Ultrasonography (US) with Doppler studies should be performed in the first 24-48 hours after the procedure and before discharge, for gross evaluation of the liver parenchyma, biliary tree and graft vasculature.
NORMAL RADIOLOGIC FINDINGS
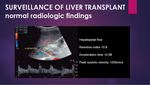
Hepatopetal flow. Sharp systolic peak < 200 cm/s with an acceleration time of around 0.08 seconds. Diastolic flow is maintained, with a resistive index between 0.5 to 0.8.
Increased RI values of more than 0.8 can be found during the early postoperative period, retunrning to normal pattern in a few days.

Fig. 2: Normal hepatic artery Doppler.
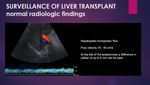
Hepatopetal monophasic flow that varies with breathing, with velocities from 16 to 40 cm/s in the immediate postoperative period. At the site of the anastomosis, a difference in caliber up to 5 mm can be seen. It is useful to evaluate PV spectrum with an intercostal approach to ensure the blood flow is hepatopetal. During the early postoperative phase, turbulent flow may be seen.

Fig. 3: Normal portal vein Doppler.
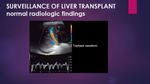
Characteristic triphasic waveform, influenced by the variations in right cardiac chambers pressure during the cardiac cycle.

Fig. 4: Normal hepatic veins Doppler.
PRINCIPAL VASCULAR COMPLICATIONS
Postsurgical changes can lead to various seemingly abnormal findings that will require close clinical and radiological follow-up.
If US findings are pathological or there is a persistent clinical suspicion for abnormalities, computed tomography (CT) is usually performed.
Contrast enhanced US can be usefull but technically difficult in the inmediate postoperative period, because the exams are usually performed in intensive care units. Also, if pathologic, surgeons will still ask for further CT evaluation. The role of MRI in these patients is limitated because their incapacity to collaborate and the duration of the test.
Vascular complications usually occur at the site of the anastomosis.
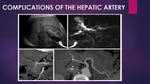
HEPATIC ARTERY COMPLICATIONS
Complications involving HA are the most common complications after a liver transplant.
Is the most common complication of a liver transplant. Its incidence is estimated to be 4-12% and usually occurs within the first 15 postoperative days.
Risk factors are increased cold ischemic time of the donor liver, ABO blood type incompatibility, small donor or recipient vessels, and acute rejection.
Unfortunately, HA thrombosis is also the second cause of graft failure in the early postoperative period. The biliary tree in a liver transplant is supplied exclusively by the HA, so hepatic artery thrombosis may lead to biliary ischemia and necrosis.
Therefore, early diagnosis and intervention is needed. HA thrombosis can be treated with thrombectomy, hepatic artery reconstruction. Nevertheless, many patients need to undergo retransplantation.
HA thrombosis is suspected when there is absence flow in HA and intrahepatic arteries on color Doppler sonography and is confirmed by CT-angiography that shows an abrupt cutoff of the hepatic artery usually at the site of anastomosis. Conventional angiography is no longer needed. MRI-angiography findings correlate well with CT and is more sensible to biliary duct pathology.

Fig. 5: Hepatic artery THROMBOSIS. The most common vascular complication after liver transplant. Ischemic cholangitis seen with US (circle) and RM due to thrombosis of the HA in the anastomosis site seen in CT-angiography (arrow).
The second most common vascular complication of orthotopic liver transplantation.
HA stenosis is related with clamp injury, intimal trauma caused by perfusion catheters at the time of surgery, or disrupted vasa vasorum leading to ischemia of the arterial ends. It can progress to HA thrombosis so, again, early diagnose and prompt intervention with surgery or arteriography and balloon angioplasty is mandatory.
Increased systolic peak velocities of more than 200 cm/s at the site of the stenosis in Doppler US or tardus-parvus intrahepatic waveform are suspicious of HA stenosis. This waveform consists in an increased acceleration time of more than 0.08 s and a decreased RI of less than 0.5. When high suspicion of HA stenosis exists, a CT angiography must be performed and will show diminished caliber or filiform flow of the HA.

Fig. 6: Hepatic artery STENOSIS (circle). Two cases of HA stenosis at the anastomosis site (A) and the native segment (C) with a slightly tardus-parvus waveform in the Doppler study.
Pseudoaneurysms of the hepatic artery are rare complications after liver transplantation. They are usually mycotic but can also be a consequence of an angioplasty. Intrahepatic or peripheral hepatic artery pseudoaneurysm can be related with liver parenchyma infections or interventional procedures such as liver biopsies. They are asymptomatic, but if they break, they can cause a major arterial hemorrhage. They can also cause haemobilia or gastrointestinal bleeding if they fistulize.
On ultrasound they are seen as cystic structures adjacent to the hepatic artery that can show a turbulent flow inside visualized as ying-yang sign. These structures enhance in CT angiography or MRI after administration of intravenous contrast. They can be treated with surgery, resecting aneurysm and reconstructing the artery or with interventional techniques, embolizing or isolating it with covered stents.

Fig. 7: Hepatic artery PSEUDOANEURISM (arrow) in coronal (A), axial (B) and sagittal (C) view.
PORTAL VEIN COMPLICATIONS
Portal vein complications are rare (1-2%). They include thrombosis and stenosis.
PV thrombosis is suspected when portal hypertension, hepatic failure and massive ascites is present. It is related with vessel misalignment, differences in the caliber of the recipient and donor portal veins, previous portal vein surgery or previous thrombosis in the recipient portal vein and hypercoagulable states.
US is very sensible in the detection of PV thrombosis seen as an hyperechoic filling defect inside a PV without color Doppler signal. Acute thrombus may be anechoic. CT and MRI angiography also depict well filling defects or narrowing.

Fig. 8: Portal vein THROMBOSIS (arrows) extended through the superior mesenteric vein (arrowheads) with alteration liver perfusion areas (circles in A, B and C).

Fig. 9: Portal vein STENOSIS (arrow) in the anastomotic site. Aliasing and increased PSV (circle with arrow) in Doppler study.
HEPATIC VEINS AND INFERIOR VENA CAVA COMPLICATIONS
Post-transplant complications in HV and IVC have a low combined incidence of less than 1 % and include thrombosis and stenosis, usually at the piggyback anastomosis.
Clinical signs such as hepatomegaly, ascites and pleural effusion are suspicious.
Focal stricture with turbulent flow and increased velocities at the anastomosis are direct signs of HV stenosis. Also, a persistent monophasic waveform on Doppler US at HV is highly suspicious of HV or IVC stenosis and a CT angiography must be performed to rule out this complication.
Treatment options include balloon angioplasty or stent placement.

Fig. 10: Inferior vena cava acute THROMBOSIS. Echogenic thrombus with no flow on Doppler in the cava lumen (circle). Filling defect in the vena cava on CT in different views (circles and arrow).

Fig. 11: Hepatic veins – vena cava anastomosis STENOSIS. Monophasic doppler flow (arrowheads) at the site of the anastomosis. Stenosis of the anastomosis on CT (arrow).