1.Clinical presentation.
Presentation is most frequently unilateral, rare bilateral, either with sudden or progressive onset.
Associated findings may include edema, tenderness, skin thickening, dimpling (“peau d´orange sign”), dermal lesions and breast masses.
2. Pathologic entities:
Different lesions either arising within the breast or from adjacent tissues may cause breast enlargement.
2.1. Breast.
2.1.1. Normal variants:
• Developmental variants ( Fig. 1 ). Unilateral premature thelarche or asymmetric development of breast buds cause size asymmetry in girls which may persist in adult breasts although they tend to reach similar size during development.

Fig. 1: Normal variants
• Unilateral breast feeding leads to breast enlargement.
• Poland syndrome consists of an agenesis of pectoralis muscle associated with breast anomalies presenting with breast asymmetry.
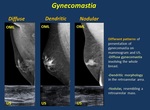
2.1.2. Gynecomastia is a benign enlargement of male breast glandular tissue ( Fig. 2 ).

Fig. 2: Gynecomastia
Idiopathic causes exceed other etiologies. Gynecomastia may be related to an imbalance in the ratio of estrogen to androgen tissue levels or end-organ responsiveness
Imaging assessment to exclude associated lesions includes mammogram (in patients older than 25 yo) and US.
2.1.3. Inflammatory-infectious conditions.
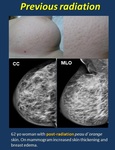
• Breast irradiation ( Fig. 3 ). Post-radiation changes are focal or diffuse depending on radiation field, typically with a linear border and non-anatomic configuration.
Mammogram. US. Both acute and chronic post-treatment changes cause increased skin thickness and breast volume. “Peau d´orange” appears acutely due to skin edema.
Radiation induces granulation tissue with contrast-enhancement on MR of the treated field at parenchyma and skin. Skin post-radiation enhancement may remain for 18 months.
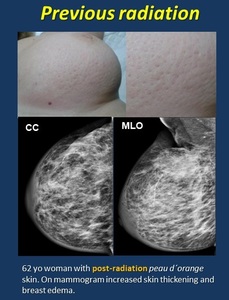
Fig. 3: Previous radiation
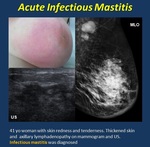
• Mastitis. ( Fig. 4 ).
Most often found in lactating women (milk stasis is retrogradely infected by bacteria), but also recurrent in non-lactating smokers or ductal ectasia. Treatment consists of antibiotics
Mammogram. Edema, trabecular thickening and focal or global asymmetry with or without skin thickening and axillary lymphadenopathy or a mass (abscess).
US . skin or breast edema.
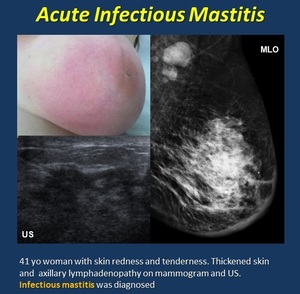
Fig. 4: Mastitis
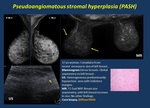
2.1.4. Pseudoangiomatous stromal hyperplasia (PASH) is a benign stromal localized mesenchymal stromal cell overgrowth.
Most frequently microscopic foci found incidentally. It occasionally presents mass appearance simulating a fibroepithelial tumor either solitary or multifocal or a diffuse massive process with asymmetry of the breast and breast enlargement, usually painless and presenting in premenopausal women ( Fig. 5 ).
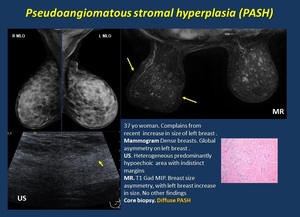
Fig. 5: Pseudoangiomatous stromal hyperplasia (PASH).
Immunohistochemistry: Positive for CD34 and PR on IHC, variable positivity for actin and desmin and negative for other endothelial markers.
Imaging findings:
• Mammogram: Circumscribed mass, focal asymmetry or no findings.
• US. Circumscribed mass, non-circumscribed hypoechoic area or no findings
• MR. Focal or segmental clumped non-mass enhancement with persistent or plateau kinetics, circumscribed mass or no findings
2.1.5. Benign tumors.
PASH, Juvenile fibroadenoma and Phyllodes tumor are rapidly growing masses and sometimes clinical perception is breast enlargement.
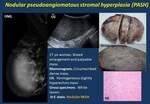
· Nodular PASH ( Fig. 6 ). Circumscribed masses on mammogram, US and MR.
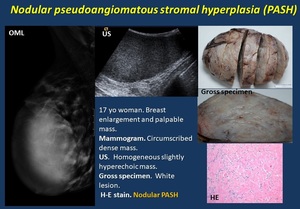
Fig. 6: Nodular pseudoangiomatous stromal hyperplasia (PASH).
· Phyllodes tumor. ( Fig. 7 ).
Benign and malignant phyllodes tumors are biphasic lesions, with different types of specialized or non-specialized stroma. Differential diagnosis with fibroadenoma is difficult on core biopsy.
Imaging:
• Mammogram. Circumscribed mass.
• US. Smooth margins and heterogeneous mass sometimes with cystic areas.
• MR. Circumscribed mass, contrast enhancement. Hyperintense T1 lesions are more frequently malignant. Iso and hypointense T2 and low ADC correlates with stromal hypercellularity.
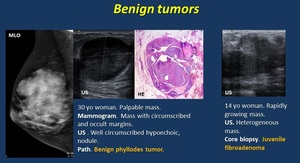
Fig. 7: Benign tumors
• Juvenile fibroadenoma.
Rapidly growing mass in young women or children.
Mammogram. US. Circumscribed mass with cystic areas that tend to be smaller and less frequent than Phyllodes tumors.
2.1.6. Malignant Tumors.
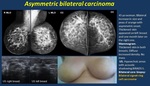
Breast cancer in young women frequently presents with higher histology grade, positive axillary nodes, hormone receptor negativity, and higher p53 and Ki-67 expression, which causes rapidly growing masses, and breast enlargement in diffuse disease ( Fig. 8 ).
Huge malignant masses are frequently caused by tumors such as sarcoma, malignant phyllodes tumor, metaplastic carcinoma, inflammatory carcinoma and lymphoma.
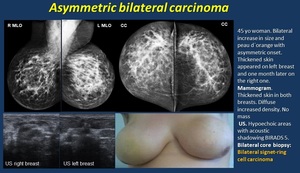
Fig. 8: Asymmetric bilateral carcinoma
• Metaplastic carcinoma ( Fig. 9 ). Large circumscribed masses, typically ER, PR, HER-2 negative. Lymph node metastases are rare, more frequent hematogenous.
Mammogram. Round or oval circumscribed-masses. Rare plemorphic, linear microcalcifications.
US. oval, round hypoechoic mass with circumscribed or indistinct margins and posterior acoustic enhancement,
MR-T2 hyperintense circumscribed mass. Contrast uptake, with curves similar to infiltrating carcinoma.
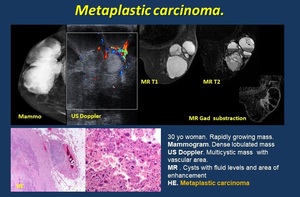
Fig. 9: Metaplastic carcinoma
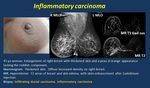
• Inflammatory breast carcinoma ( Fig. 10 ). Advanced breast cancer with increased breast size, edema and inflammatory signs. Pathological hallmark is tumor embolization: dermal lymphatic invasion with neoplastic cell emboli within the epidermal and subdermal lymphovascular drainage system.
Mammogram. Increased breast size with edema. Skin thickening. Stromal trabecular coarsening and increased density.
US. Skin thickening (more than 3 mm). Anechoic tubular dilated lymphatic channels in the subdermal fat. Parenchymal hypoechoic areas and shadowing.
MR T2. Thickened hyperintense skin.
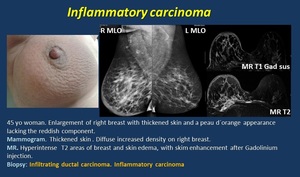
Fig. 10: Inflammatory carcinoma
Diagnostic criteria for inflammatory carcinoma Table 2
· Breast lymphoma ( Fig. 11 ).
Primary (most B-cell) or secondary lymphoma (secondary involvement of breast tissue by a systemic lymphoma/leukemia).
Mammogram. Focal or diffuse increased density with or without skin thickening and breast enlargement. Solitary round or oval mass with circumscribed margins (most common), spiculated or irregular margins, architectural distortion or focal asymmetry.
US. Hypoechoic round masses with variable transmission sometimes ill-defined or hyperechoic. Diffuse thickening.
MR. T2-weighted hyperintense, T1-weighted isointense masses. Homogeneous rapid initial and mixed delayed enhancement.

Fig. 11: Breast primary lymphoma
· Sarcoma.
Breast parenchyma primary angiosarcoma ( Fig. 12 ) or secondary (parenchyma, skin or chest wall complication of irradiation).
Huge rapidly-growing masses or diffuse infiltration and breast enlargement.
Mammogram. Focal asymmetry or huge masses.
US. Hypoechoic areas. Solid highly-vascular masses.

Fig. 12: Angiosarcoma
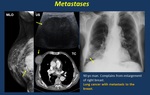
• Metastases.
Metastases from different organs may cause breast enlargement. Usually multiple sometimes isolated masses ( Fig. 13 ).

Fig. 13: Metastases
2.1.7. Breast augmentation
· Foreign-substances free injections.
Free substance injection including silicone ( Fig. 14 ), hyaluronic acid ( Fig. 15 ) causes enlargement with inflammatory reaction or palpable lumps.
Foreign-materials are evident on mammograms/US, obscuring parenchyma.
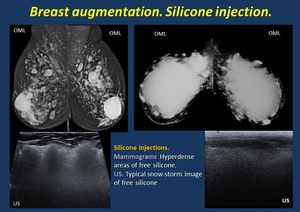
Fig. 14: Silicone injection

Fig. 15: Hyaluronic acid
• Prosthesis:
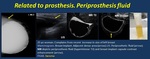
Periprosthetic fluid ( Fig. 16 ).
- Breast implant-associated anaplastic large cell lymphoma.
T-cell lymphoma arising around breast implants. Presentation 8-10 years after implant placement.
Most frequent peri-prosthetic fluid, less frequent masses.
TIP. Periprosthesis fluid collections of recent onset should undergo fine-needle-aspirationd analyzing cytology, immunohistochemistry and flow cytometry (T-cell markers).

Fig. 16: Periprosthesis fluid
-Seroma or infection cause peri-prosthetic fluid.
Implant rupture ( Fig. 17 ).
-Extracapsular implant rupture. Tear of implant shell and fibrous capsule. Mammograms, US, MR: free silicone .
-Intracapsular implant rupture. MR: linguine sign. US. Heterogeneous implant.

Fig. 17: Prosthesis rupture
2.2. Breast edema of non-mammary origin.
Systemic entities may cause acute breast enlargement.
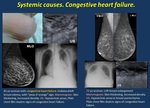
· Congestive heart failure ( Fig. 18 ).
Diffuse edema usually bilateral, sometimes unilateral or asymmetric in patients lying lateral decubitus.

Fig. 18: Congestive heart failure
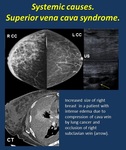
· Superior vena cava syndrome ( Fig. 19 ). Upper thorax edema including breasts.

Fig. 19: SVC Syndrome
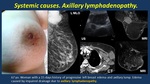
· Axillary lymphadenopathy ( Fig. 20 ) with impairment in lymphatic drainage.
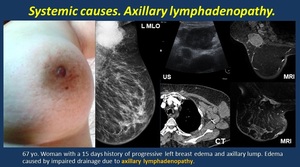
Fig. 20: Axillary lynphadenopathy
· Dermatosis: scleroderma and angioedema ( Fig. 21 ). Skin edema and increased breast size.

Fig. 21: Dermal lesions
2.2. Adjacent organs.
Pectoral major lesions including hematoma, benign (lipoma Fig. 22 ) and malignant tumors (sarcoma Fig. 23 ) simulate breast enlargement.
Properly-positioned mammograms, US and MR depict lesion location.

Fig. 22: Pectoralis muscle lipoma

Fig. 23: Pectoralis muscle liposarcoma
3. Diagnostic work-up.
The proposed diagnosic work-up for breast enlargement is presented on Fig. 24 . Keys for differential diagnosis on Table 3 .
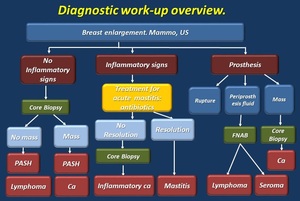
Fig. 24: Diagnostic work-up

Table 3