Spinal dural arteriovenous fisulas are the most common type of spinal vascular malformation, accounting for approximately 70% (Fig. 1).
They constitute a rare and enigmatic entity. Clinical features and structural changes were firstly recognized in 1926, pathophysiology and basics of treatment were known since 1974, and yet it remains unknown why these fistulas develop.
Epidemiology:
Most patients are middle or elderly men, which suggests an acquired origin, being trauma postulated as a potential cause.
Fig. 2

Fig. 2: Epidemiology of spinal dural arteriovenous fistulas and simplified classification of spinal vascular malformations.
Clinical manifestations:
Symptoms as well as radiologic features are related to the underlying physiopathology (Fig. 3).
Fig. 3

Fig. 3: Comprehensive review of symptomatology (which is based on the underlying physiopathology of SDAVF)
At the onset of the disease symptoms are nonspecific, often mimicking other causes of neurogenic disorders such as demyelinating disease, central canal stenosis or neoplasms (Fig. 14). This leads to a delay in diagnosis (ranging from 6- 59 months) with often permanent neurological deficits and disability.
Patients present myelopathy with motor and sensory symptoms ascending from the feet, initially fluctuating that slowly progresses over several years becoming eventually permanent. Another clinical hallmark is that symptoms can worsen after prolonged standing, exercise or Valsalva maenuvers. Sphincter dysfunction can occur.
Radicular and nonradicular back pain is also frequently encountered.
Involvement of the arms is rare, and occurs only in cervical fistulas (Fig. 12).
Acute headache due to subarachnoid hemorrhage can occur, mostly in patients with an infrequent dural fistula located at the craniocervical junction.
Physiopathology:
To fully understand physiopathology of spinal dural arteriovenous fistulas, a basic comprehensive review of spinal cord blood supply is mandatory.
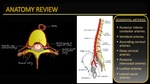
Arterial supply ( Fig. 4 ):
The spinal cord is supplied by a single anterior artery (arising at the level of the foramen magnum from vertebral arteries anastomosis) and two posterior spinal arteries (arising from either the posterior inferior cerebellar arteries or from V3/V4 segments of the vertebral arteries). These three arteries are interconnected by a pial arterial plexus (arterial vasocorona) and join inferiorly to form the cruciate anastomosis of the conus medullaris.
This supply is also reinforced at multiple levels by the radicular arteries, which originate from the posterior branches of the paired segmental arteries as they enter the dura.
Segmental arteries arise in the cervical region from the posterior inferior cerebellar arteries, the vertebral arteries as well as from branches of the ascending and deep cervical arteries. In the thoracolumbar region they arise from the intercostal and lumbar arteries. In the sacral spine they may arise from the middle or lateral sacral branches and iliolumbar arteries.
Knowledge of these segmental arteries is important because they must be assessed angiographically in order to locate the fistula.
FFig. 4

Fig. 4: Anatomy review indicating spinal cord arterial blood supply, emphasizing segmental arteries. Segmental arteries supply the spine (including the vertebral bodies, paraspinal muscles dura and nerve roots ) and the spinal cord. The dominant segmental artery in the thoracolumbar region is the artery of Adamkievicz (arising usually on the left side at the level of T9-T12).
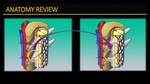
Venous drainage ( Fig. 5 ):
The cord drains through circumferential radial veins which drain into a pial venous network. Then it drains into longitudinal anastomosing veins, then into radicular veins which pierce the dura while following the exiting nerve root to reach the epidural plexus. Spinal cord venous drainage is valveless with a reflux-impending mechanism at the narrowing dural penetration.

Fig. 5: Comparative cord arterial blood supply (A) and venous drainage (B). Spinal cord venous drainage is valveless with anti-reflux mechanism at the narrowing dural penetration at the root sleeve
Spinal dural fistulas form when there is abnormal direct connection between a radicular artery and the corresponding radicular vein within the dural root sleeve ( Fig. 5 Fig. 6 ).

Fig. 6: Fistulas develop due to abnormal direct connection between a radicular artery and the corresponding radicular vein within the dural root sleeve.
This leads to a higher pressure in the radicular vein, which produces dilatation of the pial venous plexus causing congestion of the venous outflow. This leads to intramedullary edema causing chronic hypoxia and ischemia with progressive myelopathy (Fig. 6 Fig. 7 ). Long-term ischemia can lead to eventual cord infarction.

Fig. 7: Increased flow within the radicular vein leads to venous congestion causing cord edema and ischemia. Long-term ischemia can eventually lead to cord infarction.
Because the lower thoracic region has relatively less venous outflow channels relatively to the cervical region, the venous congestive cord edema is more likely to affect the most caudal part of the cord: the conus medularis. Thus, first symptoms reflect dysfunction of the conus, although the shunt is located remotely. This also explains why prolonged standing can worsen symptomatology.
The fact that the vascular pressure of the fistula can be as high as 74% of systemic arterial pressure can explain why some patients experiment worsening of symptoms during exercise, when there is systemic increase in arterial pressure.