Indications for Bowel Transplantation (Fig. 2, Fig. 3)
1) Intestinal failure due to short bowel syndrome (SBS) complicated by loss of vascular access or catheter sepsis
- SBS causes: Gastroschisis, necrotizing enterocolitis (NEC), volvulus, intestinal atresia
2) Total parenteral nutrition (TPN) induced end stage liver disease (ESLD)
3) Locally invasive malignancies such as desmoid tumors within the abdomen
4) Premalignant conditions (e.g., Gardner syndrome)
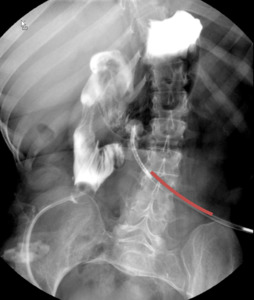
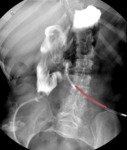
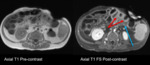
Fig. 2: UGI: Short gut after bowel resection with duodenostomy tube in place (red line)
References: Department of Radiology, UCLA Ronald Reagan Medical Center, 2019.
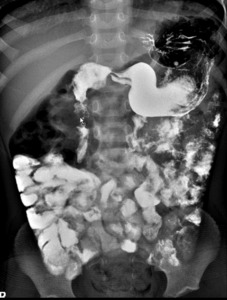
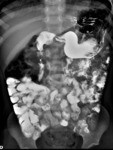
Fig. 3: UGI: Normal length small and large bowel
References: Department of Radiology, UCLA Ronald Reagan Medical Center, 2019.
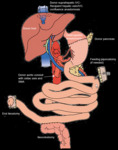
Anatomy: Multivisceral Transplant (Fig. 4)
MVTx is an effective way to treat children with SBS and irreversible liver failure from long-standing TPN. In addition to the intestines, the liver and pancreas are also harvested, allowing for an intact biliary system. Donor distal stomach is anastomosed to the recipient proximal stomach. One or both kidneys may also be transplanted at the same time if needed.
Anatomy:
Donor intestine, liver, pancreas, and distal stomach transplant
Donor aortic conduit with celiac axis and SMA attached, anastomosed to recipient aorta
Donor suprahepatic IVC-Recipient hepatic vein/IVC confluence anastomosis
Gastrogastric anastomosis
Pyloromyotomy (to prevent gastric outlet obstruction from vagal denervation)
End ileostomy (for easy access to mucosal biopsies and endoscopy)
Ileocolostomy (if patient has remaining colon)
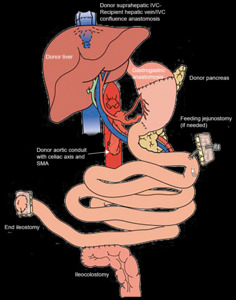
Fig. 4: Diagram: Multivisceral Transplant
References: Adapted from: Fink MP, Vincent JL, Abraham E, Moore FA, Kochanek P (2017) Textbook of critical care. 7/E. edn. Elsevier, Philadelphia, PA
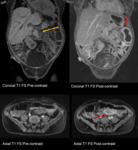
Infection: Adenovirus enteritis (Fig. 5)
High dose immunosuppression required by MVTx patients increases the risk for developing opportunistic infections. The increased risk compared to other transplant types may be due to the high dosage requirements and the microbial load inherent within the graft.
Viruses cause 2/3 of graft infections and can include cytomegalovirus (CMV), Epstein-Barr virus (EBV), and adenovirus. CMV is the most serious, since it can result in graft loss and death. EBV can cause post-transplant lymphoproliferative disease (PTLD) in up to 12.5% of patients.
Bowel wall thickening is an expected finding in the first 3 months, with persistence beyond this period suggesting pathology. Infectious enteritis can mimic rejection and the diagnosis is confirmed using endoscopic biopsy.
Imaging Features:
- thickened, enhancing bowel wall
- mesenteric fat stranding
- absence of ascites (in contrast to acute rejection)
- intra-abdominal abscesses (not shown)
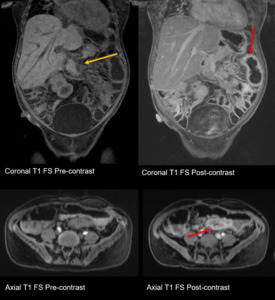
Fig. 5: Adenovirus Enteritis: Thickened, enhancing bowel wall (red arrow), mesenteric fat stranding (orange arrow), note absence of ascites (in contrast to acute rejection).
References: Department of Radiology, UCLA Ronald Reagan Medical Center, 2019.
Rejection: Acute (Fig. 6, Fig. 7)
Acute rejection is the most common cause of graft loss in the first 2 months after MVTx, occurring in up to 50-75% of patients. Caused by the onset of the cellular immunity response, it can occur anytime from 1 week post-transplant to months or even years later.
Acute rejection is diagnosed using tissue samples from endoscopic biopsies (via the ileostomy), which are performed on a regular basis following transplantation. Nevertheless, CT and MR can provide clues to the diagnosis.
An acute rejection episode can be successfully treated with immunosuppression, so prompt diagnosis can help salvage the graft. Recurrent episodes can lead to chronic rejection.
Imaging findings:
- thickened, enhancing bowel wall (focal or diffuse)
- mucosal hyperenhancement
- ascites/mesenteric edema
**findings may be similar to infection**
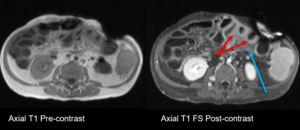
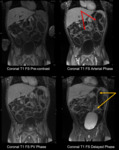
Fig. 6: Rejection, acute (MR): Thickened, enhancing bowel wall (red arrows), ascites/mesenteric edema (blue arrows)
References: Department of Radiology, UCLA Ronald Reagan Medical Center, 2019.
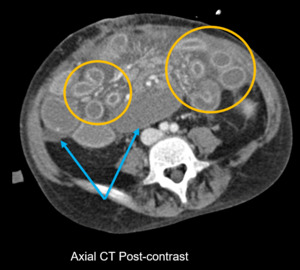
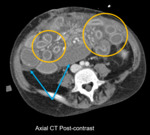
Fig. 7: Rejection, acute (CT): Mucosal hyperenhancement (orange circles), ascites/mesenteric edema (blue arrows)
References: Department of Radiology, UCLA Ronald Reagan Medical Center, 2019.
Rejection: Chronic (Fig. 8)
Chronic rejection is the main cause of late-stage graft dysfunction. Patients with chronic rejection lack specific symptoms and typically have a prolonged, vague illness course. Full-thickness biopsies are required to show non-specific fibrotic changes in the organ and vessels.
Recognition of chronic rejection is important to allow proper clinical and surgical planning for possible re-transplantation in the future, which is the only treatment.
Imaging features:
- featureless segments of bowel (loss of folds: valvulae conniventes/haustra)
- Persistent mild bowel wall hyper-enhancement (delayed enhancement, related to fibrosis)
Not shown, but may be found:
- biliary strictures and stenosis, paucity of ducts
- beaded appearance of arteries
- sclerosing encapsulating peritonitis (cocoon abdomen)
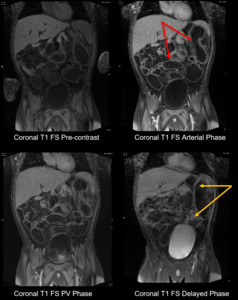
Fig. 8: Rejection, chronic: Featureless segments of bowel (red arrows), persistent mild bowel wall hyper-enhancement (orange arrows).
References: Department of Radiology, UCLA Ronald Reagan Medical Center, 2019.
Motility: Strictures in the Setting of Chronic Rejection (Fig. 9)
Bowel strictures in MVTx patients can be due to a variety of causes including chronic rejection, where there is gradual fibrosis of the transplanted structures and feeding blood vessels.
Other causes can include extensive adhesions or post-transplant lymphoproliferative disease (PTLD). In the case of the latter, infiltrative disease may circumferentially thicken the bowel wall and cause stenosis.
Imaging findings:
- short or long segment of luminal narrowing
- mural thickening
- upstream bowel dilatation
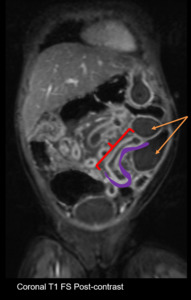
Fig. 9: Motility, strictures: Short or long segment of luminal narrowing (red), mural thickening (purple), upstream bowel dilatation (orange arrows).
References: Department of Radiology, UCLA Ronald Reagan Medical Center, 2019.
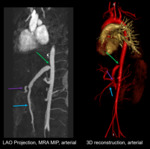
Vascular: Aortic Conduit Stenosis (Fig. 10)
For a MVTx, an aortic conduit comprised of the donor celiac axis and SMA is anastomosed to the recipient aorta. Conduit stenosis is typically an early complication. It can result in vascular insuffciency, thrombosis, and failure of the transplanted organs.
Some risk factors include faulty surgical technique, clamp injury, catheter-induced trauma, and rejection.
Imaging findings:
-reduced caliber of aortic conduit, typically at the site of anastomosis to the recipient aorta
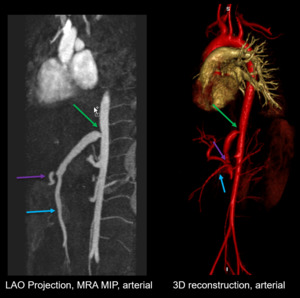
Fig. 10: Vascular, Aortic Conduit Stenosis: Reduced caliber of aortic conduit (green arrow), celiac axis (purple arrow), SMA (blue arrow).
References: Department of Radiology, UCLA Ronald Reagan Medical Center, 2019.
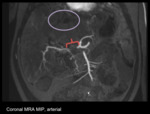
Vascular: Hepatic artery thrombosis (Fig. 11)
For a MVTx, the most serious vascular complication is arterial or venous graft thrombosis. Arterial thrombosis most commonly occurs within the first 3 months following transplantation. The risk of arterial thrombosis increases with decreased donor arterial caliber.
Imaging findings:
-filling defect in the occluded segment
-heterogeneous parenchyma of the affected organ, may be related to infarct or perfusion changes
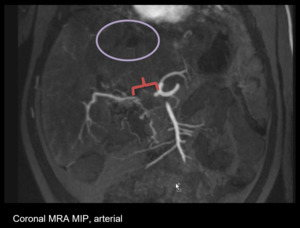
Fig. 11: Vascular, Hepatic artery thrombosis: Filling defect in the occluded segment (red bracket), heterogeneous parenchyma of the affected organ (purple oval).
References: Department of Radiology, UCLA Ronald Reagan Medical Center, 2019.
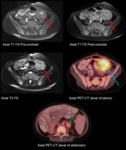
Malignancy: PTLD (Fig. 12)
Post-transplant lymphoproliferative disorder (PTLD) is a lymphoid tumor usually arising from recipient B-lymphocytes and caused by the EBV.
PTLD is more common in intestinal and MVTx patients than in other solid organ transplant patients. The disease may regress with a reduction in immunosupression, which differentiates it from other malignancies.
Imaging features:
- can occur in many extra-nodal sites including lung, bowel, bone, and brain.
- lymphadenopathy
- mass-like bowel thickening with enhancement
- lesions have increased metabolic activity on FDG-PET
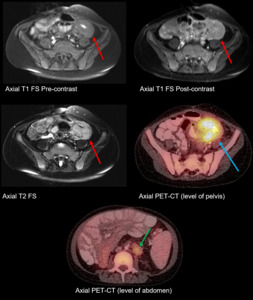
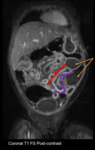
Fig. 12: Malignancy, PTLD (MR, PET-CT): Mass-like bowel thickening with enhancement (red arrows), increased metabolic activity on FDG-PET (blue arrow), hypermetabolic lymphadenopathy (green arrow).
References: Department of Radiology, UCLA Ronald Reagan Medical Center, 2019.