1. Healthy liver
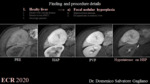
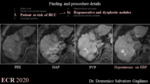
1a. Focal nodular hyperplasia (fig.1)
FNH usually occurs in relatively young women [10]. FNH is defined as a nodule composed of benign-appearing hepatocytes occurring in a liver that is otherwise histologically normal or nearly normal [11]. As reported by Grazioli et al [12], 91% of FNHs show iso- or hyperintensity on the HBP likely related to the equal or stronger OATP1B3 expression compared to background liver [13, 14] due to the activation of glutamine synthetase which is one of the major downstream targets of ß-catenin [15].
The remaining 9% of FNHs show hypointensity in the central parts of the lesion and peripheral hyperintensity giving the typical appearance of ring or doughnut-like enhancement on HBP [12-14]. The management of FNH is usually conservative [16].
1b. Hepatocellular adenoma
HCA is an uncommon benign neoplasm more frequently detected in young women with history of oral contraceptive assumption and 93% shows hypointensity on HBP [12, 17]. HCAs can be divided into eight subtypes according to their molecular/genetic and clinical features [18]. Among these, β-catenin–activated HCA own the highest risk of malignant transformation (OR 9.3), and most commonly shows iso or hyperintensity on HBP, as opposed to other subtypes [18-20]. Activation of β-catenin protein causes uncontrolled hepatocyte proliferation and overexpression of OATP1B3 responsible of iso/hyperintensity on HBP [21].
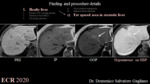
1c. Fat spared area in steatotic liver (fig. 2)
Areas of fat sparing in steatotic liver are usually located at segment II, caudate lobe or adjacent to gallbladder. Fat spared areas can be easily diagnosed through in- and opposed-phase MR images, and may appear hyperintense on HBP due to preserved or even increased parenchymal function [22].
2. Patients with vascular disease
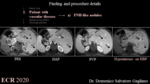
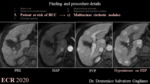
2a. FNH-Like Nodules (fig.3)
FNH-like nodules arise as a local hyperplastic response to vascular alterations, such as decreased portal venous blood flow and increased hepatic arterial blood flow and may have MRI characteristics similar to hypervascular hyperplastic nodules [23, 24]. On HBP, FNH-like nodules may show increased uptake in the periphery of the nodule (doughnut-like appearance) with a central stellate hypointense scar, as seen in FNH [23].
3. Patients at-risk of HCC
Patients at-risk of HCC require periodic surveillance for HCC screening.
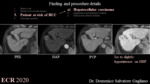
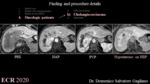
3a. Hepatocellular carcinoma (fig. 4)
Approximately 80% of cases of HCC develop in patients at-risk for HCC [25]. About 12-15% of HCCs show contrast uptake on HBP. This uptake is correlated with maintenance of hepatocyte function with upregulation of OATP1B3 and HNF 4α expression, and usually indicates good prognosis [26].
HBP uptake correlates with the presence of a greenish area at pathology due to production of bile by HCC (i.e. green HCC). Indeed, green HCCs overexpress OATP1B3 that causes bilirubin and hepatobiliary contrast uptake [27]. However, the presence of intratumoral gadoxetic acid uptake is not related to tumor differentiation [27]. Peritumoral hyperintensity of HCC on HBP may indicate peritumoral hyperplasia with glutamine synthetase and OATP1B3 expression and a higher incidence of microscopic hepatic venous invasion [28].
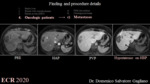
3b. Regenerative and dysplastic nodules (fig. 5)
Cirrhosis-associated regenerative nodules are innumerable well-defined rounded regions of the cirrhotic parenchyma surrounded by scar tissue and typically measuring 1-15 mm in diameter [11].
Regenerative nodules are made of hyperplastic hepatocytes that often contain ductular proliferation [29]. Regenerative nodules usually show hyperintensity on HBP.
Dysplastic nodules are observed in up to 25% of cirrhotic livers [30]. Low- and high-grade dysplastic nodules commonly show iso-/hyperintensity relative to the surrounding liver in the HBP due to higher OATP1B3 expression, but one-third of high-grade dysplastic nodules may be faintly hypointense [7].
3c. Multiacinar cirrhotic nodules (fig. 6)
Monoacinar or multiacinar regenerative nodules are well-defined regions of parenchyma that are enlarged in response to necrosis, altered circulation, or other stimuli. They may contain one (monoacinar) or multiple (multiacinar) portal tracts. The diameter of monoacinar nodules is usually 0.1–10 mm, while large multiacinar nodules are usually 5–15 mm in diameter [11].
On HBP multiacinar cirrhotic nodules usually appear as doughnut-like hyperintense nodules [31].
4. Oncologic patients
4a. FNH-Like Nodules
FNH-like nodules can occur de novo after treatment with oxaliplatin mimicking metastases at imaging, usually after a mean interval of 47.6 months after treatment [32]. The causative mechanism is considered sinusoidal obstruction syndrome [32,33].
FNH-like nodules are usually hyper- or isointense to the surrounding liver parenchyma on HBP, and a ring (or doughnut-like) pattern on HBP is observed in approximately 50% cases [32].
4b. Cholangicarcinoma (fig. 7)
Cholangiocarcinoma is the second most common primary malignancy of the liver [34].
On HBP almost all mass-forming cholangiocarcinomas show a diffuse, mainly central and inhomogeneous enhancement (called cloud) and peripheral hypointense rim [35].
This target pattern is thought to occur due to rich cellularity and vascularity in the periphery of the tumor as compared with the fibrotic center of the tumor that retains contrast agent in the extracellular space in the HBP [36].
4c. Metastases (fig. 8)
Metastases are the most common malignant liver lesions and are about 18-40 times more common that primary liver tumors [37]. Liver metastases usually originate from primary tumor of colon, breast, lung, pancreas, or stomach.
Liver metastases are usually hypovascular and colon adenocarcinoma is the most frequent source of these metastases. Hypervascular metastases typically originate from primary neuroendocrine tumors, renal cell carcinoma, thyroid carcinoma, choriocarcinoma, melanoma, and sarcomas.
On HBP, metastases may show irregular rim-like hyperintensity – which be related to a peritumoral hepatocellular or biliary reaction or the compression of hepatocytes around the tumors – or may occasionally demonstrate central areas of hepatobiliary hyperintensity due to slow accumulation of the contrast material within the intercellular matrix of the tumor (e.g. metastases from adenocarcinoma) [23].