Aortic dissection
AD represents a tear in the aortic wall, subsequent separation of the layers of the aorta with the formation of two distinct flow channels.
The risk factors include conditions that increase the stress on the wall (hypertension), and diseases with aortic medial abnormalities (connective tissue disorders, atherosclerosis, vasculitides, etc).
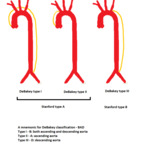
There are two main classifications used in the clinical practice - Stanford and DeBakey classification. (fig. 2) The Stanford classification is the preferred one because of its simplicity - type A AD affects the ascending aorta and requires emergent surgical treatment. Type B AD does not involve ascending aorta and requires conservative treatment with antihypertensive agents.
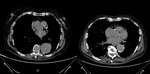
An unenhanced CT scan could show displaced intimal calcifications if present. (fig. 3)
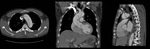
During CTA, one can identify the intimal tears and the intimomedial flap. (fig. 4-13)
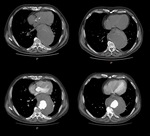
In the report, we should mention the place and the size of the entry and reentry, the luminal origins of branch vessels, the diameters of the true and false lumen, and some complications like mesenteric and renal hypoperfusion, aortic insufficiency, and coronary occlusions (fig. 14-21).
The continuity of the true lumen with the unaffected part of the aorta is the most used method to distinguish the true from the false lumen. If a lumen ends in a blind sac, it is the false one. The true lumen is usually smaller and surrounded by calcifications. Nevertheless, in long-standing aortic dissections, calcifications could be found in the outer wall of the false lumen as well. If there are three lumens, the inner one is the true lumen. The beak sign is easily detectable and presents the acute angle between the outer wall of the false lumen and the intimal flap. (fig. 22) Cobwebs (media-remnants - linear filling defects, attached to the aortic wall) and thrombosis are also indicative of a false lumen [1,3] (fig. 23-26).
The treatment for type A aortic dissection is surgical, for type B - medical. Thoracic endovascular aortic repair (TEVAR) can prevent the enlargement of the false lumen. (fig. 27-28)
Some pitfalls in the diagnosis of aortic dissection are the motion artifacts from the heart pulsation, which may simulate aortic dissection in the ascending aorta. (fig. 29) For this reason, the use of ECG synchronization is mandatory. The streak artifacts from the contrast in the left brachiocephalic vein could obscure or mimic an intimal flap in the supra-aortic branches. (fig. 30) That’s why the contrast injection should occur through a vein in the right arm [5].
Differential diagnosis of chronic AD: aortic aneurysm with mural thrombosis. If there are displaced calcifications, the diagnosis of AD is more likely. Furthermore, the false lumen usually has a spiral course. The uneven borders of the hypodense thrombotic material suggest aneurysm with thrombosis. (fig. 31)
The incomplete or subtle dissection is a limited intimal tear - a focal eccentric bulge of the aortic wall without separation of the aortic media and formation of a false lumen. It can be accompanied by subadventitial hemorrhage, which can be very difficult to differentiate from intramural hematoma with ulcer-like projection. [4]
Intramural hematoma
The classic concept is that IMH is due to spontaneous bleeding from vasa vasorum into the media, and there is no intimal tear and flap. However, there are some data about an entry tear but not re-entry one.
IMH appears as a crescent-shaped hyperattenuating thickening (60±15 HU) of the aortic wall on the native scanning, which does not enhance on the post-contrast phase. The hyperdensity is better appreciated on thicker slices using a narrow window. (fig. 32-33) There are various outcomes of IMH – resolution, enlargement, and progression to dissection or aneurysm. (fig. 34-36)
In the radiology report, one should include the Stanford classification, the maximum diameter of the IMH and the aorta, the involvement of side branches, e.g. coronary arteries, the extension to the pulmonary trunk, the presence of hemopericardium, pleural effusions, and focal contrast enhancement. (fig. 37-38) The latter includes ulcer-like projection and intramural blood pool. It is paramount to distinguish these two entities because the ulcer-like projection is related to a higher risk of progression to dissection or rupture while the intramural blood pool is not.
The ulcer-like projection or the primary intimal tear is a small zone of contrast enhancement from the aortic lumen to the intramural hematoma with a broad neck. It is believed to represent a new intimal tear. The diameter of 10-20 mm and depth 5-10 mm carries a higher risk of progression to dissection, aneurysm, or rupture. (fig. 39)
The intramural blood pool does not have a connection with the aortic lumen or it has a small one. It is reported to be a tear or pseudoaneurysm of small aortic branches, e.g. the intercostal arteries. (fig. 40)
Differential diagnosis: aortitis, contained rupture and intraluminal thrombosis. In aortitis, there's a hyperdense thicker aortic wall on a non-enhanced CT scan. The inflamed tissues, however, show contrast enhancement as opposed to IMH. [2] (fig. 41-42)
Penetrating atherosclerotic ulcer
PAU is a defect at an atherosclerotic plaque, which usually involves the descending aorta. The typical imaging finding of PAU is contrast-filled out-pouching, which separates severe atherosclerotic aortic wall changes with calcifications. They are often multiple and can be cause for saccular aneurysms. Progression towards intramural hematoma or dissection could also occur. (fig. 43-44)
Main differential diagnosis: ulceration in intimal plaque contained in the intima. They have irregular internal borders, but the external aortic wall is smooth, and there is no contrast material outside the aortic lumen (fig. 45). [1,3,4]
Unstable thoracic aortic aneurysm
The thoracic aortic aneurysms are dilatations of the aorta by more than 150%. Usually, they are asymptomatic, but the rapid growth could cause symptoms that are indistinguishable from those of AD, IMH, and PAU. The radiologists should mention any sign of impending or contained rupture.
Some key imaging findings for impending rupture are focal discontinuity of the intimal wall calcification (missing calcium sign), “tangential calcium sign”, “crescent sign” - hyperattenuating zones in the thrombus, draped aorta sign, periaortic stranding. [3] (fig. 46)
Considering all the mentioned facts, the CT protocol should include a non-enhanced scan of the thoracic aorta to confirm or exclude IMH and to detect some complications of the AAS. Then a contrast-enhanced scan in the arterial phase has to be obtained from the supra-aortic branches to the common femoral arteries. The diameters of the iliac and common femoral arteries and the presence of stenoses, atherosclerotic changes, and tortuosity should be included in the report for the endovascular treatment planning. The recommendation is to reach a contrast concentration rate of at least 250 HU in the aorta. A venous phase could be added to the protocol to appreciate the contrast differences between the true and the false lumen and to distinguish thrombosis from slow flow in the false lumen. [3,5]