Diagnostic techniques
Due to its widespread availability and short acquisition time CT is the imaging modality of choice, despite having a variable sensitivity (40-90%) and specificity (33-10%).
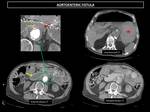
The CT protocol in our institution consists of non-contrast CT followed by contrast enhanced study on arterial, venous and delayed phases. The non-contrast study is used for detection of hyperattenuating blood content within the intestinal lumen. The arterial and venous phases are used for vascular assessment and for the detection of the site of active contrast extravasation. The delayed phase CT may be useful to reveal extravasation of contrast to the gastrointestinal lumen not shown in earlier phases Fig. 1

Fig. 1: Aortoduodenal fistula in a 80 year old male with massive hematemesis and a history of aortic graft placement 20 years earlier. Unenhanced CT showed a large hyperattenuating clot within the stomach lumen (red star). Arterial phase CT showed loss of the fat plain separating the aorta from the duodenum (red arrow) and a pseudoaneurysm formation (orange arrow). Although the site of active extravasation of contrast was not demonstrated, delayed phase CT revealed intraluminal contrast in the duodenum (blue arrow), not shown in arterial-phase CT (yellow arrow), which confirmed the presence of aortoduodenal fistula.
RM has a similar sensitivity and specificity to the CT, but its limited availability and longer acquisition time makes the RM a less adequate technique in emergency settings.
Conventional angiography is not used as a first line diagnostic technique, but may be helpful for surgical planning and for treatment.
Patients with acute gastrointestinal hemorrhage often undergo an esophagogastroduodenoscopy as the first line diagnostic technique, regardless of etiology. Although it may be a useful technique to rule out other common causes of acute gastrointestinal bleeding (peptic ulcer disease, gastric erosions, esophagitis, esophageal and gastric varices…) the sensitivity of this technique for diagnosing an aortoenteric fistula is low (50%). As a result, if there is a high suspicion of aortoenteric fistula formation, CT should be carried out first.
Radiologic signs on CT
The radiological sings that have been reported in AEF are Fig. 2 Fig. 3 :
- Ectopic gas adjacent to or within the aorta.
- Perigraft soft tissue thickening, fluid or hematoma.
- Loss of the fat plane between the aorta and the adjacent bowel wall.
- Pseudoaneurysm formation.
- Focal bowel wall thickening or retraction.
- Extravasation of aortic contrast into the intestinal lumen.

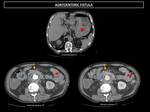
Fig. 2: Aorto-duodenal fistula in a 70 year old male who had undergone aortobifemoral graft placement 2 years earlier. Unenhanced CT showed hyperattenuating material (blood) within the stomach and the small bowel lumen (red stars), perigraft soft tissue attenuation and air (blue arrows). There is a loss of fat plane between the duodenum and the aorta (orange arrows). Although contrast extravasation to the intestinal lumen is not demonstrated, the radiological findings in a clinical context of hemorrhage are highly suggestive of aortoenteric fistula formation. As the site of active extravasation was not demonstrated, a stent graft was placed in the aortic segment adjacent to the duodenum, where most aortoenteric fistulas develop.

Fig. 3: CT of the same patient mentioned above performed after stent graft placement. Due to clinical deterioration that continued after stent graft placement, another CT scan was performed, which demonstrated the active bleeding site. Arterial phase CT showed active contrast extravasation to the duodenal lumen (blue arrows), which is a confirmatory sign of aortoduodenal fistula. Minimum intensity projection (MIP) images depicted active contrast extravasation from the aortoiliac bifurcation that extended upwards towards the duodenum (green arrow).
Most AEFs are secondary and originate from aortic graft or stent infections, which explains why many radiological signs overlap and may be present in both aortoenteric fistulas and aortic graft or stent infections without fistulization. The differential diagnosis between these entities based on imaging may be difficult . However, some signs are more prevalent with aortoenteric fistulas than with aortic graft infection without fistulizaton Fig. 4

Fig. 4
Active contrast extravasation into the intestinal lumen is a pathognomonic sign of aortoenteric fistula, but it is a rare image finding. Therefore, the absence of evident contrast extravasation in CT should not be a reason to rule out aortoenteric fistulas.
Ectopic perigraft gas and bowel wall thickening may be present in both AEF and aortic graft infections, but are more likely to be seen in AEF Fig. 5 Fig. 6.
The rest of the signs mentioned above are present with similar frequencies in AEF and aortic graft infections without fistulization. However, the demonstration of these image findings in the presence of gastrointestinal bleeding should give rise to the consideration of AEF.

Fig. 5: Aortoduodenal fistula in a 73 year old male with hematemesis and rectorrhagia who had undergone aortic graft placement 11 years earlier. Aortic phase CT revealed perigraft gas (red arrow), perigraft soft tissue attenuation and loss of the fat plane between the aorta and the duodenum. The site of active contrast extravasation was not demonstrated, but delayed phase CT showed contrast material within the duodenum and small bowel lumen (red star), not seen in the arterial phase CT (yellow star), which confirmed the diagnosis of aortoenteric fistula.

Fig. 6: Aortic stent infection in a 63 year old patient with fever and a history of aortic stent placement. CT scan showed periprosthetic air (red arrows) and soft tissue thickening adjacent to the stent-graft (orange arrow). The infected stent graft segment is in close contact with the duodenum. Although periprosthetic air is suggestive of aortoenteric fistulas, it can also be seen in prosthetic infections without fistulization.
Perigraft gas, soft tissue thickening, fluid or hematoma may be normal findings in patients who have recently undergone aortic reconstructive surgery. Persistence of ectopic gas 3-4 weeks after surgery is pathological, and may be a sign of graft infection or AEF. Soft tissue thickening, fluid or hematoma should disappear within the 2-3 months after surgery.
Differential diagnosis
The main entity to be taken into consideration in the differential diagnosis of AEF is aortic graft infection without fistulization.
Other conditions that should be considered are:
- Retroperitoneal fibrosis.
- Infected aortic aneurysms.
- Infectious aortitis.

Fig. 7: CT scan of an aortic prosthetic graft infection with perigraft soft tissue thickening (red arrow) and gas (green arrow).

Fig. 8: Aortic stent infection in a 72 year old patient with fever who had undergone aortobifemoral stent placement. CT scan revealed periprosthetic fluid collection and soft tissue attenuation in the right common iliac artery. It also showed periprosthetic air (red arrows).