SYMPTOMOLOGY
DAVF present insidiously with progressive weakness, gait disturbance and paraesthasia. Subarachnoid haemorrhage is rare but has been observed. Back pain is usually not a feature.
PATHOGENESIS
DAVFs arising at the level of the cervical cord, the craniocervical junction or skull base can cause myelopathy. In contrast to thoracolumbar DAVFs, where the feeder usually comes from radiculomedullary arteries(RMA), from our experience feeders tend to be extraspinal, involving cranial vessels. The aetiology remains the same; arterialisation of draining veins resulting in venous hypertension, ischaemia and myelopathy. Dilated, tortuoues veins can be appreciated on conventional MRI. Occasionally the arterial feeder is demonstrated on MRA.
VASCULAR NEUROANATOMY
Arterial Supply
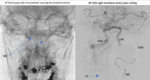
The anterior spinal artery (ASA) is formed from the V4 segments of the vertebral arteries(VA) and courses inferiorly within the anterior median aperture. Normal variants include duplicated ASA. The paired posterior spinal arteries (PSA) usually form from both V3/V4 segments and run on the posterolateral aspect of the spinal cord(Fig. 1). PSAs can be derived from PICAs. Normal variants include single PSA.
Anterior two-thirds of the cord is supplied by the ASA and the dorsal third by the PSA. A pial network forms from anastomoses between the spinal arteries to supply the cord periphery.
Spinal arteries are variably reinforced by RMA's from the vertebral, ascending cervical and/or deep cervical arteries(Fig. 2). They enter the thecal sac via the intervertebral foramina where they divide along the nerve root.
Venous Drainage
Intrinsic radial veins and a pial network drain into the anterior and posterior spinal veins (ASV and PSV respectively). The ASV is usually paired at the cervical level but can be a single vessel. The PSV can either be single/duplicated/triplicated. There are numerous anastomoses which connect the veins immediately surrounding the cord to the internal vertebral venous plexus (IVVP). Radiculomedullary veins run alongside the nerve root and drain into the IVVP. Although this is a valveless system, reflux is diminished by presence of dura. From these plexuses blood is drained into vertebral and deep cervical veins(Fig. 3). The presence of the anterior condylar confluence (ACC) allows connection between the vertebral and intracranial venous systems(Fig. 4). This is an important connection as fistulas involving intracranial venous structures cause shunting and venous hypertension in a craniocaudal direction(Fig. 12).
TIPs & TRICKS
| CORD CHANGES |
- T2 hyperintensity extending over >5 vertebrae.
- T2 hyperintensity usually homogenous with tapered margins.
- Peripheral cord sparing.
- T2 hypointense rim around cord.
|
| VESSELS |
- Perimedullary flow voids not just dorsal to the cord but also at the craniocervical junction.
- Presence of dilated, tortuous ASV/PSV.
- Presence of dilated, extraspinal veins.
|
| OTHERS |
- Variable cord enhancement
- No restricted diffusion (unless acute ischaemia)
|
CASES
Case 1:
Presentation
39 year old man presents with bilateral arm and hand paraesthesia with weakness of grip.
Initial MRI
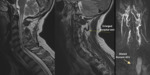
Expanded cord from C2-C6 and long segment of T2 and STIR hyperintensity with peripheral sparing. There were no perimedullary flow voids or post-contrast enhancement. Initial differentials were intramedullary neoplasm or inflammatory myelopathy. On closer inspection of the sagittal sequences, posterior to the spinous processes are large, dilated suboccipital veins(Fig. 5).
Further Imaging
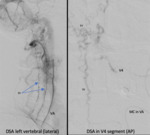
CT angiogram showed a connection between the left VA and suboccipital veins(Fig. 6). Digital Subtraction Angiography (DSA) demonstrated dilatation up to the V3/V4 junction with shunting of blood into suboccipital and spinal veins. There was also involvement of the left deep cervical artery(Fig. 7).
Treatment
Endovascular treatment with coil embolisation and WEB deployment. The left VA was occluded. The posterior circulation was fortunately supplied by the right VA and left PCOM(Fig. 8). Patient achieved full recovery.
Case 2:
Presentation
66 year old male presents with ataxia, progressive bilateral leg weakness, vomiting and urinary retention.
Initial MRI
T2 and FLAIR hyperintensity with expansion of the medulla extending into the left hemipons superiorly and cervicomedullary junction inferiorly(Fig. 9). There was no restricted diffusion or post-contrast enhancement. Initial differentials were brainstem glioma, demyelination and infarction. Infarction is unlikely because of the patients presentation and lack of restricted diffusion. You would generally not see cord expansion in subacute/chronic infarction. Similarly, with demyelination there isn’t usually cord/midbrain expansion and it is unusual to solely affect the brainstem. A second opinion was sought and on review of the post-contrast images, a dilated, tortuous anterior spinal vessel was noted. Although in the correct anatomical position for the ASA, this is too dilated and tortuous and likely represents an arterialised single ASV.
Further Imaging and Treatment
Subsequent DSA demonstrated a DAVF supplied by branches of the right ascending pharyngeal artery (APA) and middle meningeal artery (MMA)(Fig. 10). This was treated with an embolic agent and Apollo detachable tip. Unfortunately, subsequent MRIs demonstrated worsening signal abnormality extending to C6 with perimedullary flow voids(Fig. 11). Note the sparing of the cord peripheries and caudal tapering of the signal abnormality. Second DSA showed a third feeder from a dural branch of the right V3 segment(Fig. 12). MRI 6 months later showed complete resolution(Fig. 13).
CASE 3:
Presentation
78 year old made presents with headache, vomiting, left sided paresis and cerebellar signs.
Initial MRI
Long segment of homogenous T2 hyperintensity extending from the medulla to C6 which tapered caudally and spared the cord periphery. In contrast to the other cases there was restricted diffusion. Initial differentials included infarct, ischaemia and demyelination. A second opinion was sought and DAVF was queried as there were tortuous flow voids at the anterior craniocervical junction which were postulated to be arterialised condylar and superior spinal veins(Fig. 14).
Further Imaging and Treatment
Angiogram showed DAVF with feeders from a muscular branch of the left V4(Fig. 15) and C1/C2 RMA (also arising from the VA)(Fig. 16). MRI 6 months after endovascular treatment showed complete resolution.
CASE 4:
Presentation
60 year old male presents ‘off legs’ with upper motor neuron signs.
Initial MRI
Long segment of T2 hyperintensity extending from the medulla to T5 with no post-contrast enhancement or restricted diffusion. This was thought to be myelitis secondary to diffuse cervical spondylosis. MRA demonstrated a long dilated tortuous vessel anterior to the cord(Fig. 17).
Further Imaging and Treatment
DSA demonstrated DAVF with a neuromeningeal branch of the APA and branch of the MMA fistulating with condylar veins(Fig. 18). Embolic agent was used to occlude the fistula and less than a week later cord signal improved(Fig. 19). Patient went on to have a full recovery.
CASE 5:
Presentation
34 year old male with neck pain and left-sided sensory disturbance.
Initial MRI
Severe left-sided C6/C7 foraminal stenosis. Perimedullary, serpiginous flow voids were recognised which clustered around the left C6/C7 foramen. Incidentally there was no cord signal abnormality(Fig. 20).
Further Imaging and Treatment
DSA demonstrated DAVF supplied by C6/C7 RMA with involvement of the dorsal IVVP which drained caudally into the suboccipital and paravertebral veins(Fig. 21). This DAVF was considered to be silent and not contributing to patient symptomology. Patient underwent C6/C7 ACDF (no endovascular treatment). Subsequent DSA demonstrated peristent filling of the C6/C7 RMA but no early venous filling(Fig. 22). Follow up MRI showed complete resolution of the perimedullary flow voids. Patients symptoms improved and no further treatment was required.