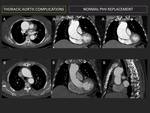
The aim when replacing heart valves is to achieve a normal function, similar to that prior to disease. PHV should adapt to the aortic root and cover the whole circumference of ascending aorta. (Fig. 2)
Complications can be divided into two groups. Herein, we describe some.
1. Haemodynamic consequences:
a- Stenosis:
Narrowing of the orifice of PHV. Certain degree of stenosis may be seen in normally functioning PHVs.
Pathologic causes include high cardiac output, prosthetic-patient mismatch and structural failure (calcification, obstruction).
At CT the orifice area can be measured with double oblique axial views using orthogonal planes.
b- Regurgitation / paravalvular leak:
Abnormal blood flow, in opposite direction to that of physiologic flow, between prosthetic ring and native annulus due to incomplete sealing. Induced by infection (most frequent cause), age related wear off, dehiscence or improper implantation.
Physiologic regurgitation may be normal. Significant regurgitation may produce heart failure or haemolytic anaemia. In these cases surgery or percutaneous closure is necessary.
At CT contrast material-filled channel in the paravalvular region is seen.
c- Stuck leaflet:
Caracterised by limited or absence of motion of a PHV leaflet due to haemodynamic or mechanical failure (space-occupying lesion).
CT evaluates opening and closing angles. Intermittent dysfunction may be missed since CT obtains data from only one cardiac cycle.
Definitive treatment requires surgical replacement.
2. Structural abnormalities:
a- Prosthetic valve-patient mismatch (PPM):
Small PHV in relation to patient’s body resulting in inadequate function, degeneration and higher mortality.
CT proves PPM and shows associated complications. The only definitive treatment is reoperation.
b- Valve calcification:
More frequent in younger patients owing to higher mechanical stress. Calcification can lead either to regurgitation or stenosis. It usually occurs at the commissural and basal region of valve cusp.
Non-enhanced-CT is the most sensitive technique; careful examination is required to distinguish calcification from structural components. Enhanced CT can also be used; HU>850 suggest calcification.
c- Dehiscence:
Separation of PHV from the annulus due to breakdown of suture. Serious complication and most common cause of early reoperation.
Some risk factors include endocarditis, ascending aortic aneurysm, regurgitation or calcification.
CT will identify a gap between the annulus and the sewing ring, with a column of contrast material showing up. The non-coronary sinus is the most frequent site.
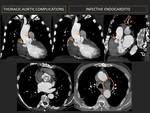
d- Infective endocarditis and abscess: (Fig. 3)
Infection can occur any time after surgery; more frequently in the first 5 years (early endocarditis 2 months).
It is diagnosed using the Duke criteria; clinical and imaging features. Early diagnosis is important as it can lead to other complications.
CT demonstrates infectious vegetation, especially large ones, as round, mobile, hypo-attenuating masses on the ventricular side of aortic valve. Other features are paravalvular leaks, wall thickening, inflammatory signs, pseudo-aneurysms, mediastinal gas, collections, extension to myocardium or pericardial fluid.
Sometimes medical treatment may be enough, others will need surgical replacement.
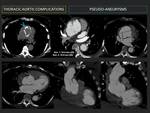
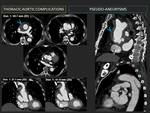
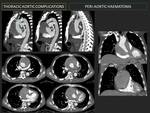
e- Pseudo-aneurysm:(Fig. 4) (Fig. 5)
Common cause for late reoperation, unless it becomes thrombosed, that appears after dehiscence of sutures.
Diagnosis is critical as their natural history includes expansion and rupture.
CT shows saccular dilatations of aorta with excellent resolution independently of contrast material; location, dimensions, necks’ size, associated thrombus; and allows surgical planning.
f- Aortic dissection:
Due to intimal tear in the aortic root, without clear association with surgery. Risk factors include wall fragility and regurgitation.
CT shows the intimal flap with true and false lumens, widening of aorta, internal displacement of intimal calcifications and ulcer-like projections.
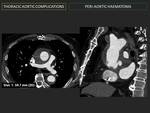
g- Periaortic haematoma: (Fig. 6) (Fig. 7)
Atypical form of aortic dissection caused by haemorrhage into the wall without intimal tear.
Non-contrast-CT manifests eccentric, high attenuation (60-70 HU) regions with inward displacement of intimal calcifications. Attenuation values will be low in relation to aortic lumen on angio-CT.
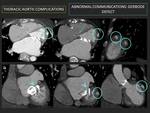
h- Abnormal communications: (Fig. 8)
Communication between two adjacent cavities caused by perforation, being the infection the most frequent cause.
Gerbode defect; left ventricle and right atrium communication; is an example.
CT shows the location of the communication between the cavities as a gap, with contrast material passing from one to another.
This complication is managed surgically.
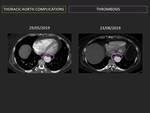
i- Thrombus: (Fig. 9)
Common complication with high mortality rates. It can occur at any time, more commonly during the first year, due to incomplete cicatrisation and endothelialisation. Incidence decreases subsequently.
CT shows irregular mobile mass without enhancement attached to PHV. It allows prediction of response to thrombolysis; complete lysis is understood with attenuation values
Treatment depends on the patients’ clinical status, size of thrombus and obstruction.
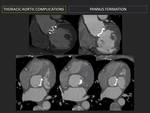
j- Pannus: (Fig. 10)
Fibrous tissue, seen more than 5 years after surgery that grows in the periannular ventricular region as a response of endocardium in normal healing process. Overproduction causes PHV dysfunction (stenosis or regurgitation). While a small amount of pannus can prevent from thrombosis, exuberant pannus favours it.
Though difficult, it is important to distinguish thrombus from pannus (Fig. 11 / Table 2).