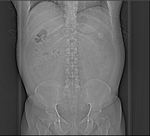
A single-energy low-dose non-contrast CT of the abdomen and pelvis was initially performed. Fig. 4 and Fig. 5
After identification of urinary tract on the raw CT data, stone targeted DECT was subsequently performed. Fig. 6 and Fig. 7
DECT was performed by targeted dual-energy scanning at two different energies of the anatomic region containing the stone and dual energy ratio for stone obtained.
Reporting renal stone on DECT should mention the below mentioned points which are required by referring surgeons to plan a holistic management strategy for the patient.
DECT differentiates between calcium-based stones and uric acid stones using dual energy (DE) ratios. DE ratio is less than 1.1 for uric acid, 1.1–1.24 for cystine, and greater than 1.24 for calcified stones. (Fig.8,Fig.9 and Fig.10)

Fig. 8: NCCT for a renal stone with DE ratio 1.44 i.e. calcium based stone

Fig. 9: DECT for a renal stone with DE ratio 1.44 i.e. calcium based stone

Fig. 10: DE ratio graph for a renal stone with DE ratio 1.44 i.e. calcium based stone
Calcium based stones include calcium oxalate monohydrate, calcium oxalate dihydrate, and calcium phosphate stones.(Fig.11,Fig.12 and Fig.13) Struvite stones have DE ratios similar to calcified stones.[3]

Fig. 11: NCCT for a renal stone of DE ratio 1.41, calcium based stone

Fig. 12: DECT for a renal stone of DE ratio 1.41, calcium based stone

Fig. 13: DE ratio graph for a renal stone of DE ratio 1.41, calcium based stone
Identifying uric acid calculi on DECT can change management. Uric acid stones can be dissolved with alkalinization of urine using oral potassium citrate solution or allopurinol. Patients with familial metabolic diseases may benefit from DECT e.g. patients with cystinuria having renal caluli of cystine composition can be treated medically using captopril.(Fig.14, Fig.15 and Fig.16) It can avoid invasive treatments such as ESWL or percutaneous nephrolithotomy (PCNL), thereby preventing potential complications like renal haemorrhage or fibrosis. Further stones composed of cystine or calcium oxalate monohydrate have a firm composition that may limit the success of ESWL.[4]
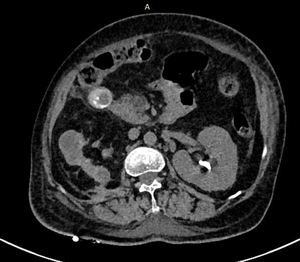
Fig. 14: NCCT of a renal calculi with DE ratio 1.22, Cystine based stone

Fig. 15: DECT of a renal calculi with DE ratio 1.22, Cystine based stone

Fig. 16: DE Ratio graph of a renal calculi with DE ratio 1.22, Cystine based stone
The simplest and most common method of assessing stone burden is measurement of stone size. Measurement of stone size helps to predict spontaneous passage of renal and ureteral stones.[1] The greatest dimension of the stone is measured to the closest millimeter at CT. (Fig.17)

Fig. 17: Measuring renal stone to assess stone burden
At CT, the internal structure can be considered to be either heterogeneous or homogeneous, needs mention. Heterogeneity in stone composition renders a stone susceptible to fragmentation with ESWL.[1] Attenuation value of stone should be reported in terms of Housfield Units.
- Number, location of calculi and determination of the presence of signs of obstruction:
Location of the stones should be reported to depict laterality,lumbar spine level the stone is at and also the calyx it is involving. Solitary and multiple calculi should be mentioned. (Fig.18 and Fig.19)

Fig. 18: Number and location of stones should be mentioned in case of multiple calculi

Fig. 19: Lower calyx stone at L2 level
Signs of obstruction like hydronephrosis, perinephric stranding, unilateral renal enlargement should be noted.(Fig.20)

Fig. 20: Dilated pelvicalyceal system with adjacent perinephric fat stranding (signs of obstruction)
- · Skin to Stone Distance:
Skin to stone distance greater than 10 cm often results in failure to achieve stone-free status following ESWL and needs a mention.[1] (Fig.21 and Fig.22)

Fig. 21: Stone to skin distance measured laterally.

Fig. 22: Stone to skin distance measured posteriorly.
Cortical thickness at the level of stone also helps to assess the success of ESWL.(Fig.23)

Fig. 23: Cortical thickness at the level of stone
- · Pre surgical planning of interventional procedures:
CT assists in the pre intervention selection of an appropriate calyx for percutaneous access and help ascertain a safe path for puncture by depicting the relationship of the kidney to various surrounding organs such as the spleen, liver, and colon.(Fig.24)

Fig. 24: Pre surgical planning on coronal reformatted CT image to ascertain safe path for percutaneous access
- · Imaging following either urologic intervention or medical therapy :
It is done essentially to:
-confirm stone-free status and identify the presence of residual stones
-rule out obstruction in the urinary system (Fig.25)

Fig. 25: Thinned out renal cortex with adjacent perinephric fat stranding.
PCNL tube seen in situ