Radiological tests used in the assessment of cerebral ischemic infarction are mainly multimodal computed tomography (CT) and magnetic resonance imaging (MRI).
MRI has high sensitivity and specificity in the early detection of cerebral ischemia, although it is currently not considered a first-choice examination in the field of hospital emergencies due to its longer acquisition time and lower availability compared to CT.
MULTIMODAL CT
The objectives of multimodal CT in patients with acute stroke are:
-
Rule out intracranial hemorrhage
-
Differentiate dead tissue (core) from potentially salvageable tissue (penumbra)
-
Identify vascular occlusion of large vessels
For this, the protocol includes the performance of a cranial CT without contrast followed by a perfusion study and CT angiography.
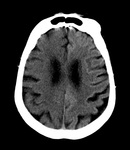
1. CT without contrast
It allows both to confirm or exclude the presence of hemorrhage and to identify early signs of ischemia (Fig. 2-3):
-
Hypodensity and loss of differentiation of the affected tissue
-
Sulcal effacement
-
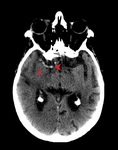
Middle cerebral artery (MCA) hyperdensity
-
Insular ribbon sign
-
Hypodensity of the lenticular nucleus
Visualization of an evident decrease in attenuation on CT without contrast is very specific for the existence of irreversible tissue damage.
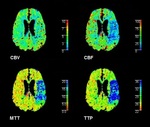
2. Perfusion study
The analysis of the increase and decrease of the signal density during the passage of the contrast provides information about brain perfusion. This is possible thanks to the different parametric maps, which allow to detect these changes through a series of variables related to cerebral blood supply: mean transit time (MTT), cerebral blood volume (CBV), cerebral blood flow (CBF) and time-to-peak (TTP).
Existence of necrosis translates into a decrease in CBV and CBF with an increase in MTT. (Fig. 4)
However, if the MTT is preserved, the perfusion alteration would correspond to ischemic penumbra. (Fig. 5)
3. CT angiography
It is a non-invasive technique that allows visualization of intracranial arteries as well as carotid and vertebral arteries, in order to detect possible filling defects in relation to stenosis or vascular occlusion.
Maximum Intensity Projection (MIP) reconstructions facilitate the visualization of these findings. (Fig. 6)
MRI SEQUENCES AND PROTOCOL
The MRI protocol commonly used in the study of ischemic infarction includes: T2-weighted turbo spin-echo (TSE T2), Fluid Attenuation Inversion Recovery (FLAIR), Diffusion-Weighted-Imaging (DWI) and ADC maps, T1-weighted turbo spin-echo (TSE T1) and MR angiography (MRA). Additionally, magnetic susceptibility sequences (SWI, T2*) can be performed.
1. DWI and ADC maps
They allow early detection of ischemic lesions since they are sensitive to the restriction of movement of extracellular water secondary to cytotoxic edema, reflecting the irreversibly damaged tissue (core).
These lesions show hyperintensity in DWI, present in the first hour and persistent for 10-14 days. In turn, they have hypointensity in the ADC map that is maximum at 2-3 days and persists for 7-10 days.
In general, hypointensity of an ischemic lesion on the ADC map indicates an evolution of the same less than one week. (Fig. 7-8)
2. FLAIR
These sequences facilitate the detection of substances with prolonged T2 by suppressing the signal intensity of the cerebrospinal fluid, which makes the areas of ischemia appear hyperintense 6-12h after the onset of symptoms due to edema. This signal intensity is maximum in chronic lesions. (Fig. 9)
3. TSE T2 sequences
It shows hyperintensity of signal in the area of the lesion after 8h of the ischemic event, which is maximal in subacute phase and subsequently persists in chronic phase. It does not allow the suppression of cerebrospinal fluid. (Fig. 10)
The three sequences described above allow 80% of ischemic strokes to be detected 24 hours after their onset.
4. TSE T1 sequences +/- intravenous contrast
In T1-weighted sequences without contrast a decrease in the signal intensity is visible at 16h after the onset of symptoms, which remains in chronic phase. (Fig. 11)
Parenchymal is the most frequent form of enhancement, appearing after the first week. If it persists more than three months it is advisable to rule out other possible underlying causes.
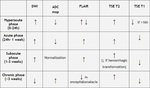
Changes in sequences referred to above during the different evolutionary phases of ischemic stroke are summarized in Table 1.
5. MRA
-
Eco-gradient sequences: Time Of Flight (TOF): based on the contrast between the signal intensity of non-stationary (blood) and stationary tissues when applying radiofrequency pulses. (Fig. 12) They allow an angiography study to be carried out without the need to administer intravenous contrast, but with the disadvantage that they can overestimate the degree of stenosis in areas of turbulent flow due to the decrease in signal intensity.
-
Intravenous contrast angiography sequences: combination of three-dimensional TOF sequences with a paramagnetic contrast medium. It creates a high resolution image of extra and intracranial vessels, being useful for detecting occlusion of large intracranial vessel, significant stenosis or arterial dissections.
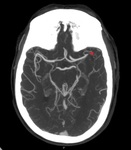
6. Susceptibility weighted imaging (SWI)
SWI is a technique based on the differences in magnetic susceptibility of various compounds (blood, iron, diamagnetic calcium).
It is highly sensitive in identification of paramagnetic substances such as hemoglobin degradation products, which show marked hypointensity, so they are especially useful for the detection of hemorrhagic transformation.
Hemorrhagic transformation usually occurs after 24-48h of the ischemic event in the form of microsangrate (Fig. 13) or parenchymal hematoma, the latter being less frequent.