After obtaining detailed history and clinical examination, patients were examined at 1.5Tesla MR and following brain MRI sequences were performed: T2 fast spin-echo, FLAIR, unenhanced T1 spin-echo and contrast-enhanced T1 spin-echo. Axial gradient-echo and diffusion-weighted were available to review. ADC maps generation and MR spectroscopy were performed.
In this exhibit, oncogenic pathways, molecular subgroups and imaging features of pediatric infratentorial tumours are presented.
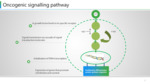
Oncogenic signalling pathway:
1. Growth factor binds to its specific receptor, resulting in transient activation of a cascade of intracellular reactions.
2. The signal is transmitted via a series of signal transduction molecules and fine-tuned by means of positive and negative regulations.
3. Gene regulatory factors are activated within the nucleus to initiate DNA transcription.
4. Gene expression promoting cell division and survival occurs.
Mutation in any of these steps may induce the development of cancer. (Figure 6)
MEDULLOBLASTOMAS:
Most common CNS embryonal neuroepithelial tumour and most common malignant solid tumour in childhood.
Histologic subtypes:
· classic
· desmoplastic/nodular,
· extensive nodularity,
· large cell/anaplastic variants.
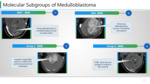
Refined into four distinct genetic subtypes:
· WNT-activated
· SHH -activated
· non WNT non SHH group 3
· non WNT non SHH group 4.
Molecular Pathways
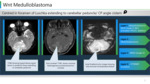
Wnt activated Medulloblastoma.—
Involves Wnt signalling pathway responsible for gene transcription regulation. Wnt protein – receptor bind to initiate a cascade of events which lead to accumulation of Bcatenin in the cytoplasm. Bcatenin translocates into the nucleus to induce gene transcription. Hallmark genetic aberrations include CTNNB1 mutation and monosomy 6.
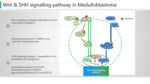
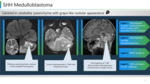
SHH activated Medulloblastoma.—
Involves SHH signalling pathway responsible for cell proliferation, survival, and angiogenesis. When PTCHD1 receptor binds with SHH ligand, the proto-oncogene SMO is released causing downstream dissociation of the GLI family transcription factors from SUFU. This results in transcriptional activation of SHH target genes. Inactivating mutations and deletions in PTCHD1, SUFU genes and genomic amplification of GLI, MYCN are recognized in SHH Medulloblastoma. (Figure 7)
non Wnt non SHH Group 3 Medulloblastoma
They harbor high-level MYC amplification including fusions with the non coding PVT1 oncogene.
non Wnt non SHH-activated Group 4 Medulloblastoma
Characterized by MYCN amplification and associated with isochromosome 17q.
Imaging Surrogates
Wnt-activated
Ø Diffusion restricting T2 hypointense signal reflecting high cellularity of medulloblastoma
Ø Cystic changes are common. Calcification/hemorrhage – rare.
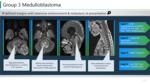
Ø Centred in foramen of Luschka extending to cerebellar peduncle/ CP angle cistern(Figure 8)
SHH medulloblastoma
Ø Mostly solid and avidly enhancing; extensive nodularity subtype show grape like appearance.
Ø More centered within the cerebellar parenchyma.(Figure 9)
non WNT non SHH medulloblastoma
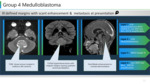
Ø Group 3 and group 4 medulloblastomas arise from a midline location involving the cerebellar vermis and fourth ventricle.
Ø Group 3 shows heterogeneous or peripheral ring enhancement.
Ø Group 4 shows relatively scant or absent enhancement.
Ø Early metastasis is common in both. Group 3 medulloblastoma show laminar or sugar coating pattern. Group 4 subtype exhibits nodular features. (Figure 10,11)
Prognosis and predictive factors
Wnt MDB demonstrate classic histologic features having excellent prognosis. SHH MDB prognosis depends on the status of Tp53 mutation. non WNT non SHH MDB have poor prognosis due to metastases during presentation. (Figure 12, 13)
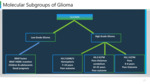
PAEDIATRIC LOW GRADE GLIOMAS
It includes pilocytic astrocytoma, ganglioglioma, pleomorphic xanthoastrocytoma, dysembryoplastic neuroepithelial tumours and diffuse astrocytomas, of which pilocytic astrocytomas are the most common.
Molecular pathways
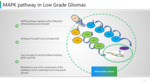
Paediatric LGG is a single-pathway disease involving the MAPK pathway. (Figure 14)
Binding of growth factor to a receptor tyrosine kinase activates the signal transducer RAS instigating two cascades of reactions, one involving BRAF, MEK, and ERK and other involving PI3K and mTOR. These ultimately converge in the nucleus to stimulate transcription factors such as MYC that drive growth promoting genes.
Majority of pilocytic astrocytomas involve BRAF fusion. while others involve BRAF V600E point mutation.
Imaging Surrogates:
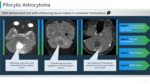
v Hypointense on T1WI, hyperintense on T2WI &FLAIR MR images and exhibit varying enhancement patterns.
v Well defined with negligible surrounding vasogenic edema.
v Pilocytic astrocytoma is usually present in the midline in posterior fossa whereas other histological subgroups are common in supratentorial location. (Figure 15)
Prognosis and predictive factors:
BRAF inhibitors are used in treating BRAF V600E mutation LGGs. BRAF fusion tumoursare resistant to BRAF inhibitors, hence treated with downstream MEK inhibitors.
PEDIATRIC HIGH GRADE GLIOMAS
HGGs include diffuse midline gliomas and other related high-grade infiltrating diffuse gliomas of childhood.
Molecular pathways
Epigenetic Regulators and HGGs:
DNA wraps around histones to form the building block of a chromosome. Histone act as an epigenetic regulator to control gene expression by modifying their protruding “tail” domain.
Molecular Subgroups:
H3 K27M and H3 G34R/V mutations in histone 3 influence gene expression in HGGs. Mutations in various cell cycle regulators like cyclin-CDK complexesand components of MAPK pathway are also implicated in pediatric HGG. (Figure 16)
Imaging surrogates:
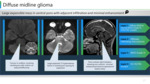
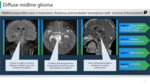
Ø Diffuse midline glioma : Large, expansile, asymmetric mass with its epicentre in pons with variable infiltration into thalamus, midbrain, and cerebellum.
Ø Exophytic spread laterally and ventrally with basilar artery encasement.
Ø Minimal enhancement. (Figure 17, 18)
Prognosis and predictive factors:
H3.1 K27Mmutant of diffuse midline glioma involving ventral pons affecting children between 4–6 years confers worse prognosis. (Figure 19)
EPENDYMOMA
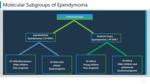
3rd most common pediatric brain tumour of neuroepithelial origin, with majority of them located in the posterior fossa. Ependymoma subgroups are summarised in the tabulation.
Molecular pathways :
Two main subgroups of supratentorial ependymomas are characterized by fusions on chromosome 11 involving RELA or YAP1 subgroup.
No mutational driver of Infratentorial ependymoma has been identified. A proportion of posterior fossa A and B tumours are characterized by a gain of chromosome 1q.
Imaging surrogates:
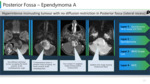
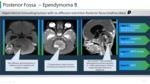
v T1 hypointense and heterogeneously high T2 signal intensity with punctate and coarse calcifications.
v Insinuation and extension of tumour through the foramina of Luschka/Magendie, encasing neurovascular structures.
v Posterior fossa A ependymoma arises from lateral recess of fourth ventricle, while posterior fossa B ependymoma arises in the midline from obex. (Figure 20, 21)
Prognosis and predictive factors
Resection of posterior fossa Type A ependymoma is difficult. Posterior fossa Type B ependymoma have better operability and clinical outcome. (Figure 22)
EMBRYONAL TUMORS:
Genetic aberrations:
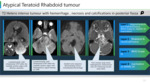
Atypical Rhabdoid Teratoid tumour is a highly aggressive posterior fossa tumour defined by inactivation of SMARCB1/INI1.
Imaging surrogates
v Heterogeneously enhancing tumour with reduced diffusivity and intratumoral hemorrhage. (Figure 23)
Prognosis and predictive factors:
Poor prognosis, ineffective to chemotherapy.
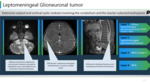
DIFFUSE LEPTOMENINGEAL GLIONEURONAL TUMOUR:
Genetic aberrations:
New rare distinct tumour entity that shows positivity for synaptophysin, GFAP and negative for IDH mutation.
Imaging surrogates
v Thick leptomeningeal enhancement and subpial nodular cysts.
v Intramedullary peripherally enhancing cystic lesion in the spine(Figure 24).
Prognosis and predictive factors:
Low grade tumour with slow progression.