Spinal cord development generally includes three phases namely gastrulation, primary neurulation and secondary neurulation.
Gastrulation involves conversion of the bilaminar embryonic disk to a trilaminar disk composed of ectoderm, mesoderm, and endoderm. Another critical event occurring along with gastrulation is the formation of notochord, cylindrical structure of mesodermal origin(Fig. 2).
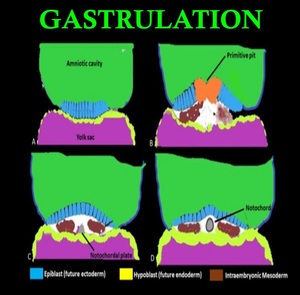
Fig. 2: Gastrulation and Development of notochord: (A) Coronal view through the bilaminar disc (B) Epiblast cells invaginate at the primitive pit creating the cells of the definitive endoderm as well as the mesoderm (C)Formation of definitive intraembryonic mesoderm and notochordal plate (D) Notochordal plate then detaches from the endoderm, and forms a tube referred to as the definitive notochord
Primary neurulation involves closure of neural plates to form neural tube. Notochord invites the ectodermal cells to differentiate into neural precursor cells, forming the neuralplate. Closure of neural tube begins at the medial hinge point in the ventral midline. Fusion proceeds in both rostral and caudal directions. During closure unfused rostral and caudal regions are referred as neuropores. Neural crest cells at the lateral border of neural plates, separates from the neural tube during closure and forms a mass dorsal to neural tube(Fig. 3).
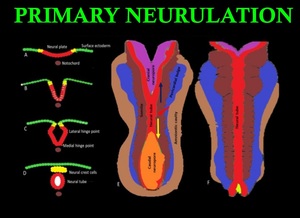
Fig. 3: Primary neurulation: The neural tube (red) and neural crest cells (yellow) are derived from surface ectoderm which forms neural plate induced by notochord (brown). (A)Future neural crest cells reside lateral to future neural tube. (B) Beginning of neural plate folding and internalization of neural plate cells. (C) Medial and lateral hinge plate facilitating folding of neural plate. (D) Separation of neural crest cells to form a mass dorsal to neural tube. (E) and (F) Dorsal view showing the closure of neural tube which begins in the center and progress in both cranial (blue arrow)and caudal (yellow arrow)direction.
Secondary neurulation involves the formation of conus medullaris and filum terminale. pluripotent tissues of the caudal eminence (tail bud), consisting of neural and mesodermal tissues, appear distal to the caudal neuropore . The caudal eminence fuses with the caudal neuropore, which then undergoes cavitation and connects to the central canal already present in the neural tube. (Fig.4).
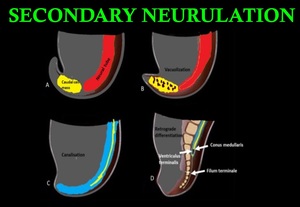
Fig. 4: Secondary neurulation: (A & B) Formation and vacuolization of caudal cell mass from the caudal end of neural tube distal to caudal neuropore. (C) Coalition of vacuoles and connection to the central canal by canalization. (D) In retrograde differentiation, cells of caudal cell mass undergo apoptosis to form conus medullaris, filum terminale and ventriculus terminalis.
In addition, recent studies suggest the presence of a unique process called “junctional neurulation,” bridging the primary and secondary neurulations.
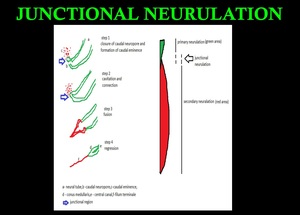
Fig. 5: Primary neurulation - appearance of the neural groove and folds to closure of caudal end of the neuropore. Secondary neurulation – formation of caudal eminence,cavitation, and connection,fusion and regression.Junctional neurulation- bridging the primary and secondary neurulations.
Failed neurulation leads to various types of spinal developmental malformations. Although primary neurulation is relatively well understood, less is known about the process of secondary neurulation or its clinical importance.
The embryonic background of spinal lipoma cases is marked by a premature disjunction between the neural and cutaneous ectoderm during the process of primary neurulation.Failed primary neurulation allows invasion of mesenchymal tissue of mesodermic origin into the neural structure, leading to the formation of a spinal lipoma. An unfused spinal cord caused by failed primary neurulation remains dysraphic in spinal lipoma.
Despite most spinal lipomas being regarded as the result of failed primary neurulation, some confusion in terms of the embryogenesis of spinal lipomas remains. Embryologists often consider closed spinal lesions to be the result of failed secondary neurulation.Indeed, the presence of a lipoma in the filum terminale, the final product of secondary neurulation, is increasingly being acknowledged. The availability of MRI has made the discovery of caudally located lipomas, which do not penetrate the dorsal aspect of the dura mater or fascia, and are unassociated with spina bifida, more common. It should be remembered that spina bifida diagnosed in such patients does not constitute true “pathological” spina bifida, but merely “physiological spina bifida” formed by the cartilaginous part of the lamina in the course of normal development.
Surgically orientated classification of spinal lipoma was first proposed by Chapman and underwent various modifications based on MRI findings.Chapman’s classification is still useful today but does not account for spinal lipomas originating from failed secondary neurulation. Some have included the filum lipoma in the classification.However, because this classification is simply based on the site and morphology of the spinal lipoma, it is now considered to have less clinical significance.
Here we classify spinal lipomas based on caudal spinal cord formation during embryogenesis. The formation of spinal lipomas from primary to secondary neurulation was considered, and the conditions were classified based on the stage of embryogenesis.





