We illustrate the subcortical T2 hypointensity presence in the following pathologies:
Meningitis and encephalitis
The presence of this phenomenon has been described in some cases of infectious origin, specifically in viral encephalitis (being more frequent in non-herpetic cases) and in meningitis of pyogenic, cryptococcal and tuberculous origin. This finding is usually associated with leptomeningeal enhancement. In cases of encephalitis, it is associated with T2 hyperintensity of the neighboring cerebral cortex.
Subcortical white matter T2 hypointensity would be a transitory phenomenon, reversible, like other typical findings, especially when the leptomeningeal enhancement disappears.
In histology, no structural abnormalities have been demonstrated in the subcortical white matter involved.
The paramagnetic effect of oxygen free radicals has been proposed as a possible causal factor, explained by a transient increase in the amount of them. Also, ferric iron accumulation would play a role. Subcortical low intensity would be visualized when there is a sufficient concentration of free radicals and ferric iron produced at a certain time in the disease process without sufficient free radical scavengers or iron chelators.
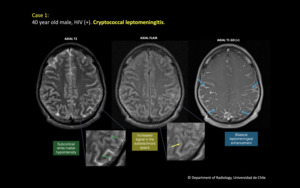
Fig. 1: 40 year old male, HIV (+). Cryptococcal leptomeningitis.
T2 FLAIR Axial: Bilateral subaracnoid space hyperintensity in both hemispheres. Subcortical white matter hypointensity is seen, best demarcated in parietal lobes.
T2 Axial: The subcortical white matter hypointensity is better demonstrated.
T1 Gd Axial: Bilateral leptomeningeal enhancement.
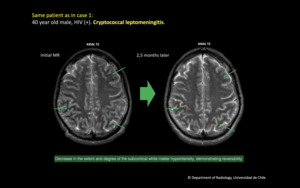
Fig. 2: Same patient as in case 1: 40 year old male, HIV (+). Cryptococcal leptomeningitis.
Initial MRI and control performed 2.5 months later. There is significant decrease in the extent and degree of the subcortical white matter hypointensity, demonstrating reversibility.
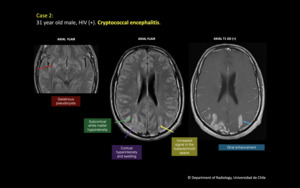
Fig. 3: 31 year old male, HIV (+). Cryptococcal encephalitis.
Increased signal in the subarachnoid space in FLAIR. There is subcortical white matter hypointensity in both parieto-occipital lobes, where there also is cortical swelling and hyperintensity, with giral and leptomeningeal enhancement. Gelatinous pseudocysts in basal ganglia.
Leptomeningeal metastasis
The phenomenon has been described in patients with adenocarcinomas (pulmonary and gastric). Usually associated to nodular leptomeningeal enhancement and secondary parenchymal lesions.
The proposed mechanism is the same as in meningitis and encephalitis, an increase in the amount of free radicals and ferric iron.
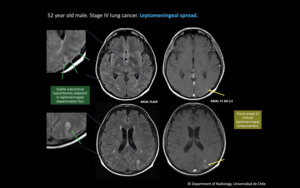
Fig. 4: 52 year old male. Stage IV lung cancer. Leptomeningeal spread.
On FLAIR there is subtle subcortical hypointensity adjacent to leptomeningeal dissemination foci. Focal areas of nodular leptomeningeal enhancement are seen in post contrast images.
Hyperosmolar hyperglycemic syndrome
Acute manifestation of hyperglycemia characterized by hyperosmolarity (> 320 mOsm/kg) and absence of ketonemia or ketoneuria. Occurs almost exclusively in elderly patients with type 2 DM. It usually is manifested as a neurological syndrome with lethargy, seizures and Babinski sign of the foot (that reverts with treatment). The mortality rate is 35 to 40%.
Subcortical white matter hypointensity can be observed in the parieto-occipital cortex, which is reversible with proper treatment. Diffusion restriction could be seen if associated with a seizure phenomenon.
The precise pathogenesis of this transient subcortical hypointensity remains uncertain. One of the possible mechanisms could be subcortical transient free radical accumulation due to excitotoxic axonal damage during seizures. Other mechanisms would be related to mineral deposition and ischemia.
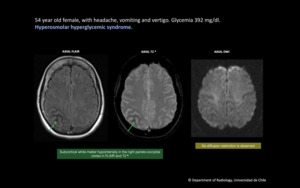
Fig. 5: 54 year old female, with headache, vomiting and vertigo. Glycemia 392 mg/dl. Hyperosmolar hyperglycemic syndrome. There is subcortical white matter hypointensity in the right parieto-occipital cortex on FLAIR and T2*. No diffusion restriction is observed.
Sturge Weber syndrome
Neurocutaneous disorder characterized by facial port wine stain, congenital glaucoma and pial angiomatosis, which compromises a cerebral hemisphere in 85% of cases.
It has been demonstrated that there is accelerated myelination, which would explain the subcortical T2 hypointensity in the affected areas. The anomalous venous plexus over the cerebral surface leads to abnormal cortical drainage and venous stasis and this, on the other hand, would condition hypoperfusion of the underlying parenchyma wich could be a factor that would favor accelerated myelination.
On imaging, a decrease in the parenchymal volume and T2 hypointensity can be observed in the white matter underlying the area of leptomeningeal enhancement.
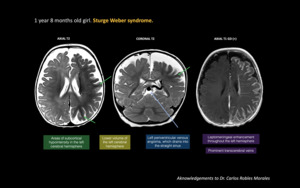
Fig. 6: 1 year 8 months old girl. Sturge Weber syndrome.
On T2 images there are areas of subcortical hypointensity in the left cerebral hemisphere. Lower volume of the left cerebral hemisphere is noted. There is also a left periventricular venous angioma, which drains into the straight sinus. On T1 post contrast image there is leptomeningeal enhancement throughout the left hemisphere and there are prominent transcerebral veins.
References: Dr. Carlos Robles Morales
Ischemia of arterial origin and ischemic-anoxic insults
In cases of acute and early subacute infarction, the subcortical white matter hypointensity can be seen adjacent to areas where the cortex showshigh intensity to isointensity. It is usually reversible, but in some cases it may persist in later stages.
In cases of severe ischemic-anoxic insults, the subcortical and the deep white matter may show lower signal on T2 weighted images, that can also be persistent in time.
One of the possible pathophysiological mechanisms that have been proposed is the production of free radicals in acute and early subacute stages of ischemia andwould be responsible for this finding in these stages. Another mechanism that has been proposed is the accumulation of non heme iron in the subcortical white matter due to interruption of axonal projections, failing its transport and this would be responsible for the lower signal intensity on T2 weighted images during late subacute and chronic stages.
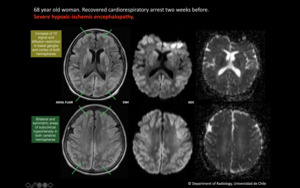
Fig. 7: 68 year old woman. Recovered cardiorespiratory arrest two weeks before. Severe hypoxic-ischemic encephalopathy.
On FLAIR there are bilateral and symmetric areas of subcortical hypointensity in both cerebral hemispheres. Also there is increase of T2 signal and diffusion restriction in basal ganglia and cortex of both hemispheres.
Cerebral venous thrombosis
Venous congestion secondary to acute or chronic obstruction of venous outflow has been proposed as the causative factor. The underlying pathophysiology appears to be the same as arterial ischemia, the accumulation of nonheme iron and/or free radicals in subcortical white matter.
It can be seen adjacent to areas of venous infarction and hemorrhagic lesions.
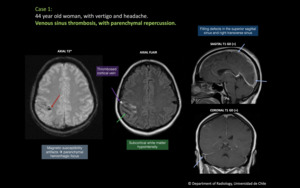
Fig. 8: 44 year old woman, with vertigo and headache. Venous sinus thrombosis, with parenchymal repercussion. There is an area in the right parietal lobe that shows subcortical white matter hypointensity on FLAIR and T2*. Also, there are magnetic susceptibility artifacts representing parenchymal hemorrhagic focus. Adjacent to the lesion there is a cortical vein that shows hyperintensity on FLAIR. The T1 post contrast images show filling defects in the superior sagittal sinus and right transverse sinus.
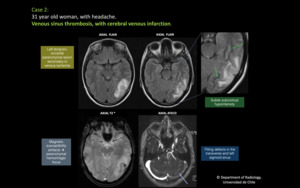
Fig. 9: 31 year old woman, with headache. Venous sinus thrombosis, with cerebral venous infarction. There is a left temporo-occipital parenchymal lesion secondary to venous ischemia, that shows high signal intensity of the cortex, that has magnetic susceptibility artifacts representing hemorrhagic focus. There is subtle subcortical hypointensity associated adjacent to the parenchymal lesion. On T1 post contrast image there are filling defects in the transverse and left sigmoid sinus.










