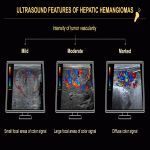
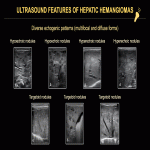
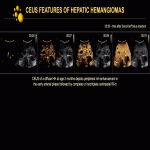
On US, HHs can present with a spectrum of imaging findings, showing variable size, echogenicity, and tumor vascularity. Lesions can appear predominantly hyperechoic, hypoechoic, isoechoic, targetoid, or as large dilated cystic vascular spaces (Fig. 9, 11). On color Doppler US, the intensity of tumor vascularity can be mild (small focal areas of color signal), moderate (large focal areas of weak color signal), or marked (diffuse color signal) (Fig. 10). They may show increased stiffness on US elastography.
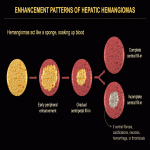
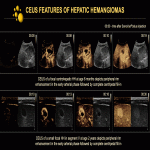
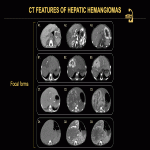
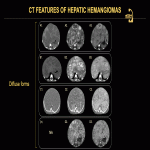
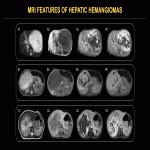
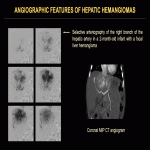
On all contrast-enhanced modalities, lesions show intense, predominantly peripheral, contrast uptake in the arterial phase, followed by complete or incomplete centripetal filling in the portal venous phase (Fig. 12). Examples of CEUS enhancement patterns are shown (Fig. 13, 14). On unenhanced CT, all lesions appear hypoattenuating relative to normal liver parenchyma, except for inconstant central or peripheral calcifications. On contrast-enhanced CT, peripheral enhancement of focal HHs in the arterial phase can either be discontinuous, corrugated, or continuous, rim-like (Fig. 15-17). On MRI, HHs appear T2-hyperintense and T1-hypointense relative to normal liver parenchyma with centripetal contrast enhancement in the arterial and portal venous phases (Fig. 18). An example of angiographic findings is shown (Fig. 19).
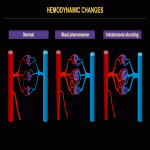
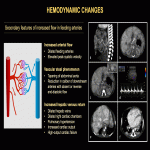
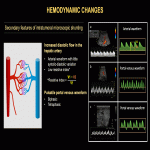
Hemodynamic changes secondary to HHs can be categorized as secondary features of: increased flow in the feeding arteries; increased hepatic venous return; or intratumoral microscopic arteriovenous shunting (often associated). Signs of increased arterial flow include: dilated feeding arteries with elevated peak systolic velocity; vascular steal phenomenon with tapering of the abdominal aorta; reduction in the caliber of downstream arteries with absent or reverse end-diastolic flow. Signs of increased hepatic venous return include: dilated hepatic veins; increased flow velocity of draining hepatic veins; dilated right cardiac chambers; pulmonary hypertension; increased cardiac output; high-output cardiac failure. Signs of intratumoral arteriovenous shunting include: arterial waveform with little systolic-diastolic variation due to increased diastolic flow with a corresponding decrease in the resistive index; pulsatile waveform with flow reversal in the portal vein (Fig. 20-22).
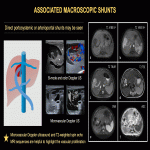
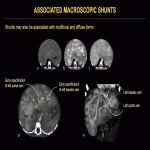
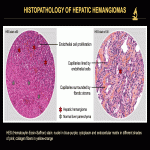
Macroscopic vascular shunts can be associated with all forms. HHs intertwined with a portosystemic shunt may be difficult to diagnose. The use of microvascular Doppler ultrasonography and T2-weighted spin echo MRI sequences can be helpful to delineate the vascular proliferation surrounding the shunt. Microvascular Doppler ultrasonography is very sensitive in identifying tumor microvascularity. T2-weighted spin echo (including diffusion-weighted) sequences show the hemangiomatous tissue as areas of increased signal intensity surrounding the shunt flow void (Fig. 23, 24).
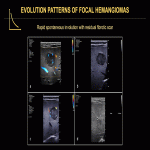
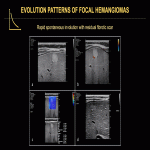
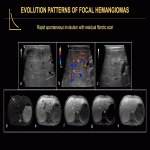
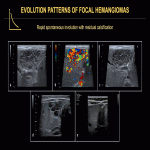
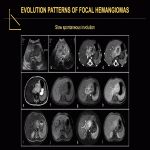
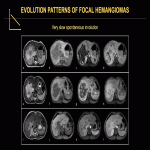
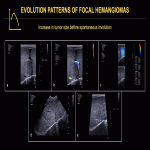
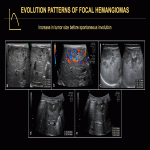
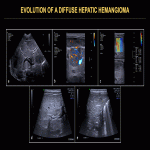
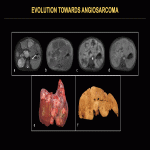
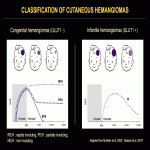
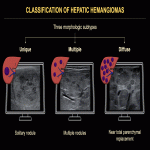
Focal HHs can exhibit several evolution patterns. They may undergo rapid or slow postnatal involution, sometimes leaving residual fibrosis or calcifications (Fig. 25-30). Some may show postnatal growth (Fig 31, 32). Multifocal and diffuse HHs usually decrease in size over time (Fig. 33). Transformation of multifocal or diffuse HHs towards angiosarcoma is exceptional (Fig. 34) [4, 5].
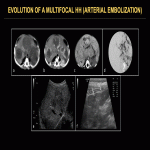
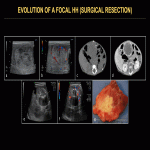
Medical treatment options for complicated HHs include symptomatic treatment and propranolol (usually for infantile forms). In case of failure to respond to medical management, therapeutic options include: hepatic artery embolization (Fig. 35), arterial ligation, surgical resection (Fig. 36) and liver transplantation [6, 7].
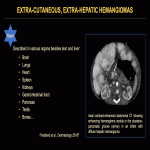
Extra-cutaneous, extra-hepatic hemangiomas are rare (Fig. 37) [8].
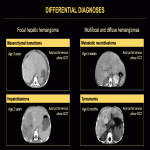
The differential diagnoses for focal HH include:
- Mesenchymal hamartoma: may also occur in the neonatal period. The usual imaging finding is a large, well-defined, solitary mass containing multiple cysts of varying sizes. They may exceptionally coexist with HHs.
- Hepatoblastoma: rarely occurs in the newborn, is not as hypervascular as HH and serum levels of alpha-fetoprotein are usually markedly elevated.
- Neonatal or infantile focal nodular hyperplasia: exceedingly rare
- Hepatic arteriovenous malformation: exceedingly rare, this vascular malformation is not associated with endothelial cell proliferation. It usually persists or worsens over time and will not usually undergo spontaneous resolution. Contrary to HH, imaging findings won't include focal areas of signal abnormality and contrast enhancement corresponding to endothelial proliferation surrounding the dilated vessels.
The differential diagnoses for multifocal and diffuse HHs include liver metastases (eg. neuroblastoma) and metabolic diseases (tyrosinemia, mitochondrial cytopathies) (Fig. 38).