Non-vascular interventions include transthoracic needle biopsy, thermal ablation of lung malignancies and drainage of thoracic fluid collections.
Thoracic Vascular interventions include stenting of superior vena cava, embolization of arteriovenous malformations, transpulmonary chemoembolization and embolization of bronchial arteries following hemoptysis.
Transthoracic needle biopsy (TTNB)
Indications:
Indeterminate pulmonary or mediastinal nodule or mass, depending on probability of malignancy and presence of metastatic disease.
Distinguishing changes following radiotherapy and surgery from recurrent disease.
Distinguishing infections from malignancies.
Contraindications
All contraindications are relative and do not pose a threat in hospital setting.
Hemorrhagic diathesis (performing radiologist should obtain recent platelet count, prothrombine time (PT), partial thromboplastine time (PPT) and international normalized ratio (INR)).
Discontinuation of anticoagulants prior to the procedure is necessary.
Other relative contraindications are deep lesions in patients with pulmonary hypertension, emphysema and/or large bullae, intractable cough and mechanical ventilation [1].
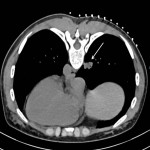
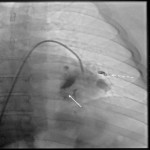
The procedure is usually performed using an aspiration needle, cutting needle or automated core-biopsy needle. First, a CT of designated area is obtained with a radiopaque grid used to determine the best approach to the lesion, as well as the depth and the angle of trajectory
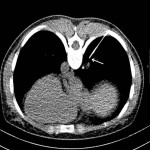
(Figure 1). Afterwards, entry point is marked and local anesthesia is administered to the puncture site. The needle is advanced to the pleura and short segment CT is performed to verify position and angle. Needle is then advanced to the predefined depth and the position is once again verified using CT, followed by sample acquisition
(Figure 2). Final CT scan is obtained after the removal of the needle to check for complications like pneumothorax, hemothorax and soft tissue hematoma. Additional X-ray is performed 2 hours after the procedure to check for delayed pneumothorax.
When multiple needle passes are required a larger needle can be used as a guidance cannula through which smaller biopsy needle can be passed repeatedly. This is called a coaxial technique.
Complications include pneumothorax, hemothorax, parenchymal hemorrhage, soft tissue hematoma, hemoptysis and air embolism.
Thermal ablation of lung malignancies
Indications
Patients with stage IA (limited to T1a and T1b) non-small cell lung carcinoma unsuitable for surgery or stereotactic radiotherapy.
Oligometastatic colorectal cancer with up to 3 lung nodules (≥ 2 cm) and contraindication to surgery.
Contraindications
Diagnosis of small cell lung cancer.
≥ Stage II NSCLC.
Presence of nodal and distant metastases.
Untreatable coagulopathies.
An Eastern Cooperative Oncology Group performance status of > 2.
A life expectancy of less than 1 year.
An end-stage lung disease and/or respiratory failure.
Lesions located < 1 cm from hilum, large vessel, main bronchi, trachea or esophagus [2].
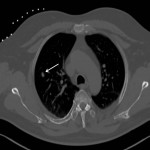
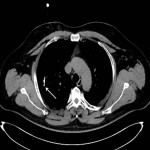
There are three thermal ablation modalities currently in use: radio-frequency ablation (RFA), microwave ablation (MWA) and cryoablation. The first two employ electricity to create high temperatures and the latter one uses pressurized argon gas to achieve temperatures around -40°C. RFA is the oldest and most well-known procedure; however, its greatest disadvantage is the “heat sink” phenomenon, a loss of heat to the blood vessels and airways near the ablated lesion. Other disadvantages of RFA include possible skin burn and the use of only one probe at a time. Both MWA and cryoablation are less sensitive to “heat sink” and allow for multiple probes. The additional advantages of cryoablation include the possibility of treating larger lesions and facilitating of an immune response through exposure of the tumor cell content to the patient’s immune cells [3]. The procedure techniques are similar to the transthoracic biopsy with some exceptions
(Figure 3 and 4). They are usually performed under general anesthesia or conscious sedation. The peripheral lesions are best approached tangentially, as this helps avoid back burn or freeze along the ablation trajectory and in cryoablation it helps contain parenchymal hemorrhage preventing hemothorax.
Complications are the same as in TTNB, with addition of bronchopleural fistula.
Superior vena cava (SVC) stenting
Indications
The main indication for SVCS is an external compression or thrombosis of SVC, leading to the Superior vena cava syndrome (SVC syndrome), the umbrella term for a group of symptoms and clinical signs associated with partial or complete obstruction of blood flow in SVC. The syndrome is usually caused by thoracic malignancies, with non-malignant causes accounting only for a third of cases. The indications for stenting or other alternative procedures depend on the severity of the symptoms, which can range from asymptomatic, mild head and neck edema, to severe and life-threatening cerebral and laryngeal edema.
Contraindications
Contraindications are only relative, since the procedure is usually palliative. In case of malignancies with good outcome chances, radiotherapy or chemotherapy should be attempted prior to stenting.
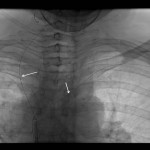
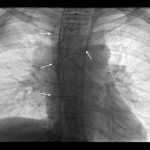
The procedure is usually performed with the patient lying supine in conscious sedation or general anesthesia. The site of venous puncture depends on the site and extent of obstruction. The femoral vein approach it the most commonly used one, however, in the case of obstruction involving brachiocephalic or subclavian vein additional approach through basilic or brachial vein can be helpful
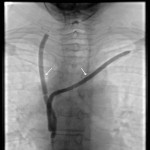
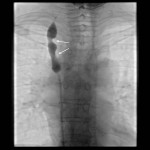
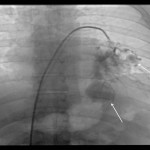
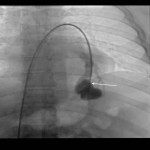
(Figure 5). Under the control of digital subtraction venography the stenosis or occlusion is traversed with guidewire, the type of which depends on the severity of stenosis. Next, several pre-dilatations with increasing balloon sizes are performed while assessing the patient’s response with each dilatation
,
,
(Figure 6, 7 and 8). The maximum diameter of the balloon should not exceed 16 mm. Various stents are at disposal for this procedure; balloon expandable, self-expanding, which can be covered or noncovered
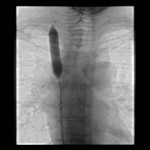
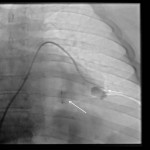
(Figure 9). The final decision on which stent to use depends on the characteristics of the lesion, anatomy and operator experience. There is no significant difference in patient mortality among different stent types [4].
Complications are rare but severe, including pericardial tamponade, SVC rupture, in-stent restenosis, stent migration, bleeding and pulmonary embolism.
Pulmonary arteriovenous malformation embolization
Pulmonary arteriovenous malformations (PAVMs) are mostly found in patients with hereditary hemorrhagic teleangictasia (HHT). Their potential for causing paradoxical emboli followed by stroke or brain abscess makes even the asymptomatic PAVMs with feeding artery diameter <3 mm eligible for intervention.
Contraindications include severe pulmonary hypertension, renal failure, early pregnancy, coagulopathy and feeding artery diameter <2 mm (the latter only for asymptomatic PAVMs); however, these are only relative and may either postpone the procedure or convert it to surgical resection.
The procedure begins with venous puncture (usually the right femoral) and selective pulmonary angiography to identify the feeder arteries
(Figure 10). The feeder arteries are then selectively catheterized with the help of a guidewire or microcatheter followed by digital subtraction angiography (DSA) to provide a road map and precisely measure the artery diameter
(Figure 11). The embolization itself may be performed using coils, detachable balloon, vascular plugs (Amplatzer and microvascular plug), or both
,
(Figures 12 and 13). The choice of embolization material depends on the feeding artery diameter (>9 mm usually warrants the use of coils) and the experience and preferences of respective operators [5].
Complications are rare and include the precipitation of pulmonary hypertension, pulmonary infarction, hemoptysis, vascular perforation, migration of embolization device and paradoxical balloon or coil embolization.