CASE REPORTS
We present five cases of EM: 4 females and 1 male,
medium age 54,8 years (range: 37- 72).
None of them had a history of coronary artery disease,
although only in one case angiography was performed to rule out significant stenosis in coronaries. No history of atherosclerotic disease was present in any patient,
and cardiovascular risk factors were only found in a 69-years-old female (smoking).
In every case except one (toxocariasis case),
eosinophilia was already known at the time of performing CMR.
Hypereosinophilic syndrome was diagnosed 4 years and 1 year before the initial CMR,
and Churg-Strauss vasculitis was diagnosed 6 years and 5 years before the initial CMR.
CMR findings were interpreted as myocardial infarction initially in 2 cases (toxocariasis and one hypereosinophilic syndrome),
although on subsequent CMR they were accurately diagnosed of EM.
Eosinophilia was present at the time of initial CMR in two cases; in the remaining three it was previously documented,
but has been resolved when performing CMR,
after therapy of underlying disease.
Only the patient with toxocariasis presented with chest pain and raised troponins suggesting an acute coronary syndrome.
One patient with hypereosinophilic syndrome presented with mild thoracic discomfort and dyspnoea due to pleuropericardial effusions,
and raised troponins were also found.
The remaining three patients did not have any cardiac symptoms,
and CMR was ordered after abnormal echocardiographic findings.
In our five patients CMR revealed subendocardial enhancement with variable extension,
diffuse in two cases,
and patchy and multifocal in the remaining three.
Enhancement was thin and regular in every case.
Papillary muscles also showed enhancement in all five cases.
Enhancement was sometimes hard to demonstrate,
and use of different sequences for delayed enhancement,
careful adjustment of inversion time to null myocardial signal or a longer time from contrast administration to imaging were needed in some cases.
In one case a small pericardial effusion was noted (hypereosinophilic syndrome that presented with pleuropericardial effusions one year ago).
Left ventricle systolic function was preserved in three cases.
A systolic dysfunction was found in one patient with a concomitant aortic valve double lesion due to a bicuspid valve and in the toxocariasis infection.
All five patients underwent CMR follow-up during a variable period: 1 year (2 CMR in two cases and 3 CMR in one case),
3 years (3 CMR in one case),
and 5 years (3 CMR in one case).
The extension of enhancement persisted without significant changes in all patients except in the toxocara infection,
where a decrease of enhancement was demonstrated.
We have not been able to document a progression to thrombotic or fibrotic stage in any patient,
so we presume that a much longer time is required for this progression.
We present CMR findings in three patients.
CASE 1
A 72-years-old male with no previous history of coronary artery disease was admitted to the hospital with chest pain and raised troponin-T levels (peak of 700 µg/l).
ECG showed a ST depression in V4-V6 leads.
A diagnosis of non-ST elevation myocardial infarction was established.
Angiography revealed unremarkable coronaries.
Echocardiography showed a mild dilatation of LV with normal ejection fraction and mild concentric hypertrophy.
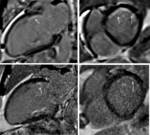
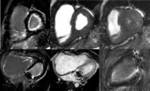
CMR (Figures 1 and 2) showed a non-dilated left ventricle with mild systolic dysfunction,
and subendocardial enhancement in two locations: inferoseptal apical segment and anterior and anterolateral basal segments.
Inferior papillary muscle also showed contrast enhancement. These findings were initially interpreted as myocardial infarctions.
Moderate eosinophilia (1900 eosinophils/mm3) lead to a diagnosis of toxocariasis (positive IgG serology) and treatment with albendazole was started,
in addition to therapy with steroids.
A second MRI three months later showed a decrease in the size of these areas of enhancement.
Eosinophilia had decreased to 800 eosinophils/mm3.
Some hours later after hospital admission,
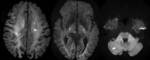
the patient also reported diplopia,
and cranial MRI revealed multiple acute embolic foci in cerebral hemispheres,
basal ganglia and right cerebellar hemispheres (Figure 3).
Cranial embolisms were considered to be caused by eosinophilia.
CASE 2
A 56-years-old female was admitted in another hospital with a history of asthenia,
polymialgias and progressive dyspnoea.
Examinations revealed pleuro-pericardial effusion that required thoracocentesis and pericardiocentesis.
Analysis of pleural and pericardial fluid revealed high levels of eosinophils,
and peripheral blood eosinophilia was also present (3600 eosinophils/mm3).
A diagnosis of hypereosinophilic syndrome was established after other causes of eosinophilia were excluded,
and treatment with prednisone and azathioprine was initiated.
During this admission,
elevated troponin-I levels were recorded (peak of 4733 µg/l),
without any related symptoms.
Echocardiography only found the pericardial effusion,
and other investigations performed during her stay at hospital were unremarkable.
She received three pulses of methylprednisolone with clinical improvement and the eosinophilia disappeared.
After discharge from hospital she was on azathioprine treatment,
and one year later was referred for a CMR due to her previous history,
although echocardiography was normal and no cardiac symptoms were present.
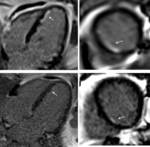
CMR (Figure 4) revealed subendocardial enhancement that persisted without changes for a one year follow-up (3 CMR).
Left ventricle was not dilated and ejection fraction was preserved.
CASE 3
We describe the case of a 40-years-old female diagnosed of Churg-Strauss vasculitis since 6 years,
with CNS,
cardiac,
skin and kidney involvement and was receiving prednisone therapy.
She had pulmonary hypertension and was in NYHA functional class II.
Mitral valve was replaced some decades ago due to rheumatic disease.
She was referred for CMR after echocardiographic finding of apex obliteration in both ventricles.
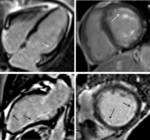
CMR (Figure 5) showed a diffuse and extensive subendocardical enhancement in left ventricle that was interpreted as a sign of EM rather than small vessel vasculitis.
Ejection fraction of LV was normal.
Right ventricle was dilated with moderate systolic dysfunction,
and severe tricuspid regurgitation with dilatation of right atrium were demonstrated.
There was myocardial thickening in both ventricles,
more severe at the inferior segments and at the apical level,
with obliteration of the apex.
This finding may suggest a progression to the fibrotic stage.
REVIEW AND DISCUSSION
EM is the cardiac manifestation of eosinophilia.
It is also known as Loffler endocarditis,
as he was the first to describe the association between cardiac pathology and eosinophilia.
It is not a frequent cardiac condition,
although it is probably underdiagnosed.
Diagnosis is challenging,
but if not treated may lead to irreversible cardiac damage.
Eosinophilia
Eosinophilia is characterised by high eosinophil blood count and organ damage due to eosinophil toxicity.
Although a percentage superior to 7% generally indicates eosinophilia,
an absolute eosinophil count is preferred to establish the diagnosis.
Eosinophilia is defined by > 500 eosinophils/mm3.
It is arbitrarily classified as mild (500-1500),
moderate (1550-5000) and severe (> 5000).
Eosinophils play an important role in the immune system,
regulating inflammation and destroying foreign substances.
However,
in the context of eosinophilia,
eosinophils may infiltrate organs and tissues and cause toxicity.
Circulating eosinophils contain granules in their cytoplasm filled with varied cationic proteins,
enzymes,
cytokines and lipid mediators. Activation of eosinophils results in release of these proteins,
with toxic effects such as cell necrosis,
inflammatory reaction,
thrombotic activation and fibroblast activation.
Causes of eosinophilia are varied:
· Reactive is the most common cause,
mainly due to an infection.
Parasites are frequent,
including trypanosome,
echinococcus and toxocara,
but other agents may be related: HIV,
tuberculosis,
etc.
Drug-induced hypersensitivity is also frequent,
and may be caused by anticonvulsants,
non-steroidal anti-inflammatory drugs and many antibiotics.
Finally,
allergic diseases may cause reactive eosinophilia.
· Systemic diseases: Churg-Strauss vasculitis,
Wegener granulomatosis,
Crohn disease,
polyarteritis nodosa,
rheumatoid arthritis…
· Malignancies,
mainly hematologic (Hodgkin and non-Hodgkin lymphoma,
leukemia,
mastocitosis),
but also solid neoplasms.
· Primary Hypereosinophilic Syndrome is a group of haematological disorders (myeloproliferative or lymphoproliferative) with unexplained and sustained eosinophilia.
It is an exclusion diagnosis after other causes of eosinophilia are ruled out.
Manifestations of eosinophilia are varied:
· Cardiac involvement is frequent,
occurring in almost half of the cases,
and is the leading cause of mortality.
· Pulmonary involvement is also frequent,
and includes eosinophilic pneumonia (acute or chronic) and pulmonary fibrosis.
· Neurologic involvement can also occur,
with varied manifestations: embolisms,
eosinophilic meningitis,
peripheral neuropathy…
· Skin lesions,
such as erithrodermia,
urticaria,
panniculitis,
fasciitis…
· Gastrointestinal involvement may produce gastritis,
enteritis or hepatitis.
Eosinophilic heart disease
Three stages are recognized in cardiac involvement due to sustained eosinophilia.
1. Acute phase: eosinophilic myocarditis.
Release of cationic proteins from eosinophilic granules induces myocardial cell necrosis.
Endomyocarditis would be a more appropriate term,
as endocardium is also damaged.
This stage may be clinically silent,
or may present with non-specific signs of myocardial inflammation,
mimicking an acute coronary syndrome or acute myocarditis. In this stage,
therapy is aimed to reduce the eosinophil count,
and also to control the inflammatory reaction.
In most severe cases,
an acute necrotising myocarditis occurs,
resulting in acute heart failure or cardiogenic shock,
with fatal outcome.
Endomyocardial injury is the most common form of cardiac eosinophilic injury.
But pericardial involvement may also be associated,
with pericardial effusion and even cardiac tamponade.
Coronary arteritis seems to be an inflammatory reaction rather than a form of primary vasculitis.
Laboratory investigations may reveal increased serum IgE,
eosinophil cationic protein (ECP) and antineutrophil cytoplasmic antibody.
Increased myocardial damage markers (troponin) are usually present.
Endomyocardial biopsy is required to establish diagnosis demonstrating eosinophils infiltrating myocardial tissue.
However,
in clinical practice it is not routinely used in many hospitals.
Relying in blood eosinophilia to establish the diagnosis can be misleading,
as in some cases EM may occur without blood eosinophilia,
and most of the patients with sustained eosinophilia do no develop EM.
Detection of eosinophil cationic protein may be an adjunct to the diagnosis of EM.
Early diagnosis is recommended to initiate therapy for a favourable outcome,
avoiding adverse fibrotic remodelling.
Corticosteroids are usually given to reduce inflammatory myocardial necrosis,
but the underlying cause of eosinophilia should also be treated.
2. Intermediate phase: thrombo-necrotic stage.
Activation of thrombomodulin in the endothelial membrane and many factors of coagulation induce formation of mural thrombi along the damaged endocardium.
Thrombi are mainly found in ventricular apex,
and may be responsible of embolisms.
Anticoagulation therapy is therefore suggested,
although no clear evidence of benefit is reported.
3. Chronic stage: fibrotic stage.
Finally,
scarring and fibrosis occur,
reflecting an irreversible damage.
Fibrosis of cardiac valves and subvalvular apparatus cause valve dysfunction.
Fibrosis of the myocardium results in restrictive myocardiopathy,
and also may be responsible of arrhythmias.
Surgery may be indicated in this stage: endocardial decortication,
valve replacement or even cardiac transplantation.
Cardiac Magnetic Resonance in eosinophilic cardiac disease
Echocardiography is insensitive to pathologic changes occurring in the acute stage,
where only an endomyocardial biopsy would have any chance to diagnosis the disease,
if clinically suspected. Ventricular size and function are usually preserved.
Ventricular thrombus may be found in the intermediate stage,
and signs of restrictive myocardiopathy may be revealed in the final stage,
but both are not specific of cardiac involvement in eosinophilia.
Therefore,
CMR appears as the non-invasive technique of choice for diagnosis of EM.
Although it is a rare disease,
a growing number of publications and case reports are appearing,
probably due to the increasing use of CMR,
allowing a more accurate diagnosis of cardiac diseases.
· Acute phase: eosinophilic myocarditis
Subendocardial enhancement is the most typical and consistent sign in this stage.
It reflects endomyocardial damage,
mainly necrosis but also inflammation. It is perhaps the only myocarditis where enhancement is found in the endocardial side instead of in the epicardial,
as occurs in typical myocarditis.
Enhancement may also be intramyocardial or even transmural.
Other findings on MR include mural thickening and myocardial edema,
due to the inflammatory reaction.
There are some reports in the literature where only myocardial edema is found,
without any myocardial enhancement.
Differential diagnosis
EM causes subendocardial enhancement,
diffuse,
patchy or in a single location.
As far as we know,
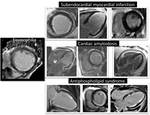
three other cardiac conditions may be considered in the differential diagnosis of subendocardial enhancement (Figure 6).
Subendocardial enhancement is almost pathognomonic of myocardial infarction,
a frequent condition due to the high prevalence of coronary artery disease.
In these cases,
a segmental abnormal wall motion (hypokinesia,
akinesia or dyskinesia) matching the area of enhancement is always found.
Enhancement is produced in a coronary artery territory,
although if multi-vessel disease is present,
more than one area of enhancement may be found.
Although clinical correlation is essential,
silent myocardial infarctions are sometimes discovered incidentally in CMR.
In addition,
a normal coronary arteriography does not allow ruling out a myocardial infarction,
as it may occur with normal coronaries.
Cardiac amyloidosis may produce a pattern of global subendocardial enhancement,
especially in the early stages.
However,
typical findings of cardiac amyloidosis usually help to establish the diagnosis: concentric myocardial hypertrophy,
enhancement in the wall of the atria and abnormal kinetics of gadolinium (with a difficult or impossible adjustment of the optimum inversion time in delayed enhancement sequences),
increased extracellular volume and native T1 septal (T1 mapping) and pericardial and pleural effusions.
Finally,
subendocardial enhancement has also been reported in antiphospholipid syndrome.
We had the opportunity to study with CMR one patient with antiphosphoplipid syndrome and normal coronary arteries and demonstrate extensive subendocardial enhancement.
For the radiologist,
an awareness of EM as a cause of subendocardial enhancement is therefore important: to avoid a misdiagnosis of myocardial infarction,
and to allow an early diagnosis of EM,
a rare condition with no typical findings in other non-invasive tests.
Although CMR is considered the imaging technique of choice for diagnosis in EM,
clinical relevance of subendocardial enhancement and potential role in follow-up have not yet been established.
In our small series,
we found similar pattern in symptomatic and asymptomatic patients,
and enhancement remained unchanged when eosinophilia resolved after treatment and clinical outcome was favourable.
· Intermediate phase: thrombotic stage
CMR accurately demonstrates endocavitary thrombus,
being superior to echocardiography,
especially if they are located in the ventricular apex,
or have a laminar morphology,
both characteristic of these cases.
In this stage,
subendocardial enhancement persists,
probably reflecting irreversible myocardial damage.
Thrombus are easily seen on postcontrast images as non-enhancing masses along the endocardial surface.
We have found that standard VIBE (volume interpolated breath-hold examination) axial sequence is highly sensitive.
· Chronic phase: fibrotic stage
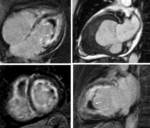
CMR reveals a mural thickening of the ventricles with obliteration of ventricular cavities and subendocardial enhancement (Figure 7).
Thrombi may persist.
Hypertrophic myocarditis may be considered as a differential diagnosis.