Results
The results of T1 and T2* imaging of the Achilles tendon of all 48 subjects were analyzed according to the normal and patient group.
Axial and sagittal morphological images,
T1 and T2* images of the AT are shown in figure 1.
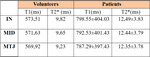
T1 and T2* mean values in each ROI of the AT for normal and patients group are summarized in table 2.
Our results show a statistically significant differences of T1 and T2* values in normal group compared to the patients group (p<0.05).
Means values of T1 and T2* in the whole AT for normal group were 571.69 ms and 14.54 ms respectively and were 818.10 ms and 18.57 ms respectively for patients.
There were no significant differences in T1 and T2* values between the different regions of interest (p>0.05) for normal group. However,
there were a significant difference for patients group.
The analysis of the variation of T1 and T2* values between normal and patient group,
showed no correlation for T1 (r=0.193) and T2* (r=-0.099).
For normal group,
there was a positive significant weak correlation between T1 and T2* (r=0.385,
p=0.002).
The plot of T1 and T2* has shown a slope equal to 0.0009 (R2 = 0.0063).
(see figure 2).
For patients group,
Person’s correlation analysis showed positive weak correlation (r=0.286,
p=0.036) between T1 and T2*.
The plot of T1 and T2* has shown a slope equal to 0.0036 (R2 = 0.17).
(see figure 2).
Discussion
T1 and T2* mapping techniques have already been used to analyze significant changes in structural integrity of tendons [5,6,7-15]. In this work,
Achilles tendon of the same subjects was imaged using the two quantitative MRI techniques (T1 and T2* mappings),
and results of the patient group were compared with those of normal subjects to define the ability of these three techniques for the diagnosis of AT tendinosis.
Our study results indicate significant differences in T1 and T2* mean values between normal and patients group.
These differences reflect degeneration within the matrix of the tendon. The variation of T1 and T2* values between normal and patient group,
indicated no correlation for T1 and T2*. Correlation analysis of T1 and T2* showed a positive significant weak correlation for normal and patient group.
Ulrich Grosse and al.
have found T1 of 658.4 ms ± 21.8 % for asymptomatic non-tendinopathic tendon and 856.1 ms ± 28.4 % for symptomatic tendons using 3D-FLASH ultrashort echo time (UTE) sequence with Variation of flip angle at 3 Tesla [16].
Vladimir Juras and al.
investigate T2* in AT evaluate a regional variation of T2* in the healthy and pathologic tendon; concluded that in volunteers,
mean T2* was 3.35±0.45 ms and in patient,
mean T2* was 6.56±1.70 ms.
[17]
For T1 mapping,
our results showed an increase of T1 values as the degenerative change increased in the Achilles tendon of patient group.
Wright et al studied patient with spondylarthritis at the Achilles tendon,
indicated that T1 mapping has the potential to identify tendon abnormality earlier in comparison to conventional MR sequences by maximizing the sensitivity to subtle changes in the tendon structure [18]. For T2* mapping,
our results showed no correlation between normal and patient group. Vladimir Juras et al.
investigated T2* in the Achilles tendon (AT),
in vivo,
showed that T2* may be a promising marker for the diagnosis of pathological changes in the AT [17].
On an other hand,
Guinel H et al.
showed that T2* measurements in the normal Achilles tendon allowed correlation with structural status by histology [19].
Our findings showed a positive significant but weak correlation betweenT1 and T2* of normal and patient group. Grosse et al.
evaluated T1 and T2* at different Achilles tendon locations in a cohort of subjects with variable tendon straining activity.
In contrast to T1,
the Achilles tendon of healthy recreational runners showed higher mean T2* values to a control group [20].
T1 and T2* values in AT suggested significant variation between the three anatomical regions of interest (ROI).
The regional variation of T1 and T2* values is very clear for patients group compared to normal group.
The difference between our results and previous published studies can be explained by the differences in scanning parameters,
MRI exam conditions and population cohort.
Moreover,
results can be influenced by several factors including the magnetic field strength of the MR system,
the fitting algorithms and the unavoidable experimental noises.
The movement of protons in Achilles tendon is reliant on the orientation of the collagen fibers.
When the collagen fibers are oriented 55˚ to the B0 dipolar interactions are reduced and T2 is increased,
causing the so called magic angle effect [21,
22].
Henkel man and al.
reported that the mean T2 of in vitro dog Achilles tendon increases from 7 to 23 ms when the orientation changed from 0° to 55° [22].
Generally,
the magic angle effect is interpreted as an artifact and results an increased signal intensity in Achilles tendon on sequences with low TE such as T1-,
PD- and GRE sequences [23].
In our study,
the magic angle effect is minimized since we compared the same region in all subjects.
UTE sequences are specifically sensitive to the magic angle effect because of the short TE [24].
Therefore,
it is essential that the Achilles tendon is placed parallel to the magnetic field when using UTE sequences.
This problem is minimized in our study since we positioned all subjects in supine position and both feet in flexion position parallel to the axis of the table.
Our study presents to some limitations.
First,
the number of subjects is small.
Second,
the population is heterogeneous and not matched for age,
BMC and gender which limits the accuracy of the results analysis.
Third,
this study needs more histological information.
Further studies in larger patient groups with histologic correlation are helpful for the evaluation of the ability of quantitative MR techniques in early diagnosis of mechanical Achilles conditions.