Several pathogens cause pulmonary infections in immunocompromised patients determining specific HRCT findings.
There are four HRCT patterns useful in differential diagnosis of infections in immunocompromised patients: focal consolidation,
nodules/masses,
small micronodules and diffuse ground glass attenuation.
In addition,
each pattern can be considered separately between non-AIDS and AIDS groups because of the peculiar HRCT findings of each infection in AIDS patients.
FOCAL CONSOLIDATION
Focal consolidation pattern consists in a homogeneous increase of parenchymal attenuation that obscures the margin of vessels and airway walls,
air bronchogram may be present,
with focal distribution.
It can be expression of bacterial pneumonia (BP),
fungal infections and tuberculosis (TB).
Bacterial pneumonia
S.
aureus,
S.
pneumoniae and Haemophilus Influenzae are the most common pathogens in immunocompromised population.
Radiological features are similar to those seen in immunocompetent patients,
showing patterns of lobar pneumonia or bronchopneumonia.
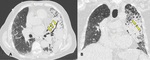
Lobar pneumonia appears on HRCT as homogeneus consolidation containing air broncograms with a segmental,
lobar or non-segmental distribution (Fig.
1); abscesses,
pneumatoceles and empyemas are relatively common complications particularly in S.
Aureus pneumonia.
Bronchopneumonia,
instead,
is characterized by bronchiolar wall thickening,
small ill-defined centrilobular nodules,
representing bronchioles impacted with inflammatory material and peribronchiolar inflammation,
and focal areas of consolidation.
Radiologic features of BP in patients with HIV infection can be similar to those in general population but almost half of patients present atypical HRCT findings as multifocal reticulonodular pattern with bilateral distribution.
Fungal infection
The typical HRCT pattern of fungal infections is characterized by solitary or multiple nodules,
but it is important to remember that less frequently these infections appear as segmental or subpleural wedge-shaped consolidations that correspond to hemorrhagic infarctions.
Tuberculosis
In immunocompromised patients mycobacterial infections can be caused either by TB or non-TB (atypical) mycobacteria and the radiological features of pulmonary infection depend on host’s immune status.
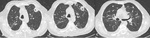
The typical presentation of primary TB is a mass or consolidation in the upper lobes that after caseous necrosis is followed by cavitation and then transbronchial spread to other parts of the lung that determine the presence of centrilobular nodules with or without tree-in-bud pattern (Fig.
2).
Frequently enlarged mediastinal nodes are present and typical tuberculous adenopathy is characterized by low attenuation with rim enhancement on contrast-enhanced CT.
It is important to underline that the peculiarity of primary TB in immunocompromised patients is the unusual location in basal segments of lower lobes,
anterior segment of the upper lobes or right middle lobe.
In severely immunocompromised patients other HRCT findings are a non-segmental distribution,
multiple cavities in a single lesion,
miliary nodules (miliary TB).
NODULES OR MASSES
On HRCT nodules appear as rounded or irregular opacity well or poorly defined measuring up to 3 cm in diameter while mass is defined as a lesion greater than 3 cm in diameter.
This pattern may be expression of fungal infection,
BP and viral infection.
Fungal infections
In immunocompromised patients there are two forms of Aspergillosis: angio-invasive (angio-IPA) and airway-invasive (airway-IPA).
Airway-IPA is more common than angio-IPA and tipically occurs in the setting of severe neutropenia and in patients with AIDS.
On HRCT bronchial invasive aspergillosis is characterized by the presence of centrilobular nodules and branching linear or nodular opacities,
giving an appearance resembling a tree-in-bud pattern.
These radiological manifestations are indistinguishable from those observed in broncopneumonias caused by other pathogens.
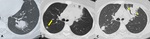
In AIDS patients airway-IPA appears as single or multiple upper lobe cavitary nodules with thick irregular walls (Fig.
3).
Angio-IPA occurs almost exclusively in patients with severe neutropenia and it is characterized by invasion and occlusion of small to medium pulmonary arteries by fungal hyphae with consequent hemorrhagic infarcts.
On HRCT early angio-IPA is characterized by the “CT-halo sign” that appears as nodules or pleural-based wedge-shaped areas of consolidation surrounded by a halo of ground-glass attenuation.
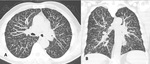
A later manifestation of angio-IPA is,
instead,
the “air-crescent sign” which is characterized by a crescent-shaped or circumferential area of radiolucency within parenchymal consolidation or nodules (Fig.
4).
Other than aspergillosis there are other forms of fungal infections such as criptococcosis,
mucormycosis,
candidiasis and nocardiosis.
The most common HRCT manifestation of pulmonary criptococcosis is solitary or multiple nodules which can be well-marginated or,
less frequently,
surrounded by ground-glass opacity.
Other radiologic findings include miliary nodules,
ground-glass opacity with a crazy-paving pattern and,
in non-AIDS patients,
airspace consolidation with cavitation.
Radiological findings of mucormycosis are very similar to those of pulmonary aspergillosis with solitary or multiple nodules and subpleural consolidation on HRCT.
Another frequent manifestation is the reversed halo sign characterized by ground-glass attenuation surrounded by consolidation.
Pulmonary candidiasis typically affects patients who are critically ill.
Candida causes pulmonary infections in two forms: hematogenous spread or aspiration.
On HRCT hematogenous type is very similar to IPA; however small centrilobular nodules are more frequent in IPA while larger nodules with a random distribution are more common in candidiasis. Candidiasis secondary to aspiration of oropharyngeal secretions,
instead,
is associated with broncopneumonia and appears on HRCT as peribronchial consolidation,
usually in a gravitationally dependent distribution.
The principal radiological features of pulmonary nocardiosis include multiple or solitary nodules,
masses and focal consolidation with frequent central cavitation. Endobronchial spread can occur after cavitation and is associated with small centrilobular nodules and bronchial wall thickening. In addition,
infection can extend to pleura and chest wall resulting in complications as pleural effusion or empyema and chest wall abscess.
SMALL MICRONODULES
Micronodules appear on HRCT as small rounded opacity with a diameter of less than 3 mm.
This pattern is not frequent in immunocompromised patients; micronodules can have either a random or centrilobular distribution ad can be expression of miliary TB,
histoplasmosis and viral infection.
Miliary tuberculosis
Hematogenous dissemination of TB results in miliary TB and in immunocompromised patients can occur either in primary and post-primary disease.
On HRCT it consists of a mixture of both sharply and poorly defined 1 to 4 mm nodules with random distribution.
Pleural effusion and mediastinal lymphoadenopathy may also be present (Fig.
5).
Histoplasmosis
HRCT appearance of pulmonary histoplasmosis is usually identical to miliary TB; in addition,
there are linear opacities and focal or patchy areas of consolidation.
Viral infection
Viruses are an important cause of infection in immunocompromised patients.
The most common pathogens involved are Citomegalovirus (CMV),
respiratory syncytial virus (RSV),
adenovirus,
herpes simplex virus and varicella-zoster virus.
Early phase of pulmonary viral infection represents a bronchitis or bronchiolitis resulting on HRCT in bronchial wall thickening and centrilobular nodules which may be surrounded by a halo of ground glass opacity such as in CMV and herpes simplex virus infections.
The next stage is represented by nectrotizing broncopneumonia that appears when viral infection affects the lung parenchyma adjacent to the terminal and respiratory bronchioles.
DIFFUSE GROUND-GLASS ATTENUATION
Ground glass opacity (GGO) is defined as slight increased opacity of lung with preservation of bronchial and vascular margins.
It may be expression not only of infectious disease such as Pneumocystis jiroveci pneumonia (PJP) and viral infection (fig.
6) but also of noninfectious disease,
for this reason differential diagnosis of this pattern may be difficult.
Pneumocystis jiroveci pneumonia
On HRCT extensive GGO is the major finding in PJP and corresponds to the presence of intra-alveolar fibrin,
debris and organisms.
It has been described a central distribution of GGO with relative peripheral sparing and a predilection for the upper lobes (Fig.
7).
The advanced disease may be characterized by septal lines with or without an intralobular septal lines superimposed on GGO,
showing the so called “crazy-paving” pattern” (Fig.
8).
Lung consolidation can also occur in this phase and tends to progress more rapidly in non-AIDS patients.
Another HRCT finding of PJP are pulmonary cysts of varying shape,
size and wall thickness associated with an increased frequency of spontaneous pneumothorax (Fig.
9).
Small nodules and tree-in-bud opacities,
instead,
are uncommon in PJP and usually indicate the presence of infectious bronchiolitis from other microorganisms.
In addition,
although rare,
interstitial fibrosis can occur in AIDS patients with low-grade chronic PJ and this condition is known as chronic Pneumocystis pneumonia.
Cytomegalovirus pneumonia
CMV pneumonia,
usually resulting from reactivation of a previous latent infection,
occurs most commonly in patients with bone marrow and solid transplantation or in patients with AIDS.
The radiologic manifestations are variable and usually consist of a mixture of pattern.
The most common HRCT findings are GGO,
consolidation and small nodules or masses with basal predominance; bronchial wall thickening,
bronchiectasis and septal thickening may be also seen (Fig.
10).
Furthermore,
in non-AIDS patients the HRCT-pattern differs from that observed in AIDS patients for the absence of very dense consolidation or masses.
These HRCT findings are very useful in differential diagnosis of CMV and PJ pneumonia because GGO with small nodules and consolidation are associated with CMV,
while extensive GGO with mosaic pattern and diffuse distribution is typical of PJP.