Component I
The pilot study was applied from 2011 to 2015 in the diagnostic radiology departments of two regional public hospitals,
considered representative because of the high risk/high volume procedures and for the potential benefit to practice and patient.
Special attention was given to breast imaging departments with good technical infrastructure and implemented quality control programmes in order to prove/enhance their ability to conduct breast screening programs.
It was created a multidisciplinary team of auditors comprising dedicated medical professionals of different specialties (radiologists,
radiographers,
radiation oncologist,
expert medical physicist and technical engineer) with high expertise in diagnostic radiology and with special interest in breast imaging.
Clinical audit was applied for the whole patient clinical pathway (structure,
process and outcome).
It was found that,
currently,
clinical audit comes across clinical activity already overloaded by accreditation and quality management evaluation systems that overlap in many aspects with it.
To simplify clinical audit process so that it does not overlap with the quality management system evaluation,
accreditation or regulatory inspections,
clinical audits were conducted focused on:
- justification of medical exposure procedures,
- optimization procedures for medical exposures,
by assessing the radiological image quality in relation to patient dose,
- clinical protocols for special procedures.
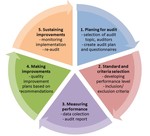
For all clinical audits applied the following five stages were performed [3] (Figure 1).
Figure 1.
The clinical audit cycle Fig. 1
Results obtained following the application of the pilot study (component I):
1.
Identification of nonconformities and improvement proposals (Table 1):
Table 1.
Examples of audit findings and improvements Table 1
2.
The breast imaging departments,
audited against specific criteria [7,
8],
proved their ability to conduct breast screening programs,
in terms of dedicated and experimented professionals,
technical infrastructure,
implemented quality assurance and quality control programmes,
number of patients addressed,
organizational and management expertise.
3.
Identification of numerous benefits of implementing clinical audit in radiological practice: increasing communication and awareness of best practices in health care facilities; practice optimization by increasing the benefit/risk ratio,
better standardization of procedures and practices; highlighting the needs in terms of resources.
4.
Highlighting some important issues related to the implementation in practice of clinical audit at national level such as: lack of fundamental understanding of the benefits of clinical audit application in practice,
clinical audits overloading activity due to overlap with other systems of quality management assessment,
accreditation and regulatory inspections,
lack of trained/certified external auditors,
shortages of qualified internal staff (in number and dedicated work time) need to clarify financing sources.
5.
Review of clinical audit procedures based on the feedback obtained after their first application taking into account the latest recommendations of the European Society of Radiology [4].
Component II
In order to elaborate a draft legislative act regarding clinical audit of medical activity with ionizing radiation were conducted the following activities:
1.
SWOT analysis on factors influencing the development of a draft legislative act on clinical audit in medical radiological practices (Table 2):
Table 2.
SWOT Analysis Table 2
2.
Drawing up a strategic plan on developing draft legislation act and establishing priority actions (Table 3) which set general objectives,
ways of achieving these objectives at national level (specific objectives) and priority activities.
Table 3.
Strategic plan – priority actions Table 3
Since the audit involves assessing radiological medical practice in relation to a referential,
there have been analyzed and selected international best practice standards and recommendations based on the latest scientific evidence specific for each type of radiological practice [5,
6,
7,
8].
Together with medical professional societies,
has been started the transposition of these recommendations,
in good-practice guidelines that were meant to be clear,
representative,
cost-effective,
flexible,
clinically applicable,
revisable,
capable for being transposed into explicit audit criteria,
which will be subject to Ministry of Health approval.
In order to increase awareness among medical specialists it has been developed an informative material on the need and benefits of clinical audit addressing both medical institutions with the personnel involvedand professional organizations.
In order to elaborate draft legislative act and harmonize national requirements on clinical audit with the European data were analyzed:
- ways of transposing EC requirements into national laws of European countries regarding audit program,
best practice standards,
training of auditors;
- the degree of practical implementation of these requirements;
- practical methods of clinical audit implementation [9].
It has been developed the draft legislative act which introduces compulsory implementation of clinical audit as professional and organizational responsibility at all public and private medical radiological practices and establishes the main responsibilities regarding the implementation and enforcement of the clinical audit.
Rules on clinical audit in medical radiological practices (diagnostic and interventional radiology,
nuclear medicine and radiotherapy),as a transposition of the "European Commission guidelines on clinical audit for medical radiological practices",
Radiation Protection no 159 [10],
describe the objectives of clinical audit,
the frequency of internal and external clinical audits,
the minimum requirements concerning topics audited,
the audit team,
the individual responsibilities,
the information from audit recordings,
and examples of standards and applicable good-practice criteria.
Since international recommendations on the criteria of good-practice and radiation protection in radiological practice and even those about the implementation of clinical audit are continuously changing,
in order to keep pace with the rapid development of technologies used in medical exposures,
it was considered useful that the rules on clinical audit in medical radiological practice should contain only general requirements for the implementation of the clinical audit,
and in the future to transpose the international recommendations into guides for each type of practice (diagnostic and interventionalradiology,
nuclear medicine and radiotherapy) which may be reviewed periodically,
in accordance with the latest international recommendations,
without the need to change the default order.
Results obtained in component II:
As a result of the activities conducted in component II of the Project were developed the following documents which will be submitted for approval by the Ministry of Health (Table 4).
Table 4. Developed national documents Table 4