Congress:
EuroSafe Imaging 2017
Keywords:
Radiation safety, Health policy and practice, Dosimetry, Plain radiographic studies, CT, Radioprotection / Radiation dose, Action 13 - Stakeholder engagement and collaboration, Action 2 - Clinical diagnostic reference levels (DRLs), Action 12 - Cooperation with international radiation protection initiatives and other stakeholders including IAEA, WHO, ICRP, HERCA, European Commission, Action 3 - Optimisation, diagnostic reference levels, image quality, Education and training, Workforce
Authors:
European Society of Radiology
DOI:
10.1594/esi2017/ESI-0030
Description of activity and work performed
The European Commission (EC) is aware that the transposition and implementation of the BSS Directive in the medical sector will constitute a major challenge for the respective national legislators and regulators in European Union Member States.
Therefore,
the EC approved a 15-month tender project in May 2016 to monitor the transposition of the BSS directive in the medical sector into Member States’ national legislation and to support its implementation.
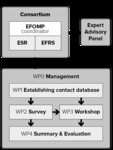
This project was awarded to a consortium headed by the European Federation of Organisations for Medical Physics ( EFOMP).
Other participating organisations are the European Society of Radiology (
EFOMP).
Other participating organisations are the European Society of Radiology ( ESR) and the European Federation of Radiographer Societies (
ESR) and the European Federation of Radiographer Societies ( EFRS).
EFRS).
In addition to the project management Work Package (WP0),
the project is broken down into four technical Work Packages and each WP covers specific tasks contributing to achieve the common objective to evaluate Member States’ activities for the transposition and implementation of the BSS Directive in the medical sector.
WP1: Identification of the competent authorities and other relevant organisations from each Member State,
Candidate State and EFTA State by establishing an electronic contact database.
WP2: Development and conducting of a survey to collect and analyse information on Member States' strategies and plans for the transposition of the BSS in the medical sector.
WP3: Organisation of a workshop to discuss findings of the survey and to identify the issues,
good practices and need for further actions.
WP4: Summary and evaluation of Member States' strategies and plans for the transposition of Council Directive 2013/59/Euratom in the medical sector.