Non-contrast CT and CT head angiogram/post-contrast CT:
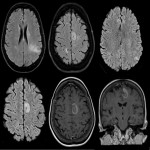
On non-contrast CT (NCCT), post-ictal findings are nonspecific including mild focal gyral swelling, effacement of cortical sulci, and decreased attenuation of the gyri, suggestive of edema. The contrast-enhanced CT images in CTP protocol are helpful in identifying underlying enhancing abnormality like brain tumors, vascular abnormalities, and previous infarct. CT angiogram assists further in demonstrating no large vessel occlusion and vascular territorial ischemia.
CTP:
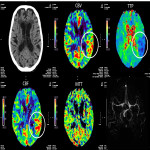
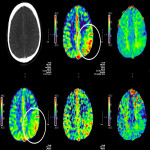
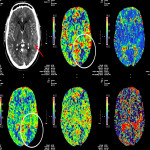
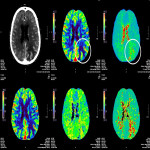
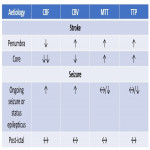
CTP findings in a classical stroke depicts hypoperfusion with increased TTP or MTT, reduced CBF and CBV in infarct. On the contrary, active seizure or status epilepticus is associated with hyperperfusion. The pattern of perfusion abnormalities includes focal cortical, multi-lobar, lobar, and hemispheric, and not respecting a particular vascular territory. Global hyperperfusion is possible in a generalised convulsive status epilepticus. See Figure 24, comparison of findings in acute ischemic stroke and seizure on CTP.
Perfusion abnormalities in the ictal and post-ictal spectrum are highly variable. Hyperperfusion may be seen in the epileptogenic zone and/or the propagation pathway during active seizure or status epilepticus. Depending on timing of CTP from onset of symptoms, changes can be as subtle as reduced TTP or MTT and normal CBF or CBV. In the post-ictal state, perfusion is usually normal. Once the seizure subsides, there may be decreased CBF in the epileptogenic zone. Bitemporal hyperperfusion though rare is because of inter-hemispheric propagation from one temporal lobe to the contralateral side, a described finding in temporal lobe epilepsy[i].
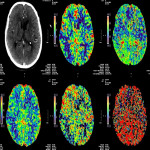
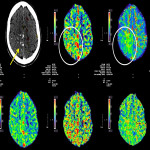
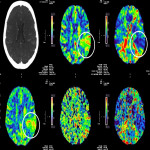
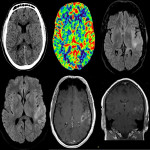
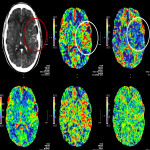
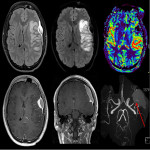
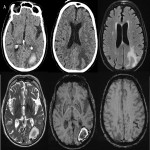
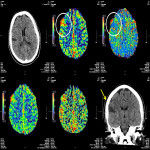
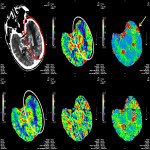
The three CTP characteristics are hyperperfusion (increased CBV or CBF, decreased TTP or MTT), hypoperfusion (decreased CBV or CBF, increased TTP or MTT), normal perfusion (normal CBV or CBF, normal/decreased TTP or MTT). Hyperperfusion pattern is illustrated in Figure 1, with EEG confirming status epilepticus. Hypoperfusion pattern is illustrated in Figures 2 and 4, in keeping with postictal state. Gugger et al documented 55% patients with focal neurological deficits ultimately diagnosed with seizures have normal perfusion, 23% had hyperperfusion and 21% had hypoperfusion pattern[ii].
MRI:
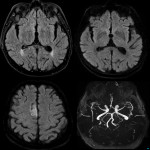
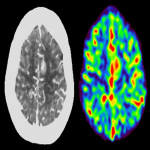
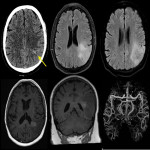
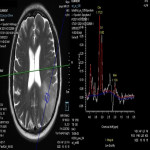
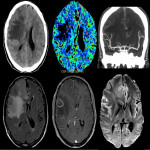
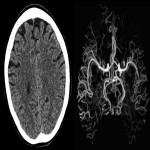
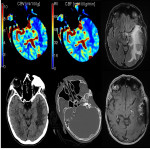
Postictal MRI findings include gyral swelling, increased signal intensity on T2-weighted and FLAIR images, and enhancement. Diffusion weighted imaging (DWI) is particularly useful as in acute phase cortical restricted diffusion is often seen as a result of cortical metabolic strain. This finding is often confirmatory with appropriate clinical history. MRA is complimentary to CTA in excluding vascular malformation and gold standard being DSA (Fig 8). Advanced neuroimaging, MR Spectroscopy, MR Perfusion is helpful in problem solving cases confirming or excluding neoplasm as a cause of seizures (Fig 14, 22). Structural findings such as hippocampal atrophy and hyperintense T2/FLAIR signal seen in temporal lobe epilepsy, or polymicrogyria, malformation of cortical development can also be appreciated.
Discussion:
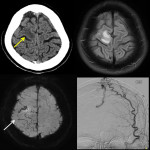
Causes of seizure can be classified as primary (idiopathic) and secondary (structural, metabolic, or reactive/inflammatory). Common secondary causes include trauma or insult, haemorrhage, vascular lesion, space-occupying mass, degenerative disease, infection, and hypoxia. Vascular subset includes but not limited to venous sinus thrombosis, arterial venous malformation, developmental venous angioma, cavernoma, and capillary telangiectasia. Figure 8 is an example of dural arterio-venous fistula resulting in adjacent oedema.
Figures 11, 14, 17, 19 are examples of glioblastomas presenting with acute seizures. Hence, it is important to correlate with the CT angiogram or post-contrast CT head for underlying vascular lesions or enhancing tumours.
Kramer et al highlights the relationship between cortical spreading depolarisation (CSD) and seizure [iii]. CSD is a disruption of the cortical electrical activity due to a loss of ion homeostasis, hence altering the threshold for seizure. Additionally, epileptiform activity and CSD can potentiate each other. Neurological insults such as seizure, subarachnoid haemorrhage, stroke, and traumatic brain injury disrupt the blood-brain barrier, resulting in activation of NMDA receptors, influx of intracellular cations and loss of normal membrane potential, hence leading to depression of cortical activity. The biochemical imbalance further lowers the threshold for epileptiform activity.
The relationship between vascular response and seizure is also relevant. As the depression of electrical activity accompanying CSD may be the cause of postictal state. The loss of ion homeostasis creates an acidic environment which promotes vasoconstriction. This correlates to the hypoperfusion pattern on CTP. Figure 23 is an example of radiographic left MCA vasospasm in the setting of subdural hematoma, requiring intra-arterial verapamil treatment.
Figure 9 illustrates a sequalae of cerebral oedema and lobar haemorrhage from CAA. As described above, abnormal vascular response from CSD may contribute to the delayed cerebral ischemia resulting from the haemorrhage, potentiating epileptiform activity. A subset of cerebral amyloid angiopathy, so-called CAA-related inflammation, often presents as headache, confusion, visual symptoms, or seizure. Inflammatory responses to the amyloid depositions in the tunica medial and adventitia leads to CSD events, hence resulting in seizure without intraparenchymal haemorrhage.
The hypermetabolic demands in sustained seizure results in vasogenic and/or cytotoxic oedema, resulting in cell death. On MR, the most common findings are increased T2/FLAIR signal, restricted diffusion having a gryiform pattern.
Figure 4 is an example of seizure secondary to severe electrolyte derangement requiring aggressive calcium, potassium and phosphate replacement. The decrease in magnesium and potassium level results in imbalance of the sodium and calcium channels via NMDA receptors, resulting in osmotic shift, damaged neurons, and cortical activity depression.
The difficulty lies in differentiating hypoperfusion seizure from acute ischemic stroke which both have reduced perfusion and MTT/TTP. The most consistent differentiator between seizure-related hypoperfusion and stroke is that the former does not respect vascular territory and hypoperfusion is only modest and not as severe as in ischemic stroke. The distribution of perfusion abnormality is almost always cortical or subcortical whereas in ischemia it, involves both cortex and white matter (Fig 24). Multilobar and hemispheric distribution sometimes can be an intriguing finding. In hemispheric perfusion abnormality, it is essential to correlate with CT angiogram (CTA) for absence of vessel occlusion.
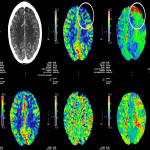
Post-stroke seizure is clinically challenging as 29% are non-convulsive seizures, hence CTP may be helpful[iv]. There is compelling evidence documenting the frequency and duration of CSD events are associated with the size of the infarct[v]. Figure 6 illustrates the hyperperfusion characteristic favouring a seizure focus adjacent to the region of gliosis.
Risk of seizures varies between 40-60% in brain tumours, occasionally presenting as stroke mimic[vi].
As described above, cerebral insult is associated with CSD events, including radiation necrosis (Fig 20). The pathophysiology of radiation necrosis is multi-factorial, including disruption of vascular permeability, fibrinoid necrosis of small vessels, and glial cells demyelination. Hypoxia caused by endothelial cell damage leads to VEGF production, inducing neo-angiogenesis of leaky capillaries and resulting in oedema. Management options include, steroid, antiepileptics, bevacizumab, hyperbaric oxygen therapy or resection.