Mesenteric fibromatosis (MF) is an example of an idiopathic fibrosing disorder, a group of locally aggressive fibroproliferative conditions which can infiltrate and recur. They can occur in the mediastinum, small and large bowel and mesentery. [1]
MF can occur throughout adulthood with no race or gender predilection. Interestingly, 13% of patients with MF are found to have the Gardner variant of familial adenomatous polyposis, with previous abdominal surgery as an important risk factor for the development of MF. Bowel obstruction, fistula formation and perforation are complications of MF. Mucosal ulceration can also occur due to vasculature compromise. Homogeneous soft-tissue attenuation is seen in tumours with collagen-dense stroma; in contrast, hypodense/hypointense appearances are common with a myxoid stroma. CT and MRI contrast enhancement can be homogeneous or heterogeneous. [1]
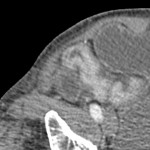
An 82 year old man presented to A&E with nausea, vomiting, abdominal pain and PR bleeding with no significant background abdominal or surgical history. Basic bloods were grossly unremarkable and physical examination elicited tenderness in the right lower abdomen. On CT, there was short segment hyperenhancement and thickening of the terminal ileum with luminal narrowing, and resultant bowel obstruction. The patient underwent a right hemicolectomy, pathological specimens revealed MF. Post-operative recovery was unremarkable.
Rosai-Dorfman disease (RDD), also known as sinus histiocytosis with massive lymphadenopathy, is a rare non-Langerhans cell reactive histiocytic disorder characterised by macrophage proliferation. [2, 3] Although idiopathic, autoimmune diseases and viruses such as human herpesvirus 6 and Epstein-Barr virus have been postulated as potential causes. The pathogenesis involves recruitment of marrow monocytes from the peripheral blood into lymph node sinuses or extranodal sites with subsequent transformation into the immunophenotypically distinct RDD histiocytes. These histiocytes can release cytokines, such as tumour necrosis factor–α, which result in fever and other systemic symptoms. [2]
There is a slight male predominance, with children and young adults most commonly affected; there also is a predilection for those of West African and Caribbean descent. [2, 3] The most common clinical presentation of RDD is non-specific painless lymphadenopathy (most commonly cervical) accompanied by fever, an elevated erythrocyte sedimentation rate, and mild anaemia. Extranodal disease occurs in nearly half of patients and can involve nearly every organ system, with the head and neck being the most common sites of involvement. [2, 3]
Although variable, overall RDD has a good prognosis; many patients have an indolent clinical course and even spontaneous resolution. Extranodal disease correlates with a worse prognosis. Surgical resection is the mainstay of treatment, if deemed appropriate because of vital organ involvement or complications. Radiotherapy and long-term corticosteroid therapy have been used with mixed results, while chemotherapy has generally proven ineffective. [3]
Lymphadenopathy has non-specific imaging appearances and often enhances. [3] Uptake of gallium and FDG has been demonstrated in the abnormal lymph nodes. Imaging manifestations of RDD overlap with a variety of neoplastic conditions (lymphoma, Kaposi sarcoma and carcinoma metastases) and infectious/inflammatory conditions (tuberculosis, Wegener granulomatosis and sarcoidosis). There is typically enhancement of the involved organ with an infiltrative or mass-like appearance. [2]
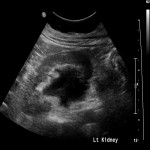
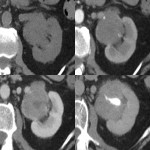
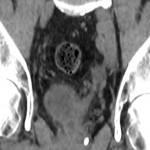
A 53 year old female presented to her GP with a few day history of lower abdominal pain, diarrhoea and fevers with no significant background history. An US study revealed a large well-defined homogeneous hypoechoic renal hilar mass. Subsequent multiphase CT demonstrated delayed homogeneous enhancement and narrowing of the traversing ureter without causing obstruction. A 36 mm soft tissue mesenteric mass was also seen in the right pelvis with similar enhancement characteristics. RDD of the renal hilar mass was confirmed on CT guided biopsy and the patient was discharged without further intervention. Both masses are stable on follow up imaging.
Inflammatory myofibroblastic tumour (IMT), also known as inflammatory pseudotumour, is a rare lesion characterized by reactive non-neoplastic proliferation of myofibroblastic spindle cells and inflammatory cell infiltration within a myxoid stroma. Some authors consider IMT a low-grade fibrosarcoma with inflammatory cells. IMT can occur in nearly every site in the body and is thought to result from inflammation following minor trauma/surgery. Immune/autoimmune and infectious causes have also been proposed. Organisms have been found in IMTs, such as Epstein-Barr virus in nodal disease and mycoplasma in pulmonary disease. [1]
IMT most commonly affects children and young adults. Hepatic IMT occurs mainly in Asian countries. Patients can present with fevers, thrombocystosis and hypergammaglobulinaemia. Organ-specific signs and symptoms have also been noted, such as obstructive jaundice and cholangitis in hepatic IMT. [1]
IMT has non-specific appearances with heterogeneous and variable enhancement on post-contrast CT (often delayed and persistent due to fibrous content). Calcifications can occur and are more common in children. Depending on the organ of involvement, IMT can appear hyper or hypoechoic on US and have varying signal characteristics on MRI. A wide range of non-aggressive (such as well circumscribed margins) and aggressive (such as ulceration and infiltrating margins) features have been described. [1]
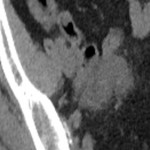
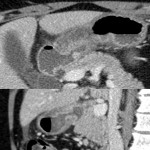
An 82 year old man presented to his GP with lower abdominal discomfort and impaired renal function on a background of recurrent low grade urothelial cancer of the bladder and chronic kidney disease. A non-contrast CT study revealed soft tissue thickening and luminal narrowing of the left distal ureter. There was marked proximal hydroureteronephrosis without stranding. The biopsy taken via cystoscopy yielded IMT. The patient remains under active surveillance.
A 30 year old man presented to A&E with a 4 hour history of worsening sharp stabbing abdominal pain, which was worse on movement and radiated to the shoulders. This was on a background of excessive NSAID use, smoking and occasional ETOH. Bloods revealed deranged electrolytes and mildly elevated lipase and WCC. Upper abdominal free gas and fluid was seen on CT, with marked oedematous thickening of the gastric antrum, initially thought to be an ulcer. The patient underwent a laparotomy and washout with sewing of the ulcer, pathological specimens revealed IMT. Post-operative recovery was unremarkable.