Keywords:
Artificial Intelligence, Oncology, Radiation physics, Experimental, Image manipulation / Reconstruction, Neural networks, Brachytherapy, Dosimetry, Radiation therapy / Oncology, Cancer, Quality assurance, Radiotherapy techniques
Authors:
K. Koo, M. Hanlon, R. Smith, J. Millar, H. Elsaleh; Melbourne, VIC/AU
DOI:
10.26044/ranzcr2021/R-0456
Results
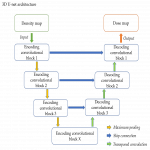
Monte Carlo dosimetry was generated for 10 random source positions in a water phantom, 8 of which were used in model training. We found network depth, choice of loss function and initial neuron weights to be the most critical model hyperparameters requiring optimisation. As an example, the impact of network depth on model training speed and final accuracy are illustrated in Figure 2. We see increasing accuracy with deeper networks, but there are diminishing returns beyond 3 layers. Nevertheless, a 6-layer network, being the most accurate, was selected as the final model for presentation.
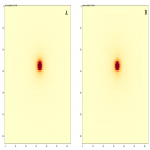
When we applied the trained model to the validation set, dosimetry prediction for a single source took an average of 0.12 seconds (range 0.10-0.15 seconds). The resulting dose map (Figure 3B) resembles the ground truth data (Figure 3A), with less than 1% of all voxels having dosimetric errors greater than 2% of the putative prescription dose, but there remains significant absolute percentage error in low dose voxels.
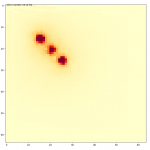
Even though all training and validation data utilised a single source position, we tasked the model with predicting dose for three simultaneous positions. The model successfully produced a dose map for all sources, taking no longer to do so than for a single position (Figure 4).