Anatomy and Function:
Perivascular spaces (PVS) in the brain are channel-like extensions of the extracerebral fluid space. Interposed between penetrating cerebral blood vessels and the surrounding parenchyma, these fluid-filled spaces are lined by the invaginating pial membrane and a barrier of astrocytic endfeet with tight junctions, the glia limitans. The walls of the PVS differ between anatomical regions; Type 2 ‘Cortical’ PVS through the cortical grey matter and subcortical white matter have a single leptomeningeal layer, whereas Type 1 PVS centred in the basal ganglia along the lenticulostriate vessels have an additional layer against the blood vessel wall.1
PVS allow interstitial fluid (ISF) transport alongside the penetrating vessels and allow the exchange of CSF and ISF with the surrounding brain parenchyma. This exchange is regulated by Aquaporin 4 (AQP4) water channels that are predominantly localized to the astrocytic endfeet of the PVS surrounding penetrating arteries, capillaries and veins.2 PVS flow has also been shown to correlate closely with arterial pulsation in line with the cardiac cycle.3 The arterial pulsation is believed to increase the CSF/ISF exchange rate between the PVS and the surrounding parenchyma.4 This process is postulated to create a flow of interstitial fluid through the parenchyma towards the perivenular spaces, allowing for clearance of solutes from the parenchyma such as amyloid-beta(Aβ).2, 5, 6 This functional CNS drainage role has been termed the ‘glymphatic system’.
Pathophysiology:
Previous studies have noted the correlation between PVS dilatation and their quantity with neurodegenerative disorders, vascular pathologies, metabolic disorders, chronic alcoholism as well as normal ageing.7-9 A build-up of proteins in the parenchyma and along the microvasculature is a feature of multiple neurodegenerative diseases, such as Alzheimer’s Disease (AD) with Aβ.10, 11 Impaired function of the glymphatic system may lead to an accumulation of waste proteins in the parenchyma and along PVS causing dilatation of these channels.
Sleep influence on PVS:
Animal studies have demonstrated an increase in CSF/ISF exchange, a 60% increase in interstitial volume and an increase in waste clearance during sleep.12, 13 Sleep deprivation studies in animals and humans have shown an increase in Aβ levels in the CSF, the hippocampus, and the thalamus after one night of sleep deprivation.14 It is postulated the increase in interstitial volume allows for more efficient CSF/ISF exchange promoting solute clearance.13 Sleep deprivation has also been noted to cause AQP4 depolarization, which reduces CSF/ISF exchange along the PVS.15
Effects of Ageing:
The ageing brain has reduced cerebral arterial wall pulsatility, loss of perivascular AQP4 water channel polarity, and the reduced clearance of interstitial solutes such as Aβ and tau, all of which reduce the effectiveness of the glymphatic system.10,16 Hypertension increases arterial stiffness and reduces perivascular pumping, which decreases the overall flow in the PVS.3
Effects of Cerebrovascular Accidents (Stroke) on the glymphatic system:
Mouse and primate studies using contrast-enhanced MRI have demonstrated severe deficits in glymphatic perfusion and solute clearance after subarachnoid haemorrhage (SAH) and ischemic strokes. Intraventricular injection of fibrinolytic agents after SAH improved glymphatic perfusion and reduced histological injury in rodent brains.16, 17
Neurodegenerative Diseases:
A buildup of waste proteins is a common feature of Alzheimer’s disease (AD), Parkinson’s disease, ALS, and other neurodegenerative diseases.11, 18 CSF clearance is reduced in patients with AD with an inverse relationship with Aβ accumulation.19 Disruption of AQP4 channel distribution along PVS was associated with reduced cognition and AD.20 Thus, there has been considerable research interest in evaluating PVS metrics as a biomarker for neurodegenerative disease. In Parkinson’s disease, PVS volume and visible number are increased in affected regions from the early stages of disease progression.22 In Multiple sclerosis, enlarged PVS are a feature associated with cognitive decline.21
Traumatic Brain Injuries:
TBI is a risk factor for neurodegenerative disease later in life.22 In mice, TBI causes significantly reduced perivascular CSF/ISF exchange and decreased parenchymal clearance of tracers along with disruption of perivascular AQP4 channels.23 TBI have been found to lead to an increased accumulation in phosphorylated tau.23
Imaging of Perivascular Spaces:
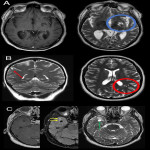
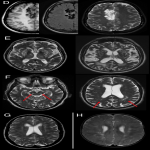
Enlarged PVS are described on MRI as well-circumscribed T2 hyperintense, T1 hypointense lesions with no associated enhancement or diffusion restriction.1 They follow CSF signal on all MRI sequences.1 Though predominantly seen in older age groups as part of normal aging brain, or related to vascular and neuroinflammatory disease or TBI25, 26, they are also seen on routine MRI as an incidental finding.
Although uncommon in routine practice, visual scoring systems of PVS exist, which include parameters describing their number (multiplicity); location (anatomical region); size (maximum diameter) and shape on Axial T2 Weighted imaging.8 Combining T1 and T2 weighted imaging has been used to increase the visibility of PVS for quantification.27
Glymphatic Imaging:
Animal studies using tracers injected into CSF spaces demonstrate distribution along perivascular channels on light microscopy of serial brain slices. Tracer entry into parenchyma was dependent upon arterial pulsations.28 In vivo, two-photon imaging using intracisternal fluorescent tracer injection in mice demonstrate the flow of CSF down perivascular spaces of arteries and exchange with the surrounding ISF.2 Dynamic contrast-enhanced MRI has helped visualise CSF/ISF exchange in perivascular spaces and interstitial solute clearance in rodents.5
Tracer studies of the glymphatic system rely on intrathecal injection of gadolinium-based contrast agents (GBCA), not possible in human trials due to the risk of gadolinium toxicity at high doses.29, 30 However, it has been approved for studying select conditions including idiopathic normal pressure hydrocephalus (iNPH).6 This population demonstrated similar CSF flow along PVS as in rodents with glymphatic flow also increasing during sleep. Interestingly, patients with iNPH had delayed tracer clearance compared to controls.6, 31
Human intravenous GBCA tracer MRI studies demonstrate an increased leakage rate into the CSF space in patients with AD, visualised in the CSF and parenchyma with heavily T2 weighted and FLAIR imaging.32, 33 PET-MRI studies in humans, with intravenous radiotracers crossing the blood-brain-barrier, demonstrate impaired CSF distribution leading to increased deposition of Aβ in specific brain regions from a single night of sleep deprivation.14, 19
Multiple MRI techniques are available to evaluate CSF flow that do not rely on injected contrast. However, many have not yet been used for evaluating flow in PVS. These include spatial modulation of magnetization (SPAMM), Time-spatial labelling inversion pulse (Time-SLIP) MRI, and phase contrast methods such s 4D-PC.34-36 Multi-echo time arterial spin labelling (ASL), has been used to non-invasively assess glymphatic flow through AQP4 channels.37 Diffusion Tensor Imaging (DTI) has been adopted in a technique called Diffusion Tensor Image Analysis Along the Perivascular Space (DTI-ALPS) to measure the motion of water movement along PVS.38 In certain regions of the brain, perivascular channels are perpendicular to the fibre tracts, allowing quantification of perivascular diffusivity independent of the fibre tract diffusivity.38 Perivascular diffusivity was significantly correlated with mini-mental state examination in patients with AD, and with severity of iNPH.39 DTI studies of PVS fluid movement have demonstrated with cardiac gating that cerebral vascular pulsation drives flow.40 DTI and structural imaging methods have been used together to provide an objective analysis of PVS changes to investigate their role as imaging markers of disease.22

![Fig 1: [Adapted from Ramirez et. al.] Illustration of the structure and physiology of perivascular spaces and the glymphatic system. The perivascular spaces surround the penetrating blood vessels. The glia limitans creates a barrier between the PVS and the parenchyma with AQP4 channels. It demonstrates a potential cause for an enlarged perivascular space where metabolic waste and inflammatory cells disrupt the outflow of PVS causing an increase in the diameter of the channel.7](https://epos.myesr.org/posterimage/ranzcr/ranzcr2021/159047/media/905508?maxheight=150&maxwidth=150)
![Fig 2: [Adapted from Rasmussen et. al.] This model demonstrates some of the examples of disruptions of PVS and the glymphatic system. CSF flow occurs from periarterial spaces filtering through the parenchyma towards the perivenular spaces. 20](https://epos.myesr.org/posterimage/ranzcr/ranzcr2021/159047/media/905509?maxheight=150&maxwidth=150)
![Fig 5: [Adapted from Rasmussen et. al.24] illustrate the range of imaging modalities used to assess PVS and the glymphatic system. Ex vivo imaging using microscopy in brain slices of animal models with 20 μm to 100 μm thickness after injection of tracer into CSF space. Two-photon laser scanning microscopy is used with a surgical bone window created in living animals to assess PVS flow with high spatial resolution.24](https://epos.myesr.org/posterimage/ranzcr/ranzcr2021/159047/media/905563?maxheight=150&maxwidth=150)
