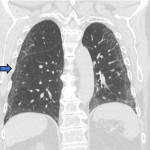
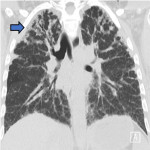
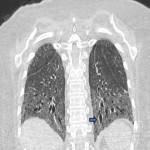
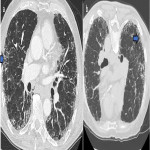
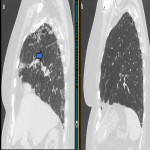
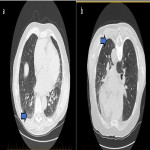
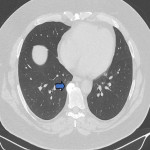
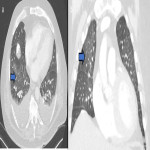
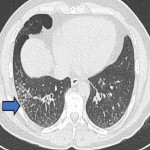
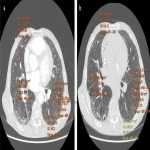
ILA is subclassified based on the zones of the lung affected (upper, mid and/or lower), its distribution (peripheral or central) and whether fibrosis is present [1,2]. Examples of ILA in different locations are shown in Figures 1 and 2.
Fibrosis is indicated by architectural distortion and traction bronchiectasis (shown in Figure 3), with or without honeycombing (shown in Figure 4) [1,2].
Fissural retraction is also a useful feature of fibrosis (shown in Figure 5).
Other findings of ILA include ground-glass opacities (shown in Figure 3), reticular opacities (shown in Figure 4), and non-emphysematous cysts [1,2].
When peripheral reticulation is observed, it is important to ensure that the findings are not due to dependent changes which are not ILA. This can be achieved by reviewing for changes between supine and prone studies, as ILA should persist in both positions [1,2]. Figure 4 shows an example of non-dependent change of ILA, whereas an example of dependent change is illustrated in Figure 6.
Other findings which are not considered ILA include paraspinal fibrosis in association with osteophytes (shown in Figure 7), interstitial oedema (shown in Figure 8), centrilobular nodules (Figure 9) and patchy ground-glass and tree-in-bud opacities [1,2].
Patients over the age of 65 can have senescent lung changes that should be accepted as normal variants rather than being attributed to pathology [3]. These include areas of ground glass opacities, especially near osteophytes, mosaic attenuation and hyperexpansion. As long as patients are asymptomatic, and if findings are stable based on progress studies, they can be considered paraphysiological.
Before deciding if there is ILA, adequacy of technique needs to be assessed. Post-contrast scans can lead to increased density of the lung parenchyma, occasionally leading to the erroneous interpretation that there is ground glass density, as shown in Figure 10 [4]. Having a consistent window and width level can help sensitivity.
Management of ILA:
Once ILA is found, the first step is to evaluate for potential ILD. ILD should be considered if there is extensive disease affecting multiple zones on CT, or if the patient has supportive findings on clinical examination or PFT. If any of these factors are present, management is delegated to a respiratory specialist in conjunction with the multidisciplinary team [1].
If there are no features to suggest ILD, the radiologist should look for ILA patterns associated with an increased risk of disease progression [1,2]. These are:
- Nonfibrotic ILA with basal and peripheral predominance
- Fibrotic ILA with basal and peripheral predominance without honeycombing
- Fibrotic ILA with basal and peripheral predominance with honeycombing
In these cases, or if there is a clinical risk factor for progression (such as smoking or chemoradiotherapy), active monitoring is an appropriate approach. This involves repeat PFT and clinical examination in 3-12 months and repeat CT at 12-24 months, or sooner if there is evidence of deterioration. In the absence of risk factors for progression, the management of ILA is conservative with patient education and reassessment if symptoms arise [1,2].