Keywords:
Lung, CT, Stereotactic radiotherapy, Cancer
Authors:
C. Chong, H. Tan, C. Harper, R. Chee, E. Ng, S. Mincham, C. Vaz, E. Leong, T. Lim
DOI:
10.26044/ranzcr2022/R-0088
Methods and materials
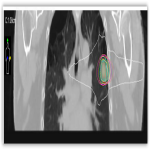
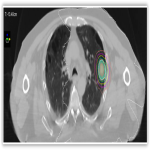
This institutional research ethics board approved study with biopsy proven Stage I NSCLC treated with SABR between September 2010 to November 2018 were retrospectively reviewed/
All patients were discussed at the thoracic multidisciplinary meeting and had attempted biopsy unless deemed medically unsafe. Patients received treatment as per departmental protocol and were inversely planned with Pinnacle and Monaco. All patients were treated to a dose of 48Gy in 4 fractions (12 Gy per fraction), 2 fractions per week over 2 weeks with dynamic conformal arc therapy (DCAT) utilising intensity-modulated radiation therapy, prescribed to the 65-90% isodose. Treatment prescriptions of 52Gy/4#, 60Gy/8#, 50Gy/8# and 54Gy/10# were also utilised.
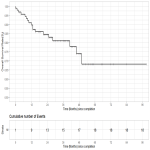
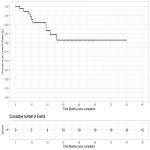
Competing risks analysis was used to estimate the rate of local control (LC) using death as a competing risk factor.
Overall survival (OS) and progression-free survival (PFS) were estimated using the Kaplan-Meier method.
Predictors of LC, OS and PFS were evaluated using univariable Cox regression and Fine & Gray competing risks regression, respectively.
The SABR- related toxicities were recorded using common terminology criteria for adverse events (CTCAE v.5).