Purpose
With an increasingly ageing population, at least 25% of patients presenting with early stage NSCLC are not amenable to surgical resection (1). Over the past 10 years stereotactic ablative radiotherapy (SABR) or Stereotactic Body Radiotherapy (SBRT) is fast becoming a curative option for these patients with improvements in technology and accuracy in delivery.
To evaluate the clinical outcomes and toxicity profile of Stage I NSCLC patients treated with SABR in our institution
Methods and materials
This institutional research ethics board approved study with biopsy proven Stage I NSCLC treated with SABR between September 2010 to November 2018 were retrospectively reviewed/
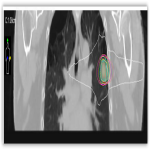
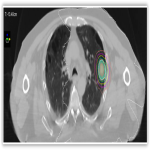
All patients were discussed at the thoracic multidisciplinary meeting and had attempted biopsy unless deemed medically unsafe. Patients received treatment as per departmental protocol and were inversely planned with Pinnacle and Monaco. All patients were treated to a dose of 48Gy in 4 fractions (12 Gy per fraction), 2 fractions per week over 2 weeks with dynamic conformal arc therapy...
Results
Patient Characteristics
A total of 93 patients were included in the analysis and the mean follow up was 30.3 (range, 1-99) months. SABR doses ranged from 48 - 60Gy in 4 -10 fractions and the mean biological equivalent dose (BED10) was 105.3Gy.
The median age was 77 (range, 54-94) years. There were 50 patients with tumour size less than 2cm and 42 patients with tumour size > 2cm. Majority of histology subtype was adenocarcinoma (n=57).
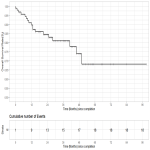
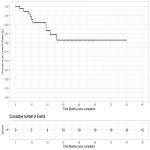
Local Control and Overall Survival
The 1, 2 and 3-year...
Conclusion
SABR remains an effective and safe treatment modality for medically inoperable Stage I NSCLC, with an excellent early and medium-term local control and survival, with acceptable toxicity as illustrated in our study. Our findings add to existing body of literature to support the utility of SABR in medically inoperable Stage I NSCLC.
References
1.Ricardi U, Filippi AR, Guarneri A, Giglioli FR, Ciammella P, Franco P, et al. Stereotactic body radiation therapy for early stage non-small cell lung cancer: results of a prospective trial. Lung Cancer. 2010;68(1):72-