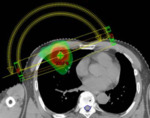
Learning objectives
Describe the clinical background of targeted lung cancer therapies,
namely targeted chemotherapy,
immunotherapy,
stereotactic radiotherapy and image guided percutaneous ablation.
Outline novel patterns of treatment response observed in patients receiving targeted therapies.
Recognize the imaging findings of adverse events associated with targeted therapies.
Background
Lung cancer is a leading cause of cancer related morbidity and mortality.
Historically medical therapy for advanced lung cancer relied on cytotoxic chemotherapy.
Drug development focused on specific cell signaling pathways,
coupled with the molecular characterization of tumor subtypes has led to the development of targeted drugs Fig. 1.
More recently,
immunotherapy has emerged as a promising treatment strategy for lung cancer.
Recent developments in interventional oncology and radiation therapy such as percutaneous ablative therapies and novel forms of radiation therapy includingstereotactic body radiation therapy...
Findings and procedure details
Targeted Therapies
Targeted medical treatments act on molecular signaling pathways crucial to cancer cell survival; and are more specific in targeting tumor cells.
Drug nomenclature reflects the site of action of the drug Fig. 5; which can be broadly divided into monoclonal antibodies acting on the cell surface/circulating growth factors,
and small molecule inhibitors acting within the cell Fig. 1.
The targeted drugs most commonly used in lung cancer are agents targeted at specific genetic aberrations,
anti-angiogenic agents and checkpoint inhibitor immunotherapy.
Agents targeted at...
Conclusion
When assessing patients receiving targeted lung cancer therapies,
radiologists need to be aware of nontraditional pattern of tumor response and adverse events.
Personal information
E.
O'Dwyer,Department of Radiology,
Memorial Sloan Kettering Cancer Center,
1275 York Ave,
New York,
10065,
USA
email:
[email protected]
D.
Halpenny,Department of Radiology,
Memorial Sloan Kettering Cancer Center,
1275 York Ave,
New York,
10065,
USA
References
Ding PN et al: Risk of treatment-related toxicities from EGFR tyrosine kinase inhibitors: A meta-analysis of clinical trials of gefitinib,
erlotinib,
and afatinib in advanced EGFR-mutated non–small cell lung cancer.
J Thorac Oncol 12:633-643,
2017
Gemma A et al: Final safety and efficacy of erlotinib in the phase 4 POLARSTAR surveillance study of 10 708 Japanese patients with non–small-cell lung cancer.
Cancer Sci 105:1584-1590,
2014
Choi H et al.
Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated...