Nodular parenchymal pulmonary amyloidosis:
Epidemiology:
This is the most common manifestation of pulmonary amyloidosis. Nodular parenchymal amloidosis is typically a feature of primary pulmonary/localized amyloid and therefore occurs in the absence of systemic amyloid.
Clinical findings:
Nodular parenchymal amyloidosis is usually an incidental finding on CXR. However it can cause mass effect symptoms such as cough, haemoptysis or recurrent infections due to airway obstruction.
Radiological findings:
· single/multiple nodules
· nodules and masses varying in size from 5mm to 15cm1
· location is usually usually subpleural or peripheral
· contour may be smooth or slightly lobulated
· spiculated amyloidomas have been described which resemble carcinoma3
· can calcify (uncommon, 20% in one series 3, up to 50% in others1)
· cavitation may occur rarely
· remain stable over time, may increase but never regress
· may be associated with lymphadenopathy or pleural effusions (the latter not due to cardiac failure secondary to senile cardiac amyloid)
· PET: Parenchymal nodules may be positve or negative for 18-FDG uptake, which emphasises the importance of tissue biopsy to confirm suspected malignancy before instituting curative treatment 8,9,10,11.
Fig 1a:
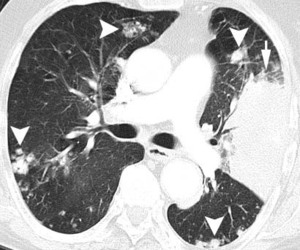
Fig.: 56-year-old man with biopsy-proven localised pulmonary amyloidosis. Axial CT (lung windows, a, c and e; bone windows, b, d and f) shows multiple bilateral pulmonary nodules that are predominantly peribronchovascular and lower zone in distribution, with both lobulated and spiculated contours (arrowheads). Many of these pulmonary nodules are partially calcified. There is also a more confluent area of consolidation in the lingula which appears hyperdense and contains intenal fleck of calcium (arrows, a and b). The appearances may easily mimic disseminated pulmonary metastases, pulmonary lymphoma or even invasive or semi-invasive fungal pneumonia.
Fig 1b
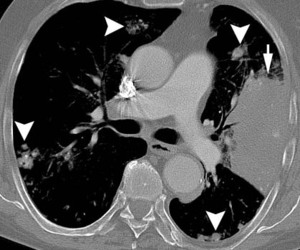
Fig.: 56-year-old man with biopsy-proven localised pulmonary amyloidosis. Axial CT (lung windows, a, c and e; bone windows, b, d and f) shows multiple bilateral pulmonary nodules that are predominantly peribronchovascular and lower zone in distribution, with both lobulated and spiculated contours (arrowheads). Many of these pulmonary nodules are partially calcified. There is also a more confluent area of consolidation in the lingula which appears hyperdense and contains intenal fleck of calcium (arrows, a and b). The appearances may easily mimic disseminated pulmonary metastases, pulmonary lymphoma or even invasive or semi-invasive fungal pneumonia.
Fig 1c:
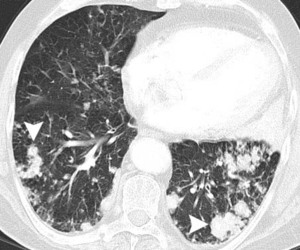
Fig.: 56-year-old man with biopsy-proven localised pulmonary amyloidosis. Axial CT (lung windows, a, c and e; bone windows, b, d and f) shows multiple bilateral pulmonary nodules that are predominantly peribronchovascular and lower zone in distribution, with both lobulated and spiculated contours (arrowheads). Many of these pulmonary nodules are partially calcified. There is also a more confluent area of consolidation in the lingula which appears hyperdense and contains intenal fleck of calcium (arrows, a and b). The appearances may easily mimic disseminated pulmonary metastases, pulmonary lymphoma or even invasive or semi-invasive fungal pneumonia.
Fig 1d:
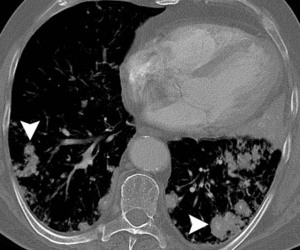
Fig.: 56-year-old man with biopsy-proven localised pulmonary amyloidosis. Axial CT (lung windows, a, c and e; bone windows, b, d and f) shows multiple bilateral pulmonary nodules that are predominantly peribronchovascular and lower zone in distribution, with both lobulated and spiculated contours (arrowheads). Many of these pulmonary nodules are partially calcified. There is also a more confluent area of consolidation in the lingula which appears hyperdense and contains intenal fleck of calcium (arrows, a and b). The appearances may easily mimic disseminated pulmonary metastases, pulmonary lymphoma or even invasive or semi-invasive fungal pneumonia.
Fig 1e:
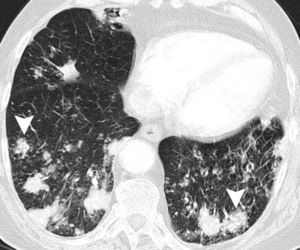
Fig.: 56-year-old man with biopsy-proven localised pulmonary amyloidosis. Axial CT (lung windows, a, c and e; bone windows, b, d and f) shows multiple bilateral pulmonary nodules that are predominantly peribronchovascular and lower zone in distribution, with both lobulated and spiculated contours (arrowheads). Many of these pulmonary nodules are partially calcified. There is also a more confluent area of consolidation in the lingula which appears hyperdense and contains intenal fleck of calcium (arrows, a and b). The appearances may easily mimic disseminated pulmonary metastases, pulmonary lymphoma or even invasive or semi-invasive fungal pneumonia.
Fig 1f:
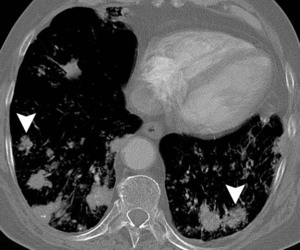
Fig.: 56-year-old man with biopsy-proven localised pulmonary amyloidosis. Axial CT (lung windows, a, c and e; bone windows, b, d and f) shows multiple bilateral pulmonary nodules that are predominantly peribronchovascular and lower zone in distribution, with both lobulated and spiculated contours (arrowheads). Many of these pulmonary nodules are partially calcified. There is also a more confluent area of consolidation in the lingula which appears hyperdense and contains intenal fleck of calcium (arrows, a and b). The appearances may easily mimic disseminated pulmonary metastases, pulmonary lymphoma or even invasive or semi-invasive fungal pneumonia.
Treatment:
Therapy is not usually required except where recurrent pneumonia due to obstruction may necessitate surgical resection.
Wide differential diagnosis.
Solitary pulmonary nodule >5mm:
· Granuloma including tuberculoma
· Infection (Pneumoococcal ‘round’ pneumonia, hydatid disease, fungal infection such as histoplasmoma)
· Benign (adenoma, hamartoma) or malignant (primary bronchial carcinoma or metastasis) tumours
· Vascular (Infarction, haematoma, AV malformation)
· Amyloidoma
Multiple pulmonary nodules:
· malignancy (metastases, lymphoma)
· immunological causes (Wegener’s, Rheumatoid nodules, Caplan’s syndrome)
· infection (fungal pneumonia, TB)
Multiple calcified pulmonary nodules:
· lung cancer engulfing a granuloma (eccentric calcification) or primary squamous cell lung cancer
· metastases (osteosarcoma, osteochondroma, mucinous adenocarcinoma of the colon/breast, papillary thyroid cancer)
· inhalational (silicosis, coal worker’s pneumoconiosis)
· infection (healed miliary TB, histoplasmosis)
Diffuse interstitial/alveolar septal amyloidosis
Epidemiology:
Primarily associated with primary systemic amyloidosis and uncommonly occuring as localized disease, it is the least common form of respiratory amyloidosis.
Clinical findings:
Diffuse interstitial amyloidosis presents with progressive dyspnoea.
Radiological findings:
Radiologically this disorder can look like interstitial fibrosis or pulmonary oedema
· CXR: reticular or reticulonodular pattern due to diffuse interstitial parenchymal involvement, +/- calcification or ossification of abnormal areas
· CXR: Less commonly it may present as a small nodular pattern mimicking sarcoidosis or miliary TB.
· HRCT: multiple pulmonary nodules 2mm-15mm in diameter, interlobular septal thickening and interlobular linear opacities which are more marked in the bases and periphery3
· HRCT: less common findings are areas of ground glass attenuation, consolidation, traction bronchiectasis, honeycombing, and foci of calcification within nodules 3,12.
Differential diagnosis for interlobular septal thickening and lines3:
· Lymphangitis carcinomatosis
· Idiopathic pulmonary fibrosis
· Pulmonary oedema
· Lymphoma
Fig 2a:
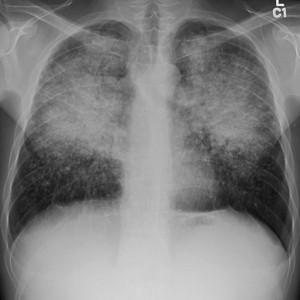
Fig.: Fig. 2 (a-f). 40-year-old man with known primary systemic amyloidosis with progressive stable dyspnoea on exertion. (a) Frontal CXR shows diffuse bilateral perihilar consolidation in a ‘batwing’ distribution suggestive of cardiogenic pulmonary oedema. However, the heart size is normal.
Fig 2b:
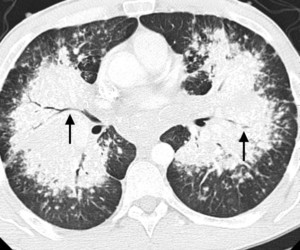
Fig.: Fig. 2 (a-f). 40-year-old man with known primary systemic amyloidosis with progressive stable dyspnoea on exertion. (b, c) Axial CT (lung windows, b and bone windows, c) confirm the presence of bilateral perihilar consolidation. There are air-bronchograms present (black arrows, b) in addition to speckled calcifications (short arrows, c) and ‘eggshell’-type calcified lymphadenopathy in the subcarinal region (arrowhead, c).
Fig 2c:
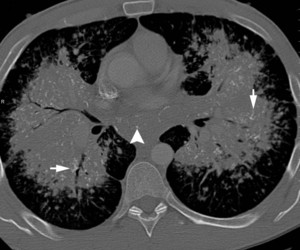
Fig.: Fig. 2 (a-f). 40-year-old man with known primary systemic amyloidosis with progressive stable dyspnoea on exertion. (b, c) Axial CT (lung windows, b and bone windows, c) confirm the presence of bilateral perihilar consolidation. There are air-bronchograms present (black arrows, b) in addition to speckled calcifications (short arrows, c) and ‘eggshell’-type calcified lymphadenopathy in the subcarinal region (arrowhead, c).
Fig 2d:
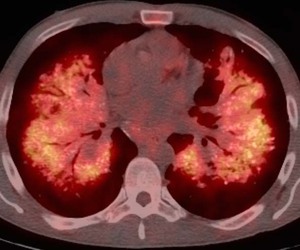
Fig.: Fig. 2 (a-f). 40-year-old man with known primary systemic amyloidosis with progressive stable dyspnoea on exertion. (d) Axial PET/CT scan shows intense FDG uptake throughout the parenchymal consolidation, which is not typical of pulmonary oedema.
Fig 2e:
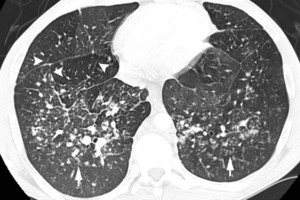
Fig.: Fig. 2 (a-f). 40-year-old man with known primary systemic amyloidosis with progressive stable dyspnoea on exertion. (e, f) Axial CT (lung windows) at the level of the lower lobes shows clusters of small pulmonary nodules (short arrows, e) in addition to thickening of interlobular septa (arrowheads, e and f) demarcating secondary pulmonary lobules (long arrow, f). By comparison with previous imaging studies, the patient was known to have pulmonary amyloidosis of the alveolar-septal type. This case is remarkable for the peculiar perihilar distribution of the alveolar consolidation, mimicking cardiogenic pulmonary oedema. However, the presence of calcifications and the intense FDG uptake is not consistent with simple pulmonary oedema, whereas these findings are characteristic of pulmonary amyloidosis.
Fig 2f:
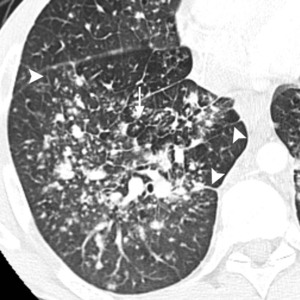
Fig.: Fig. 2 (a-f). 40-year-old man with known primary systemic amyloidosis with progressive stable dyspnoea on exertion. (e, f) Axial CT (lung windows) at the level of the lower lobes shows clusters of small pulmonary nodules (short arrows, e) in addition to thickening of interlobular septa (arrowheads, e and f) demarcating secondary pulmonary lobules (long arrow, f). By comparison with previous imaging studies, the patient was known to have pulmonary amyloidosis of the alveolar-septal type. This case is remarkable for the peculiar perihilar distribution of the alveolar consolidation, mimicking cardiogenic pulmonary oedema. However, the presence of calcifications and the intense FDG uptake is not consistent with simple pulmonary oedema, whereas these findings are characteristic of pulmonary amyloidosis.
Treatment:
Diffuse interstitial amyloidosis is a progressive condition associated with a poor prognosis. The median survival is 16 months from diagnosis of pulmonary involvement. It may be that diffuse interstitial amyloidosis is a marker of an otherwise fatal disease rather than being directly related to death5.
Laryngotracheobronchial amyloidosis
Epidemiology:
Laryngotracheobronchial (LTB) amyloidosis is associated with primary localized but not systemic amyloidosis.
Clinical findings:
The clinical course is highly variable. Patients may be assymptomatic or present with obstructive symptoms due to laryngeal/tracheal/bronchial stenosis such as dyspneoa, stridor, recurrent pneumonia/atelectasis or haemoptysis. Recurrent pneumonia can lead to respiratory failure. Amyloid can calcify and some authors have suggested a link between amyloid and tracheobronchopathia osteoplastica, a rare benign condition characterised by an abnormal presence of calcium deposits and bony tissue in the tracheobronchial mucosa but this has not been confirmed in a large group of patients 5,13,14.
Radiological findings:
LTB amyloidosis can take the form of intraluminal tumour-like masses (amyloidomas) or submucosal plaques and can be focal, multifocal or diffuse. Plaques may be calcified.
· CXR: rarely an intraluminal tracheal mass may be discernible, calcification may projected over the trachea/bronchi
· CT appearances: tracheal narrowing and wall thickening
· Can occur as isolated laryngeal thickening on CT, the appearance of which resembles laryngeal carcinoma
Fig 3a:
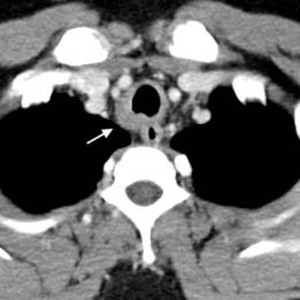
Fig.: Fig. 3 (a, b). 52-year-old man investigated for wheeze. Axial CT (soft tissue windows) shows marked nodular thickening of the tracheal wall (arrows). Subsequent biopsy confirmed tracheal amyloidosis. There was no evidence of amyloidosis elsewhere.
Fig 3b:
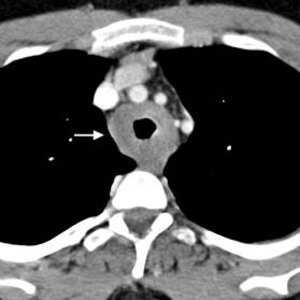
Fig.: Fig. 3 (a, b). 52-year-old man investigated for wheeze. Axial CT (soft tissue windows) shows marked nodular thickening of the tracheal wall (arrows). Subsequent biopsy confirmed tracheal amyloidosis. There was no evidence of amyloidosis elsewhere.
Treatment:
Bronchoscopic resection, laser ablation and external beam radiation have also been used to treat intra-luminal masses causing obstructive symptoms.
Hilar/mediastinal lymphadenopathy
Epidemiology:
Mediastinal lymphadenopathy may occur in systemic amyloidosis. Rarely, hilar/mediastinal lymphadenopathy may be associated with pulmonary parenchymal amyloidosis in primary systemic amyloidosis. Only a few cases have been reported of hilar/mediastinal lymphadenopathy in the absence of pulmonary parenchymal involvement (ie. with laryngotreacheobronchial amyloidosis or on its own as a manifestation of localized amyloidosis) and then, strictly speaking, it is a form of thoracic (primary localized amyloidosis) rather than pulmonary amyloidosis15.
Radiological findings:
· CXR, CT: Lymphadenopathy with homogeneous low attenuation and stippled or ‘eggshell’-type calcification is a characteristic finding of amyloidosis15.
Fig 4a:
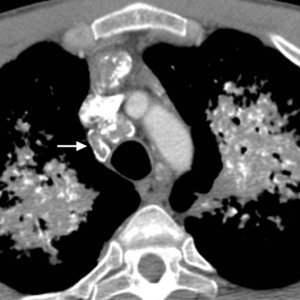
Fig.: Fig. 4 (a-d). Same patient as in Fig. 2 (40-year-old man with known primary systemic amyloidosis). (a-c) Axial CT (soft tissue windows) shows prominent mediastinal lymphadenopathy with ‘eggshell’-type and stippled internal calcifications (arrows).
Fig 4b:
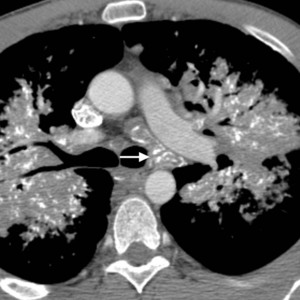
Fig.: Fig. 4 (a-d). Same patient as in Fig. 2 (40-year-old man with known primary systemic amyloidosis). (d) Axial PET/CT shows low-grade uptake up FDG by a non-calcified lymph node in the right axilla (arrowhead) but virtually no FDG uptake by the calcified mediastinal lymphadenopathy (arrow), highlighting the variable appearance of amyloidosis on PET/CT imaging.
Fig 4c:
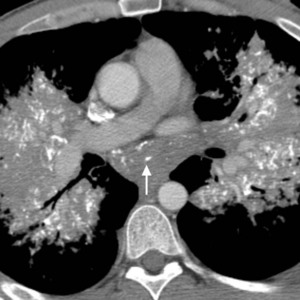
Fig.: Fig. 4 (a-d). Same patient as in Fig. 2 (40-year-old man with known primary systemic amyloidosis). (a-c) Axial CT (soft tissue windows) shows prominent mediastinal lymphadenopathy with ‘eggshell’-type and stippled internal calcifications (arrows).
Fig 4d:
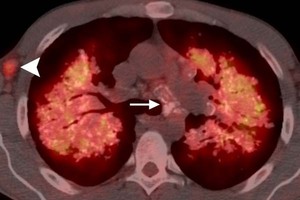
Fig.: Fig. 4 (a-d). Same patient as in Fig. 2 (40-year-old man with known primary systemic amyloidosis). (d) Axial PET/CT shows low-grade uptake up FDG by a non-calcified lymph node in the right axilla (arrowhead) but virtually no FDG uptake by the calcified mediastinal lymphadenopathy (arrow), highlighting the variable appearance of amyloidosis on PET/CT imaging.
Differential diagnosis for mediastinal lymphadenopathy:
· metastases
· malignant lymhomas
· sarcoidosis
· infections such as TB or fungal infections (histoplasmosis or coccidiomycosis)
· silicosis (look for egg-shell calcification)
Stippled/speckled calcification in lymph nodes:
· amyloidosis
· rare in lymphoma unless treated
· metastatic mucinous adenocarcinoma
· Castlemans disease



















