Neurovascular bundles are often located inside conical or tubular pathways in the human body, which are muscular, osseous, fibrous or mixed in nature. The adductor canal, firstly described by John Hunter in 1786, is a fibromuscular canal which contains the superficial femoral vessels and saphenous nerve along with a variable amount of fibrous tissue [1]. This aponeurotic tunnel is located in the middle third of the thigh and runs from the apex of the femoral triangle (Scarpa’s triangle) to an opening in adductor magnus through which the femoral vessels reach the popliteal fossa. It is conical or pyramid-shaped and triangular in section. Its boundaries are represented anterolaterally by vastus medialis, posteriorly by adductor longus and adductor magnus muscles and anteromedially by a strong aponeurosis that extends between the adductors across the vessels to vastus medialis (vastoadductor membrane). Sartorius is anterior [1, 2]. The adductor canal is a relevant anatomical region for many reasons, in particular the hiatal region may be a preferential site for the developing of atherosclerotic plaques and may play a role in two different (vascular and nervous) entrapment syndromes.
The compression of the saphenous nerve at the adductor hiatus is believed to be a possible cause of pain on the medial aspect of the knee [3]. The saphenous nerve neuralgia should be remembered as a differential diagnosis of pain referred to the knee region but this nervous entrapment syndrome is so infrequent that diagnosis is rarely made [3]. Symptoms may be successfully relieved by saphenous nerve block alone or combined with surgical exploration and decompression, especially in those cases with an evident compression at the hiatus [4].
The vascular entrapment syndrome related to arterial compression is much more frequent than saphenous nerve entrapment. It presents as a claudication syndrome during strong physical exercise, typically in young males. The hypertrophy of muscular structure of the adductor hiatus may play an important role in the pathophysiological mechanism [5]. Lower limb arteriography sometimes reveals a short, probably extrinsic, compression of the femoral artery in the distal thigh with few or absolutely no other signs of atherosclerotic lesions at other site. Frequently the arteriography doesn’t show any extrinsic compression, this suggesting a dynamic compression mechanism. Definite diagnosis is often made during surgical exploration when a fibrous band is encountered in the interior of the adductor canal or, most frequently, the musculoaponeurotic structures of adductor hiatus encroaches on the femoral artery. This claudication syndrome is relieved by section of the fibrous band and/or adductor hiatus, coupled or not with an autologous or synthetic femoral artery graft. Pathoanatomical findings reveal tunica intima proliferation and tears on the stenosed segment, which may be induced by long-standing, dynamic extrinsic compression [6, 7, 8, 9].
In addiction stenotic and occlusive arterial disease of the femoropopliteal segment is located in the distal part of the adductor canal in 70% of patients with intermittent claudication [10, 11].
Some theories suggest a local mechanical factor, while others suggest that local hemodynamic circumstances facilitate vascular disease in the region of the adductor canal hiatus [12].
According to the mechanical theory, intimal proliferation is caused by continuous microtraumas from the systolic hammering of the arterial wall against the adductor magnus tendon [12]. Others believe that intimal tears are caused by direct compression [6, 8] or traction [13] of the femoral artery in the hiatal region.
According to the hemodynamic theories, plaque formation occurs more frequently because of the "S" shaped curve of the femoral artery [14] or the high incidence of vascular branching in the hiatal region [10].
In literature the is only one paper about US anatomy of the adductor canal and it is a morphological study about US location of the adductor canal hiatus [15]. The US examinations were performed with an old generation 5-MHz mechanical-sector scanning head with a focal distance of 3.0 cm. More recently the introduction into clinical practice of sonographic equipment with high-resolution transducers, together with the development of new, increasingly sophisticated postprocessing algorithms to optimize image resolution, has led to an increased use of high-resolution ultrasound in assessment of the peripheral nervous system, including the saphenous nerve. In recent years large interest is arising in regional
anesthesia and pain medicine fields for the ultrasound-guided block of the saphenous nerve at different levels in the adductor canal [16-18]. This increasingly interest is related to continuing challenge of providing sensory anesthesia of the lower extremity without impairing disposition (quadriceps muscle weakness) of the ambulatory surgery patient. saphenous nerve block is essential for complete anesthesia of the foot and ankle because it represents the terminal branch of the posterior division of the femoral nerve and provides sensory innervation to the medial, anteromedial and posteromedial aspects of the lower extremity from the distal thigh to the medial malleolus.
The first of these peripheral anesthesiology works emphasized the importance of correct saphenous nerve detection in Hunter’s canal when performing a peripheral nerve block using an ultrasonographic in-plane approach and avoiding unintentional puncture of major vessels [16]. In this work the Author asserted that the saphenous nerve can often be imaged where it pierces the vastoadductor membrane, but this first work was lacking of a complete and detailed US iconography of the anatomical basis for this anesthesiologic procedure.
More recently in a descriptive prospective study about the feasibility and efficacy of real-time ultrasound-guided block of the saphenous nerve in the adductor canal, Manickam and coll. asserted that during the
examination of the lower third of the thigh using axial US scans the femoral artery was seen "diving" deep and progressively moving away from the anterior muscle plane (sartorius and vastus medialis muscle); this area was identified as the adductor hiatus and the peripheral block location was selected 2 to 3 cm proximally in the distal adductor canal [17]. It is important to note that with this procedure the sensitive block corresponded to the distribution of both the sartorial and infrapatellar branches of the saphenous nerve. Another, more selective, approach to saphenous nerve block is described by Horn and coll. using the saphenous branch of the descending genicular artery as a reference structure for US-guided peripheral nerve block out of the adductor canal [18].
In our anatomical study we pointed out the presence of some important anatomical landmarks to standardize the examination technique of the adductor canal.
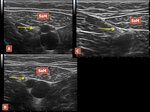
We divided the Hunter’s canal in three segments: the first part (proximal) is that segment of the canal where the posterior wall is formed by adductor longus muscle; the second part is bounded posteriorly by adductor magnus and the third part is the hiatal region. In the first part of the canal we observed the constant presence of a perforating venous vessel which connects the superficial femoral vein with the great saphenous vein. In all subjects of our examinations the saphenous nerve at the apex of the Scarpa’s triangle is lateral to the superficial femoral artery and becomes more anterior near the emergence of the communicating vein, which may represent an Hunterian perforator [19; 20]. In the distal part of the second segment the nerve is clearly appreciable between the anterior aspect of the arterial wall and the deep fascia of sartorius muscle, which is the more anterior boundary (Figure 11).
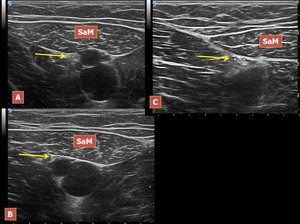
Fig.: Figure 11. A, B, C The saphenous nerve at three different levels in the Hunter’s canal. Legend: SaM, sartorius muscle; arrow, saphenous nerve.
The hiatal region was identified by the origin of the descending genicular artery. The descending genicular artery arises just proximal to the adductor opening and immediately supplies a saphenous branch. It then descends posterior to vastus medialis and anterior to the rounded tendon of adductor magnus, to reach the medial side of the knee, where it anastomoses with the medial superior genicular artery (Figures 12; 13).
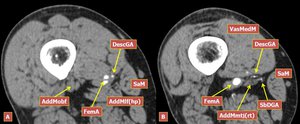
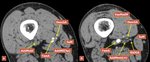
Fig.: Figure 12. A, B Two MDCT angiography axial slices of the thigh in a patient with severe atherosclerosis of the femoral artery demonstrating the origin of the descending genicular artery and its bifurcation which gives rise to the saphenous branch. Legend: SaM, sartorius muscle; FemA, femoral artery; DescGA, descending genicular artery; SbDGA, saphenous branch of the descending genicular artery; VasMedM, vastus medialis muscle; AddMlf(hp), adductor magnus longitudinal fibers (hamstring part); AddMmtj(rt), adductor magnus myotendinous junction (origin of the rounded tendon).
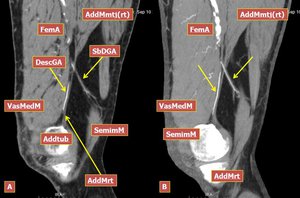
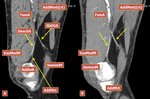
Fig.: Figure 13. A, B Two MDCT angiography reconstructed sagittal images of the same patient of Figure 12 showing the bifurcation of the descending genicular artery, its saphenous branch and the relations between the descending genicular artery and the rounded tendon of adductor magnus. Legend: SemimM, semimembranosus muscle; FemA, femoral artery; DescGA, descending genicular artery; SbDGA, saphenous branch of the descending genicular artery; VasMedM, Vastus medialis muscle; AddMmtj(rt), adductor magnus myotendinous junction (origin of the rounded tendon); AddTub, adductor tubercle; AddMrt, adductor magnus rounded tendon.
The descending genicular artery, below the origin of the saphenous branch, runs inferiorly along with the adductor magnus tendon to the medial region of the knee. Using the color-Doppler module the descending genicular artery may be identified anteriorly to the rounded tendon of adductor magnus as a pulsatile vascular structure. At this level the femoral vessels were seen moving deeply in a postero-lateral direction toward the popliteal fossa. The saphenous nerve separates from the femoral vessels and accompanies the descending genicular artery toward the roof of the adductor canal, which here is represented by the vastoadductor membrane, appearing on US axial scans as a double hyperechogenic line. This fascia is formed by tendinous fibers which spread laterally from the rounded tendon of the adductor magnus toward the vastus medialis and end in the medial intermuscular septum and it may be pierced by the saphenous nerve and the descending genicular artery [20]. However this aponeurotic structure is not dissociable in vivo on US images from the deep muscular fascia of sartorius and adductor magnus muscles and a more precise anatomical correlation should be performed by US examination of a dissecting room preparation. Unfortunately it was impossible to demonstrate the saphenous nerve, accompanied by the saphenous branch of the descending genicular artery, piercing the vastoadductor membrane, probably due to problems of correct insonation related to its oblique course. However, in all examinations, the small saphenous branch of the descending genicular artery was appreciable passing through the roof of the adductor canal applying the color-Doppler module.
We found that the adductor canal hiatus anatomical limits were best assessed by coronal oblique US scans. The most important reference landmark is the adductor magnus rounded tendon which forms the medial wall of the adductor opening and attacks to the adductor tubercle of the medial femoral condyle. The descending genicular artery was easily appreciated anterior to the rounded tendon using the color-Doppler module. The superior limit of the Hunter’s canal hiatus is represented by the oblique fibers of the adductor part of adductor magnus muscle, which insert on the medial margin of the linea aspera. The deflection site of the superficial femoral vessels were correctly assessed in all subjects of our examination by coronal oblique scans, using the adductor magnus myotendinous junction or the rounded tendon itself as a reference landmark.
In conclusion HRUS technique was able to depict Hunter’s canal boundaries and its neurovascular content with high anatomical detail in all subjects of our study. In addiction we found several anatomical structures in the adductor canal which may be regarded as US reference landmarks for a standard US assessment of this region.