HISTORY OF MAMMOPLASTY
Breast implants have been used for about four decades for both reconstructive and aesthetic purposes.
For the first time,
in 1854,
Velpeau described some techniques which modified and corrected altered shapes and sizes of the breast.
Later in 1889 Gersuny used local paraffin injections to increase breast volume,
and even though it could be noted a cosmetic amelioration,
there were numerous complications such as solid masses- granulomas,
cutaneous erosion and cutaneous fistulas,
migration,
blindness or pulmonary embolism.
In 1895,
Czerny trasplanted a patient’s lipoma to her to fill out a defect caused by removal of an adenoma.
E.Lexer in 1925 and Berson in 1944 proposed other autologous materials like dermis grafts and fat dermis graft,
from the gluteal region,
but the results were too unpredictable,
with atrophy noted to be up to 50%,
and giant calcifications and cysts of fat necrosis were frequently observed.[1-2]
The first injection of liquid silicon appeared in the fifties and it was performed for breast augmentation; in that period the silicone was considered as an inert substance with the advantage that its consistence approached that of normal breast tissue,
although important complications occurred,
such as hard,
painful nodules and/or inflammation.
[1,3-5]
In the same period plastic implants,
most in the form of a sponge made from polyethylene,
Teflon and silicone sponge were experimented for breast reconstruction and augmentation.
Pangmann is considered the first who seriously studied the use of sponges for breast augmentation; in fact he introduced a sponge-like implant made of polyvinyl alcohol (PVA) to promote implant integration into the surrounding breast tissue by growth into sponge pores.[1,5-6].
Soon some complications appeared such as incrustation and scar retraction reducing volume and softness of the implant; sometimes the implant contracted into a rock-hard formation,
seroma formation,
fistulization and infection.
The design of the implant was revolutioned by T.
Cronin and F.Gerow; they wrapped a smooth and thin envelope of rubbery silicone elastomer (polysiloxisanes) around a soft but firm silicone gel compound[1,7].
Compared to the already existing implants,
these new implants conserved better their form,
softness with less local tissue reaction.
This “natural feel” Silastic gel prosthesis became the implant of choice and the golden standard. In 1969 Cronin and Gerow made the envelope thinner and stronger and redesigned the shape to a tear-drop,
resembling a normal looking breast; in addition, Dacron patches were integrated on the posterior side of the implants in order to stabilize its position because of the integration of these patches to the pectoralis fascia.
But it was found that Dacron-backed prosthesis reacted unfavourably to any tendency to capsular contracture; so thin-walled discoid non-backed prostheses became the favoured shape and design[1].
Ashley described a new textured gel-filled implant with a polyurethane foam coating the silicone shell,
which resulted in decreased capsular contracture (2-15% sub glandularly,
compared to 20-59% when smooth implants are used)and remained soft more consistently than the smooth surface implants(fibroblast proliferation into the polyurethane in many directions)[1].
In the 80’s H.
Becker introduced a permanent inflatable implant to augment breast volume for aesthetic reasons; the advantages of these implants were a shorter scar and the possibility to adjust the volume preoperatively and postoperatively to obtain a perfect symmetry[1,8].
In 1990 the implant with an anatomical shape,
based on the teardrop designed first introduced by Cronin and Gerow,
was reintroduced,
because it was suppose to bring a better aesthetic result to the upper pole of the breast,
which is often too full when round implants are used [1].
So,
over the years,
three generations of silicone gel-filled breast implants have been manufactured,
based primarily on the type of silicone gel and type of elastomeric shell.
The first one was manufactured between approximately 1960 to the mid 1970s.
These silicone implants had a thick elastomeric shell with firm silicone gel.
The second generation was created between the mid 1970s and the late 1980s; they had a thin elastomeric shell,
with the silicone gel being less viscous.
The third generation of silicone gel-filled breast implants was manufactured between the late 1980s until the present and they have a multilayer shell with a barrier layer and thick silicone gel.
[9]
Saline filled implants have been introduced for the first time by Arion in 1965,
but since then they comprised only 10% of the implants placed; showed advantages consisting in the fact that saline is wholly harmless and if the silicone elastomer ruptures,
the saline solution is rapidly absorbed by the surrounding tissues. Other advantages were to be harmless and a very low capsular contracture rates,
especially when placed behind the pectoralis muscle (1-6%).
But on the other hand,
they showed a less natural appearance (folds,
consistence) and an incidence rate of deflation of 1-16%(1).
To date,
over 240 breast implant styles from American manufacturers alone [10].
The actual number of styles is far larger because many implants of a single style from a single manufacturer evolved through many variants over the years.
Also,
the "custom" implant type alone is diverse,
but single-lumen and double-lumen silicone implants are the most commonly encountered.
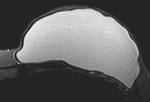
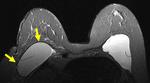
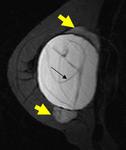
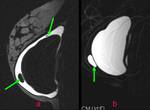
A single-lumen silicone implant (Fig.1) has an outer silicone Silastic shell containing homogeneous high-signal-intensity viscous silicone on T2-weighted images.
A double-lumen silicone implant typically has an inner lumen of high-signal-intensity silicone surrounded by a smaller outer lumen that contains saline and has different signal intensities,
depending on the pulse sequence.
A variety of other types of implants are occasionally placed,
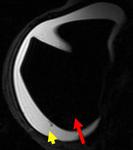
including reverse double lumen implants (saline in the inner lumen and silicone in the outer lumen). (Figg.2-3).
The implants are usually oval and have a smooth or textured surface.
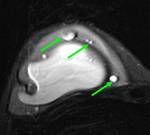
After surgical placement,
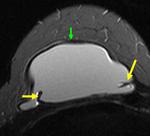
a thin fibrous capsule (scar tissue) normally forms around the prosthesis (Fig 4).
EPIDEMIOLOGY
Recently cosmetic breast implants have become more popular throughout the world.
Since their introduction in the1960s,
silicone breast implants have been placed in more than 2 million American women.
Of these implants,
80% have been placed for augmentation mammoplasty and remainder for breast reconstruction.
However,
there is an insufficient knowledge about the frequency and severity of local complications such as rupture, and capsular contracture has been the focus of many reviews.
[11-14].
BREAST RECONSTRUCTION
The techniques of breast reconstruction have evolved and matured over the past 25 years.
Methods of breast reconstruction include implant and autologous tissue reconstruction.
Breast reconstruction with use of implants is the simplest technique.
Breast implants may be surgically placed with two techniques placed anterior or posterior to the pectoralis major muscle.
In the first case the prostheses are located subglandularly (anterior to the pectoralis major muscle).
The advantages of this placement are:
•good control of breast shape;
•absence of breast shape variations during pectoralis major muscle contraction;
•good control of submammary fold position and its shape;
•post-operative course quicker than the sub-pectoral placement.
The disadvantages are:
-Higher risk of visibility or palpability of implant margins (in patients with thin skin or with big implants);
- higher visibility of capsular contracture,
if present;
- increase of capsular contracture incidence.
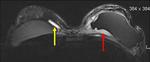
Another placement is subpectoral,
that is posterior to the pectoralis major muscle (Fig.5).
The advantages of this placement are:
-lower risk of visibility or palpability of implant margins;
-suitable for patients with thin skin;
-lower incidence of capsular contracture;
- lower visibility of capsular contracture,
if present.
The disadvantages are:
•Less control of breast shape;
•lateral displacement of implant and increasing of inframammary space;
•breast shape variations during pectoralis major muscle contraction;
•higher risk of upward displacement of the prosthesis;
•post-operative course longer.
Surgery-related Complications
Complications secondary to surgery include seroma,
infections,
hemorrhage,
flap necrosis,
lymphedema and axillary contracture[15-16]
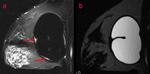
Seroma is a serous fluid collection in the axillary dead space or over the anterior chest wall; it is the most common complication of breast surgery,
with reported rates as high as 60% [17,18].
Infection is the second most common complication rating 3.6% [19],
characterized by either cellulitis or tissue suppuration.
Abscess secondary to a necrotizing infection is relatively minor,
and aggressive wound management resulted in healing of the abscess [20].
(Figg.
6-7).
Haematoma is a haematic fluid collection (Figg.
8-9).
Guidelines of breast implants
Post-operative care depends on the surgical procedure used and individual patient recovery and response.
Patients need to be monitored for a minimum of ten weeks following the surgical procedure.
From the literature we have three possible scenarios in the work-up of women with possible breast implant rupture.
In an asymptomatic woman the best estimate for breast implant rupture varies from 6.5 to 8%.
In this patient ultrasonography (US) is the best initial diagnostic tool.
If US shows no rupture,
the probability decreases 2.2%,
which obviates any work-up.
Instead if US shows rupture the probability a true rupture increases to a 37.8%,
which may not be high enough to recommend explanation.
But if MRI is positive for a rupture,
the probability of true rupture increases to 94–98% which is a high value to recommend implant removal.
The most recent U.S.
Food and Drug Administration guidelines recommend magnetic resonance imaging (MRI) examination 3 years after silicone implants are placed and every 2 years there after to evaluate for silent implant rupture.[21,22]
The Medicines and Healthcare Regulatory Authority recommends removal of these implants if there are problems,
such as implant rupture.
MR findings should be correlated with clinical history,
physical examination.
Silicone implant rupture has been linked to breast pain,
parasthesiaes,
asymmetric swelling,
breast contracture,
decrease in breast implant volume.
The incidence of silicone implant rupture is 15 to 33 percent in symptomatic women with pain or capsular contracture.
This incidence,
however,
is dependent on the generation of implant and the length of implantation.
[23-25]
Breast implant rupture
Complications related to breast implants include:
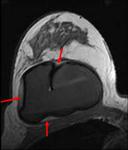
-fibrous or calcific capsular contracture (Figg.
10-11),
-rupture and leakage,
-localized pain,
-deformity,
-paresthesias.
In addition,
it has been reported that silicone-gel implants may be associated with the development of generalized autoimmune disorders [26].
MRI TECHNIQUE
High field strength magnets of 1.0±1.5 T are preferred in the case of silicone breast implants because they are able to use MRI sequences that can appropriately suppress or emphasize signal from water,
fat or silicone.
Moreover,
these types of magnets are better suited to detect implant ruptures.
Low field magnets can also be used to image silicone breast implants,
but the number of MRI sequences to suppress or emphasize the above-mentioned is limited; moreover a lack of adequate homogeneity prevents chemically selective fat suppression and may compromise image quality.
There is a linear relationship between magnetic field strength (B0) and signal-to-noise ratio (SNR).
With greater B0,
the SNR is higher,
and images with higher spatial resolution can be obtained in a relatively short acquisition time if appropriate pulse sequences are used.
The magnetic field should be homogeneous across the entire field of view,
which includes both breasts.
In the diagnosis of implant ruptures several types of sequences without contrast injection can be used.
Generally the silicone gel in the implant has a homogeneous low signal intensity on T1-weighted images and a high signal intensity on T2-weighted images.
It is possible to use either silicone-suppressed or silicone-selective sequences.
We can combine this with fat-suppression and produce a “silicone-only” image.
Mild compression applied to the breast in the lateral- to-medial direction decreases the amount of tissue to be imaged in that direction and thereby decreases the image acquisition time,
an effect that is particularly useful for imaging in the axial
and sagittal planes.
Compression also decreases patient motion during each sequence and between sequences and thus enables the avoidance of signal misregistration on subtracted images.
A variety of imaging protocols can be used to evaluate the breast.
MRI examinations in our department,
were performed with 1.5T clinical scanner with a dedicated bilateral breast surface coil.
Although there is no standard recommendation,
we advocate bilateral breast imaging for several reasons,
including the usefulness of assessing symmetry and evaluation of the contro-lateral breast in patients with newly diagnosed breast carcinoma.
The scanner was a high field whole body scanner doing 2D and 3D scans with fat saturation and silicone excitation properties.
The scanning protocol included:
•T2 TIRM axial (parameters: field of view was 30 or 10 cm; matrix size was 448x448; section thickness 4mm,
TR: 9570 TE 95)
•T1 FLASH 3d axial (parameters: field of view was 30 or 10 cm; matrix size was 448x448; section thickness 2mm TR 16 TE4,76)
•T2 TIRM coronal (parameters field of view was 40 or 10 cm; matrix size was 448x448; section thickness 4mm,
TR: 9570 TE 95 :)
•T2 TIRM sagital silicone-selective (parameters: field of view was 24 or 10 cm; matrix size was 256x256; section thickness 4mm,
TR: 6260 TE 89,
bandwidth,
13 kHz)
•T2 TIRM sagittal water selective (parameters: field of view was 24 or 10 cm; matrix size was 256x256; section thickness 4mm,
TR: 6260 TE 89,
bandwidth,
13 kHz)
Informed consent was obtained for all cases.
Breast implants: MRI role
MRI has two roles in patients with breast implants:
-detection of implant rupture,
especially in intracapsular rupture;
-in carcinoma detection in patients in which the prosthesis impairs sufficient evaluation of the breast tissue;
•MRI is the most sensitive and specific study to evaluate breast implant rupture (sensitivity 74–96% and specificity 94–98%) but because of its high costs,
is not suitable as a screening tool and should only be used in cases of US suspected rupture or after radical mastectomy.
In these cases,
MRI is the method of choice.[27,28]
•Mammography is ideal for breast parenchymal evaluation and obvious extracapsular silicone implant rupture.
•However it is of limited use in the evaluation of implants because a portion of the implant is never imaged and the internal structure is obscured by the radiopaque silicone,
so it fails,
however,
to consistently detect intracapsular implant rupture [29,30]
•US is valuable in the evaluation of implant integrity when extracapsular silicone or a collapsed shell floating within the silicone bed may be confidently identified
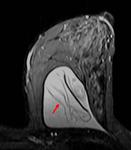
INTRACAPSULAR implant rupture is defined as rupture of the implant shell (elastomeric envelope) with silicone leakage that does not macroscopically extend beyond the fibrous capsule that commonly forms around silicone implants.
The most reliable magnetic resonance criteria is the presence of multiple curvilinear low-signal-intensity lines seen within the high –signal-intensity silicone gel,
the so-called “linguine sign” (Fig.12).
These curvilinear lines represent the collapsed implant shell floating within the silicone gel.
Without other magnetic resonance evidence of implant rupture such as the collapsed implant shell or free silicone within the breast parenchyma,
water droplets or small amounts of air within a silicone implant are not reliable signs of implant rupture.
An uncollapsed silicone implant rupture is defined as a tear of the silicone implant shell; the implant shell does not collapse or only partially collapses.
The linguine sign will not be present.
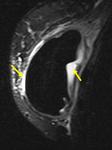
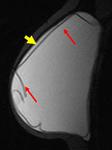
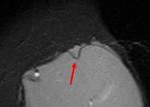
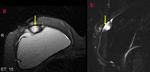
The magnetic resonance finding of uncollapsed rupture is the presence of free silicone outside the implant shell but still contained by the fibrous capsule. The sub-capsular line (Figg.13-14-15) is an early variant of linguine sign; hypodense lines run almost parallel to the fibrous capsule and just beneath this.
The beginning and the ending of each line can be followed to the surface of the implant.
A small amount of free silicone is on the outside of the silicone shell,
separating the shell and the fibrous capsule.
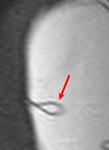
The orientation parallel to the fibrous capsule helps to differentiate sub-capsular lines from radial folds (Fig.2), which show a sharp angulation with the capsule.
This is a certain sign of rupture.[31;32]
As possible diagnostic signs suggesting an early intracapsular implant rupture there are the teardrop sign (Fig.16),
representing the invagination of the silicone membrane containing a droplet of silicone,
and the noose sign or key-hole sign (Fig.17),
that is a local invagination of the silicone membrane,
where the two membranes do not touch; a small amount of free silicone is seen on the outside.[31]
Unlike rupture,
gel bleed is microscopic silicone leakage through an intact implant shell.[ 34,40-42]
Extracapsular silicone implant rupture (Fig.18),
the less common type of implant rupture,
is defined as rupture of both the implant shell and the fibrous capsule with macroscopic silicone leakage that extends beyond the fibrous capsule into surrounding tissues.
Focal areas of high signal intensity,
representing free silicone,
can be identified on MRI.
In addition to free silicone in the surrounding breast parenchyma,
the linguine sign is often present with extra-capsular ruptures.[33-39]
Occasionally,
round foci with water/serum can be seen mixing with the silicone gel within the implant.
They can be seen if steroid or antibiotics were installed into the lumen preoperatively,
or if there is an influx of serum/water into the implant through a defect membrane.
Reynolds et al reasoned that the silicone envelope acts as a semi-permeable barrier and that fluid from the surrounding tissue enters the implant,
causing the droplet.
They appear as small hypointense elements in the silicone.
The presence of many droplets is accepted as being indicative for implant rupture and,
in this case,
they are usually seen in combination with the collapsed envelope.
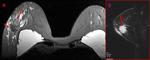
In double-lumen implants rupture,
the “salad-oil sign” appears (Figg.
19-20). It could be an additional sign of rupture,
but it could be seen in implants injected with povidone-iodine,
saline or steroids.[32].The train rail sign appears as two hypointense parallel lines in close proximity forming a double-contoured sub-capsular line within the silicone gel.
It represents both membranes in a double lumen implant that have ruptured and it is a certain sign of rupture.[31].