Keywords:
Urinary Tract / Bladder, MR, Diagnostic procedure, Contrast agent-intravenous
Authors:
S. Donaldson1, B. Carrington1, R. Cowan1, J. Lyons1, D. Buckley2, S. Bonington1; 1Manchester/UK, 2Leeds/UK
DOI:
10.1594/ecr2011/C-2308
Purpose
Background
- Bladder cancer is the fourth most common cancer in men and twelfth most common cancer in women in the UK [1].
- The five-year survival rate for muscle-invasive bladder cancer is < 50% [2].
- Increasingly,
treatments are being used which include:
- Concurrent chemo-radiotherapy with bladder sparing.
- Neo-adjuvant chemotherapy + surgery or radiotherapy.
The clinical problem
- Treatment of bladder cancer with neo-adjuvant chemotherapy affects normal tissues as well as tumours.
- This results in normal tissue toxicity.
- Some patients experience adverse bowel and bladder effects either acutely or in the long-term.
- Identifying patients likely to develop these effects is difficult.
- Identification of these patients would allow treatment modification and / or closer monitoring of those likely to develop treatment effects.
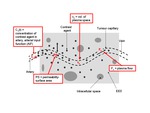
What is DCE-MRI?
Dynamic contrast-enhanced (DCE-) MRI involves an intravenous injection of paramagnetic contrast agent.
This enters tumour due to the increased leakiness of tumour blood vessels.
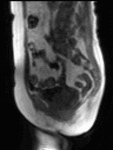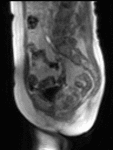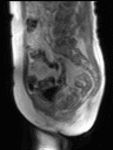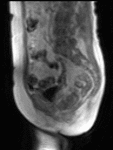
It shortens the T1 value of tumour resulting in signal enhancement on a T1-weighted MR image (Fig.
1).
This signal change can be monitored over time with rapid MR sequences.
Analysis of the DCE-MRI signal-time course can be carried out.
Tracer kinetic analysis of DCE-MRI data
DCE-MRI data can be analysed with a tracer kinetic model,
e.g.
two-compartment exchange model (2CXM [6]) to obtain estimates of microvascular parameters (Fig.
2).
Aims
To investigate whether perfusion measured with DCE-MRI in normal tissues before and after neo-adjuvant chemotherapy in patients treated for bladder cancer:
a) Are significantly different;
b) Can predict those patients who go on to develop normal tissue effects.