If any of the links to figures or videos don´t work,
please view them by clicking on the sidebar image
SECTION 1: BASIC CONCEPTS
The understanding of the basic bubble acoustic properties and their interaction with the sound waves is necessary to explain some of the issues (Fig. 2,
Fig. 3 )
1.1 Microbubbles,
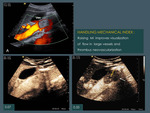
acoustic power and mechanical index.
Microbubbles oscillate in a sound field in a non-linear way.
Listening to the harmonic signal associated to non-linear oscillation,
and suppressing the linear tissue signal permits seeing exclusively the signal due to the presence of bubbles,
thus achieving contrast- specific imaging.
The response of the microbubbles to the sound field mainly depends on the acoustic power of the incident wave [6],
which is related to the sound wave amplitude (Fig. 2 ) and roughly to the mechanical index (MI).
MI and power are displayed on the screen and may be varied by means of button handling.
Microbubbles oscillate at low acoustic power (low MI),
but they are frail and disrupt when the acoustic power is high (high MI) (Fig. 3 ).
Increasing progressively MI will disrupt more and more bubbles.
In the different machines,
acoustic power is expressed as a percentage of the total power.
In most cases the grey-scale conventional explorations are performed with 100% of the acoustic power.
This would break all the bubbles very quickly,
so CEUS explorations are performed with a very low power of insonation (0.20-1%),
and low MI.
The values of the MI are expressed differently in the different machines,
and cannot be extrapolated.
Each user must learn the MI values from the technician of applications of his/her machine.
1.2 Microbubbles resonance frequency in an acoustic field is 2-3 Mhz,
which happens to be very similar to the frequencies employed for US diagnostic purposes with a convex 3.5-4 Mhz probe.
Thus,
it can be said that microbubbles are in tune with the frequencies of convex abdominal transducers.
In high-frequency imaging,
such as in the linear probes,
the driving frequency is away from the resonance frequency of the bubbles and only a small proportion of the bubble population will undergo nonlinear oscillation,
so the image signal is less intense.
1.3 Microbubbles and time. The extent to which bubble destruction occurs also depends upon the duration of exposure.
The longer the exposure,
the higher the destruction.
1.4 The higher the dose,
the higher the number of microbubbles and the higher the image signal …up to a limit. There is a concentration threshold over which the imaging signal does not increase linearly,
with a major draw-back: Attenuation increases and obscures deep structures.
SECTION 2: HANDLING VARIABLES.
TIPS AND TRICKS
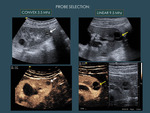
2.1HANDLING FREQUENCY AND PROBES
The choice of frequency and probe depends on the lesion or organ to be studied considering the following variables:
- Depth of the lesion or organ
- Size of the lesion or organ
- Multiple or single lesion
2.1.1 A convex 4Mhz multifrequency-probe is the most widely used probe to study abdominal organs.
With this probe,
the emitted wave will achieve the maximum bubble resonance,
and thus,
the maximum signal intensity.
Also,
the large field of view permits the exploration of whole organs,
and the study of large or multiple lesions.
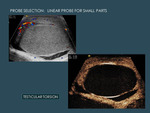
2.1.2 A 9 MHz linear probe is recommended for small parts (breast,
testicle,
parotid glands) (Fig. 4 ) or superficial small lesions (lymph nodes,
soft tissues).
Intra-abdominal or thoracic small superficial lesions may benefit from the linear probe too,
due to the higher resolution related to the higher frequency (Fig. 5 ).
Using higher frequency probes is a trade-off between resolution and signal intensity: the higher resolution is convenient for improving visibility of superficial small lesions,
but the bubble signal will be lower,
due to the suboptimal bubble resonance.
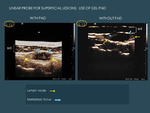
This can be partially compensated by increasing the administered dose (see section 2.3).In the authors´ experience,
when the structure is very superficial the interposition of a gel pad improves the visualization of the bubbles (Fig. 6 ).
In the case of superficial abdominal or thoracic lesion,
the authors recommend using a trapezoidal window to widen the field of view.
2.2.
HANDLING DOSAGE
The intravenous recommended dose of sulfur hexafluoride is 2.4 ml if the convex probe is used,
administered as a bolus,
flushed with 5-10 ml of saline [1].
A 1.2 ml dose has been proven to be enough for renal enhancement and for pediatric intravenous use. When a high-frequency probe is used,
the recommended dose is 4.8.
This is because as was explained above (section 1.2),
bubbles do not oscillate optimally under higher frequencies; a double dose will double the number of bubbles and this will improve the bubble signal.
OTHER DOSE ADMINISTRATION
A second or a third dose can be used:
2.2.1 To evaluate another organ or other lesions. For instance: to evaluate the contralateral kidney,
or to evaluate other intestinal segment.
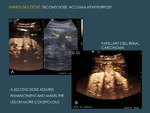
2.2.2 To evaluate again the lesion already studied.
- To reevaluate the temporal and spatial enhancement of a lesion with characterization purposes.
In this case it is desirable to break all of the bubbles before the reevaluation to monitor replenishment,
so a high mechanical index flash is recommended to break them.
This can be achieved by a specific high MI flash present in the different machines for this purpose,
or by raising manually the acoustic power or the MI,
which will break the bubbles in the whole image sector.
Another way to do it is by inducing a color Doppler flash.
At least five minutes must de waited before repeating the exploration for characterizing focal liver lesions.
- To evaluate if there is enhancement or to delineate or study better the enhanced parts in lesions with very subtle enhancement or in which it is not clear if there is enhancement or if there is no enhancement at all. Some examples are: in complex cystic lesions in which the septal enhancement or a small mural nodule may be difficult to see; in poorly vascularized lesions to demonstrate bubble circulation (to distinguish hypovascular metastasis from necrotic abscess) or to better delineate or characterize the lesion (papillary renal carcinoma) (Fig. 7 ).
In this situation,
the purpose is accumulating persistent bubbles of the previous dose with the bubbles of the new dose,
so no burst flash is to be employed.
2.3.HANDLING MECHANICAL INDEX
Many explorations may be performed satisfactorily with the basal default value MI established in the presets of the machines at installation .
However,
MI must be considered as a dynamic parameter ( www.ceusinfo.com) that must be tailored to each situation because raising /and lowering the MI may improve the visualization of the lesions.
This is especially so in superficial or deep lesions.
But other benefits may be attained.
Handling of MI can be useful in the following situations:
www.ceusinfo.com) that must be tailored to each situation because raising /and lowering the MI may improve the visualization of the lesions.
This is especially so in superficial or deep lesions.
But other benefits may be attained.
Handling of MI can be useful in the following situations:
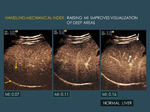
2.3.1 To evaluate deep lesions or areas.
The higher the MI,
the deeper the penetration of the beam,
so in deep lesions it is useful to raise manually progressively the MI until the lesion or area are clearly seen (Fig. 8 ).
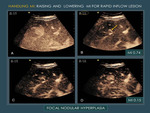
2.3.2 To reevaluate the vascular distribution and enhancement direction during the arterial phase (centripetal or centrifugal) in rapid fill-in focal lesions in which there is a very short time to evaluate the fill-in.
A slight gradual MI rise will break the bubbles in the parenchymal slow-flow microvasculature,
but rapid bubble in-flow in bigger vessels will permit to depict them,
obtaining a vascular map similar to angiography.
Posteriorly,
MI may be lowered to evaluate the lesion fill-in pattern (Fig. 9 ) (video 1 CLICK ON SIDEBAR Fig. 10 ).
In fact,
some authors havecalled this maneuver “pseudoangiography” ( www.ceusinfo.com ).
www.ceusinfo.com ).
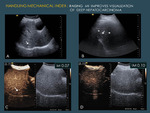
2.3.3 To improve visualization of hepatocarcinoma. Some authors recommend starting with a slightly higher MI to make more conspicuous the hypervascular arterial in-flow.
In our experience this is especially useful in small and deep hepatocarcinomas (Fig. 11 ).
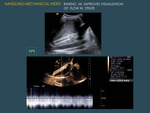
2.3.4 To evaluate medium or large size vessels.
MI may be dynamically raised and adjusted to optimize the visualization only of the vessels.
This is especially useful in the evaluation of vessels in liver and renal transplantation,
and in the evaluation of stents or TIPS,
in which the stent walls may prevent an optimal Doppler exploration and doubts concerning permeability or a partial thrombosis may appear (Fig. 12 ,Fig. 13 ).
2.3.5 To minimize bubble rupture in hemangiomas, it is recommended to LOWER the MI.
In this way more bubbles will survive and there will be more time to demonstrate the slow centripetal fill-in of classical hemangiomas.
2.4.
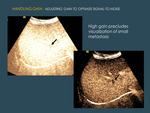
HANDLING GAIN
Gain is a post-process that does not influence the enhancement of lesions,
but it may influence the quality of the images because setting gain too high causes image noise,
and this may influence the radiologist subjective perception (Fig. 14 ).
The gain must be adjusted so as to reduce noise,
without losing the bubble signal.
The pre-contrast gain level should be set immediately below the level where noise is just barely seen.
Gain can be real-time adjusted,
except when quantification is going to be performed; in this case,
gain must not be changed during the clips to be used for quantification purposes.
2.5.HANDLING FOCAL ZONE
The electronic focalization of the ultrasound beam is related to increased bubble rupture in the focalized area,
so the focal zone must be kept deep to the area of interest to minimize bubble rupture and to ensure a more uniform acoustic field.
2.6.MAP SELECTION
All the machines offer different post-process maps with different colors and hues and different contrast degrees.
Each user must find in his/her machine the map or maps in which lesions seem to be more conspicuous or the map with which the user feels more comfortable to work with.
Once the ideal map or few maps are chosen,
the authors recommend using always the same one.
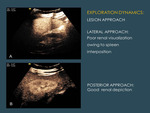
2.7.EXPLORATION DYNAMICS
The exploration dynamics must be adapted to the purpose of the study:
2.7.1 Focal lesion characterization. After having chosen the ideal scanning plane (for example in-plane with an afferent vessel of the lesion,
or the plane in which septa are more prominent),
initial arterial phase in-plane evaluation is recommended.
Then,
fanning of the lesion in different scanning planes will permit to study the whole lesion comparing its enhancement with the adjacent parenchyma and reducing bubble rupture ( VIDEO 2 CLICK ON SIDEBAR Fig.
15 ).
In the case of hemangiomas,
interrupted exploration is recommended so as to minimize bubble rupture and to avoid false washout that may create a diagnostic problem.
This can be achieved by staying in plane and freezing the image (recommended in the case of small or not conspicuous lesions to avoid losing the lesion) or exploring other areas to return at intervals to study the lesion.Continuous scanning is not needed in most lesions,
and interval exploration may be used during portal and late phases so as to preserve the bubbles
2.7.2 Focal lesion quantification. The plane must be maintained during the clip used for quantification,
so the probe must be kept still.
Otherwise when quantification is performed,
part of the region of interest (ROI) may fall out of the lesion.
In this case (which may also happen if the breathing movements are big or if the patient moves),
surrounding tissue or structures enhancement will contribute to the time-intensity curves (TICs),
thus yielding miss-information.
The degree of error will depend on the amount and the duration of extra-lesional tissue moving in and out of the ROI.
In some cases,
cutting the clip and eliminating the part in which the out-off-plane information appears will solve the problem.
Considering quantification,
standardization of the procedures is of outmost importance.[7]
2.7.3 Focal lesion detection. The organ must be exhaustively studied in a systematic way.
Some authors recommend standardized exploration systematics (with a standardized sonoexam for the detection of metastasis) which improve the interpretation of the studies by different observers and are very useful for comparison purposes.
2.7.4 Vascular evaluation. When small or medium size vessels are the purpose of the study (for instance arteries in hepatic and renal transplantation) care must be taken to choose the scanning plane with the aid of color Doppler,
and to remain still,
because the information obtained during the first seconds of enhancement is important and must not be lost because of being off-plane.
Other dynamic exploration factors:
2.7.5 Dual imaging should be used whenever it is very important to remain in plane and the basal grey image is needed to keep the references (small focal lesion,
small vessel,
small nodule or septum in a complex cystic mass) at least until the lesion,
vessel or septum is conspicuous on the CEUS images.
The drawback of dual imaging is that the image quality of the grey-scale image is downgraded because the image is obtained using a very low acoustic power (Fig. 11) .
Also,
the bubbles are actually seen to some extent in grey-scale imaging,
sometimes making the vessels or the lesion less conspicuous. Because of this reasons,
the lesion is sometimes “lost” on the grey- scale image,
rendering dual imaging useless.
2.7.6 The scanning position to scan the lesion must be optimized also.
Very deep lesions will be difficult to see,
even though the MI is raised,
so it is advised to choose another approach that brings the lesion closer to the probe.
In the liver,
oftentimes this is achieved with an intercostal approach,
which has the drawback of the rib shadows which may obscure partially the lesion during breathing.
In the case of the left kidney,
if parenchymal phase is important for clinical reasons (for example,
to see focal pyelonephritis foci),
it is recommended not to choose an approach in which the spleen lays between the probe and the kidney because the high bubble concentration of the spleen parenchyma will produce strong attenuation that will render kidney evaluation difficult (Fig. 16 )
2. 8 HANDLING DATA AND INFORMATION.
Evaluation of enhancement during all phases is very important,
especially when characterization is the purpose of the exploration.
Saving clips of the arterial phase (or cortical phase in the kidney) is mandatory,
we recommend at least 40s clips.
From this moment on,
the authors recommend shorter representative clips of the rest of the exploration to be saved.
There is no need to store the whole exploration,
as this will overload the PACS.
Workstation review of the exam is strongly recommended,
since it very often aids to the diagnosis or shows small overlooked lesions.
It also permits a slow-motion view that can sometimes be of great value to clarify details.
Saving representative still frame images of all phases is still recommended,
just in case the video saving fails.
For quantification purposes,
linear data are preferred for analysis by the software,
but most clinical users do not have access to them [7] and linearized log-compressed data are used instead.
This may modify information to some extent,
and cause variability in the results.
If our machine does not offer the possibility of linear data based TICs,
this cannot be solved; but standardization of the data acquisition,
of the exploration settings and of the exploration protocols will reduce the variations to certain extent.
SECTION 3.
ARTEFACTS,
PROBLEMS AND LIMITATIONS
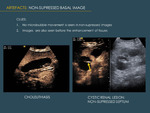
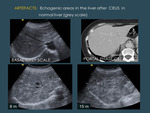
3.1 NON-SUPRESSION OF STRUCTURES WITH HIGH ACOUSTIC IMPEDANCE
Interfaces with very high acoustic impedance or else specular interfaces (for example the diaphragm) are not effectively suppressed by image suppression techniques .
This may happen in biliary stones,
in lesions with a fatty component or calcifications,
or in some septa in cystic lesions (Fig. 17).
The problem is that non-suppression can preclude or make difficult the evaluation of enhancement and washout.
Also,
it may be confused for enhancement.
Non-suppressed echoes are visible from the beginning of the CEUS exploration,
from time zero before the contrast arrives,
they don´t change with time and no bubble movement is detected.These features help to distinguish it from real enhancement.
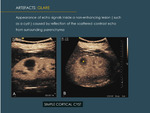
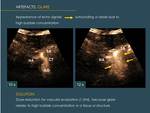
3.2 GLARE
This problem refers to the appearance of hazy echoes inside a non-enhancing lesion (which may simulate enhancement in the lesion Fig. 18 ); or surrounding a vessel (which may obscure the vessel limits and surrounding structures Fig. 19 ).
This artefact is related to high contrast concentration.
It most probably occurs due to reflection of the intense scattered contrast echoes from the surrounding parenchyma in the lesions (www.ceusinfo.com); or from the intensely enhanced vessels in the surrounding tissues.
This effect increases if the lesion is deep,
and is especially intense if the lesion is close to the reflecting lung surface or the intensely enhancing kidney.
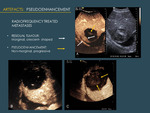
3.3 PSEUDOENHANCEMENT
Pseudoenhancement refers to the appearance of echoes inside a non-enhancing lesion,
which may be confused for enhancement.
Most commonly it happens in radiofrequency ablated lesions,
particularly in heterogeneous or echogenic deep lesions.
But it may also occur in other lesions with heterogeneous content,
such as type II hydatid-cysts.
Pseudoenhancement is not seen from the beginning.
Rather,
it follows enhancement of the parenchyma between the probe and the lesion; it begins relatively late (portal phase) and progresses over time,
typically in a non-marginal location.
No wash-out is seen. This temporal and location pattern distinguishes it from real tumor-recurrence enhancement in radiofrequency ablated lesions,
which shows early enhancement at the margin of the ablation zone (Fig. 20).
Some authors [8] explain that pseudoenhancement is probably due to nonlinear ultrasound propagation through intervening microbubble-perfused tissue,
which increases with bubble concentration.
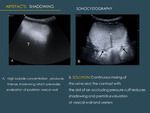
3.4 SHADOWING
The highly enhanced parenchyma can produce attenuation of the sound beam and preclude correct visualization of deep tissues or structures.
This problem is dose related and occurs when high contrast dose is administered,
as was explained previously,
and this is one of the reasons that may influence the dosage.
But even when the dosage is correct,
this problem occurs almost always to some extent.
Raising the MI will disrupt some of the superficial bubbles and will make the beam penetrate more and see better the deep areas. Shadowing also occurs when the left kidney is examined during late phase with the interposed spleen,
because the spleen persists very highly enhanced until very late and produces attenuation over the kidney (Fig.
18) .
This problem happens also during sonocystography.
In this case the bubbles are not in the vascular bed,
but inside a cavity,
so they tend to float over the urine,
on the top of the bladder,
and produce dense shadowing.
The continuous movement of the bladder content can be achieved with the aid of a dynamic pressure-cuff that moves and pushes into the bladder the mixed saline and contrast through a vesical catheter (Fig. 21 ).
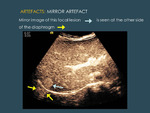
3.5 MIRROR ARTEFACTS may happen during CEUS, in the same locations as in grey-scale US (Fig. 22) .
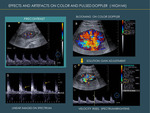
3.6 ARTEFACTS IN HIGH MI EXAMINATIONS
Microbubbles enhance Doppler signal and can produce the following artefacts in high MI examinations (Fig. 23) :
- Blooming refers to color saturation with appearance of color in pixels outside the vessel limits.
This may be minimized lowering the general gain.
- Rise of the spectral Doppler velocities.
The presence of the microbubbles can artificially rise the velocities 30%.
- Brightening and broadening of the spectrum
- Appearance of linear artefacts in the Doppler spectrum
Microbubbles are barely seen in basal grey-scale high MI examinations.
Very rarely,
the authors have seen after a normal CEUS examination with normally enhanced liver a post- contrast grey-scale patchy liver ( Fig. 24) .
Interestingly,
the effect can be long lasting (up to 20 minutes).
We don´t have an explanation for this finding.
3.7.PROBLEMS RELATED TO CONTRAST ADMINISTRATION
Preparation and handling of the contrast is easy if the instructions are followed.
Care must be taken to re-shake the contrast before its administration if some circumstance delays its administration.
The use of smaller than 20g diameter needles due to very frail peripheral veins or the use of long or complex connection systems may be related to some bubble destruction.
3.8 LIMITATIONS
Some of the limitations of CEUS are inherent in medical ultrasound imaging,
such as a big body habitus,
a poor acoustic window or fatty liver.
Others are specific to CEUS including,
among others,
the limited survival of the microbubbles,
which implies a temporal limit; the attenuation related to the presence of bubbles which limits the visualization of deep lesions; the difficulty in evaluating multiple lesions during all of the phases,
and the hemodynamical variations among patients.
A wise examiner is the one who is familiar with the limitations of the technique and who masters the handling of the different variables to obtain the best performance.
A major shortcoming with CEUS examinations is their operator-dependent nature as compared with computed tomography (CT) and magnetic resonance (MR) imaging.
Standardizing the examination dynamics,
the clips or images saved,
the software used for quantification,
and reviewing difficult explorations in consensus can reduce the operator-dependent limitations of CEUS [9].
In the future,
computed-aided diagnosis may help to overcome this problem to some extent.
Handling of the different variables is a trade-off too,
because it may further add variability to the explorations. Another problem is that clinicians feel more comfortable and understand better the more anatomic CT and MR images than the CEUS images.