Pancreatic imaging: guidelines: MDCT or MRI?
- if pancreatitis: MDCT classically first line
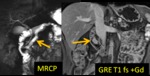
- if suspiction of biliary pancreatitis and possible stone in the CBD: MRI
- if suspiction of a tumour: MRI ≥ MDCT
- anatomic variants type pancreas divisum: MRI ≥ MDCT
- ductal anomalies: MRI ≥ MDCT
- MDCT + I MRI + Gd
- Pro’s of MDCT: shows calcifications,
generally less prone to technical and interpretive errors,
faster,
more available,
more practical for acutely ill patients
- US,
EUS,
ERCP and PET/CT: no first line exam’s
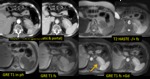
MDCT protocol for known/suspected panceatic tumour
- water as oral contrast
- nonenhanced images (liver – pancreas)
- pancreatic phase (+/- 125ml C at 4ml/sec,
35-45sec delay,
1-2mm for angio and 3D ductal anatomy and 5mm)
- hepatic parenchymal phase (+/- 70sec delay)
MR protocol for known/suspected panceatic tumour
- water as oral contrast Fig. 1
- T2 +/- f.s.
,
axial and coronal
- MRCP
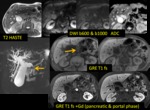
- DWI (b 50-600-1000)
- GRE T1 in/op phase ,
axial
- 2D or 3D fat-suppressed GRE; +/- bolus dynamic Gd-enhancement (pancreatic phase and hepatic parenchymal phase)
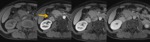
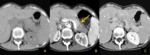
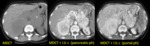
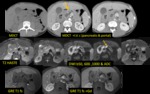
Ductal adenocarcinoma: CT Findings Fig. 2
- focal mass (95%)
< low density area (75%)
< ill-defined borders
< hypo-enhancing mass
– duct dilation
< pancreatic duct (50%)
< CBD (40%)
– focal narrowing
< “missing duct sign” at the level of the tumour
– atrophy of the tail (20%)
– dilated collateral veins (12%)
– duodenal invasion
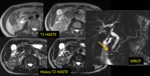
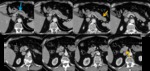
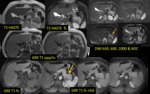
Ductal adenocarcinoma: MR Findings Fig. 3 Fig. 4
T1: usually hypointense,
isointense if surrounding
pancreatic tissue is abnormal (chronic pancreatitis)
T2: iso- or slightly hyperintense
- hypo-enhancing after I.V.
Gd (rapid bolus)
- DWI: hyperintense on high b-values & low ADC
- double duct sign: in pancreatic head cancer
- stenosis or apparent occlusion
- usually irregular with abrupt termination
- lenght should correspond to size of mass
Special types of pancreatitis mimicking adenocarcarcinoma:
- Groove pancreatitis or paraduodenal pancreatitis
- Autoimmune pancreatitis
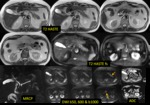
Groove pancreatitis
- special type of chronic pancreatitis near the minor papilla
- 3 types:
- pure (head of the pancreas is spared)
- segmental (pancreatic head and the ducts are affected)
- non segmental (sec.
to established chron.
pancreatitis)
- hallmark: scar tissue with fibrosis in the pancreaticoduodenal groove
- duodenum: involved by a chronic inflammatory process (cystic dystrophy)
imaging findings important to differentiate from carcinoma:
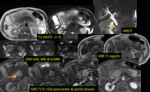
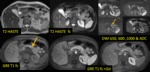
- sheet-like mass in the groove (most important diagnostic clue !) Fig. 5 Fig. 6
– hypodense on CT
– MRI: T1: hypo
T2 & DWI: hypo-iso-hyper,
depending on the stage
– Gd: homogeneous delayed enhancement < to the fibrous nature
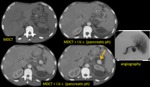
- inflammatory changes in the pancreatic parenchyma
– CT: hypodense pancreatic head
– MRI: hypo T1 ,
hyper T2,
DWI hyper
– enlargement
- regular common bile duct tapering Fig. 7
- normal pancreatic duct or duct penetrating sign Fig. 8
- PET-scan may be positive in an acute stage,
mimicking a carcinoma Fig. 9
Cystic dystrophy of the duodenum Fig. 10
– dilated stomach
– thickened duodenal wall
– deep location of the cysts
– narrowing of the duodenal lumen
– signs of groove pancreatitis
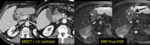
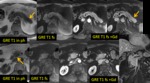
Autoimmune pancreatitis
- IgG4 related pathology,
can be multifocal
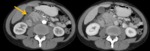
CT findings: Fig. 11 Fig. 12
– edema with loss of the classical lobulation pattern
– no pseudocysts nor atrophy or major ductal dilatation
– no calcifications
– unenhanced CT reveals a slightly hypodense homogenous mass,
hypo-attenuating in the pancreatic contrast phase (most important diagnostic clue to differentiate from neuro-endocrine tumour!),
and becoming homogeneously hyperdense at the delayed phase (most important diagnostic clue to differentiate from adenocarcinoma!)
– dilatation of the common bile duct when pancreatic head is involved
MR findings: Fig. 13 Fig. 14
- diffuse enlargment (sausage-like) or focally swollen pancreas with loss of the classical lobulation pattern
- homogeneously hypointense on T1-weighted images even more pronounced with fat suppression implementation
- homogenous and slightly hyperintense on T2-weighted images and clearly hyperintense with fat suppression
- DWI: hyperintense & low ADC
- no peripancreatic fluid effusions nor pseudocysts
- no pancreatic atrophy
- moderate ductal compression and only minor upstream dilatation
- clear-cut demarcation of the lesion with the normal pancreatic parenchyma
PET-scan may be positive in an acute stage,
mimicking a carcinoma
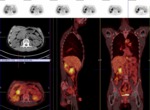
NET: Neuro Endocrine Tumours
- related with Multiple Endocrine Neoplasia (MEN)
- pancreatic islet cell tumor MDCT findings: Fig. 15
– lesions:
- often < 2cm,
and multiple in MEN1 (sens 70-80%)
- if ≥ 2cm,
calcifications: often malignant
- periampullary lesions: water as oral contrast necessary for optimal distension of the duodenum
– usually hypervascular (look also for liver mets)
– solid or ring-enhancement if cystic (delayed scans)
– only in advanced stages ductal involvement
– large tumours may show cystic & necrotic areas
- pancreatic islet cell tumor MRI findings: Fig. 16
– lesions:
- often < 2cm,
and multiple (sensitivity MR > CT)
- ≥ 2cm: often malignant
- SI:
- T1: hypo
- T2: hyper (unless high collagen content)
- DWI: high SI & low ADC
- dynamic contrast-enhanced sequences: usually hypervascular
differential diagnosis NET:
- (intra-peri)pancreatic accessory spleen Fig. 17
- aneurysms Fig. 18
- metastases Renal Cell Carcinoma or melanoma (see poster
M.
Eyselbergs ECR 2013)
clue to a correct diagnosis:
- same density/SI and contrast behaviour as spleen or vessels
- known RCC or melanoma
Pancreatic lymphoma
- primary pancreatic lymphoma is rare (0.5–2% of pancreatic tumours)
- mostly high-grade B cell (up to 75%)
- non-specific clinical findings (abdominal pain and weight loss)
- primary vs direct extension from adjacent peripancreatic lymphadenopathy
Two patterns of pancreatic lymphoma are described:
- large infiltrating lesion with poorly defined contours Fig. 19
< DD pancreatitis
In patients with diffuse infiltration of the pancreatic gland
without clinical or radiological signs of pancreatitis,
the
radiologist should be alert to the possibility of pancreatic
lymphoma
- rounded,
well-delineated mass Fig. 20
solitary
< DD adenocarcinoma and neuro-endocrine tumours
multiple
< DD metastases and granulomatous disease
Radiological clues suggesting lymphoma
< only mild dilatation of Wirsung’s duct
< infiltration of retroperitoneal or abdominal organs
< enlarged lymph nodes below the renal veins
< encasement of blood vessels but no thrombus or occlusions
< enhancement after IV contrast is more pronounced in the
pancreatic phase than in cases of adenocarcinoma but
less than in cases of NETs.
< strong diffusion restriction with low ADC.