The working group “Image-guided Minimally Invasive Breast Biopsies” (MIBB) of the Swiss Society of Senology (SGS) recordsall minimally invasive breast biopsies (VAB) performed in Switzerland for the purposes of documentation and quality assurance on behalf of the Federal Office of Public Health [BundesamtesfürGesundheit (BAG)] within the scope of legal specifications.
The MIBB working group has also developed quality guidelines which define minimal standards for the execution of a vacuum biopsy for the physician and the institute.
Stereotactic and sonographic VAB have been recorded since 2008.
MRI-guided biopsies have been recorded only since 2009.
As any subsequent surgery must also be recorded in addition to the histopathological result of the VAB,
the completion of a case may be delayed by up to 3 months.
The period between 2009 and 2011 was selected as the study period.
A total of 9113 records of vacuum-guided breast biopsies including the specification of the biopsy method were entered in the database during the study period between 2009 and 2011.
For data collection purposes,
ADJUMED-Services AG (Zurich,
Switzerland) provided an online questionnaire (one per intervention) to be completed by the institutions.
This database forms the basis of the present study.
The data used were available in anonymised form so that the identities of patients,
surgeons and institutions could not be ascertained.
As part of the information provided to patients before a vacuum biopsy,
a standardised information protocol informs the patients that the collected data “...
are made available to specialists and authorities (…) in anonymised form for evaluation within the scope of quality assurance ...” and that these data would only be published in anonymised form after the declaration of their consent[1],
whereby the respective surgeon/institution is responsible for informing the patient.
The responsible ethics committee confirmed that the evaluation and publication of data within the scope of this study is permissible.
For further evaluation,
all mammographically guided biopsies,
which were performed using either a stereotaxis table with the patient supine (n = 5340) or an “upright” device with the patient seated (n = 564),
were included in the “stereotactic vacuum biopsy” method.
A distinction between these two procedural variations was maintained only for the analysis of complications and the chronological development of the biopsy methods.
We refer to the literature with regard to the technical implementation of image-guided VAB [2,3]
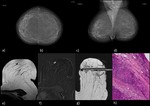
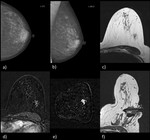
Images 1 and 2 display examples of MRI-guided vacuum biopsies performed at the institute of the lead author.
In both patients,
the second-look ultrasound was negative.
Histology resulted in one invasive carcinoma each.
Categorical variables were compared between groups using the chi-square test.
P-values less than 0.05 are considered statistically significant.
IBM SPSS Statistics version 20 (SPSS Inc.,
Chicago,
IL,
USA) was used for statistical analyses.
When single methods were compared with each other,
a Bonferroni correction was used to address the problem of multiple comparisons.
Because two methods (stereotactic biopsy und sonographic biopsy) respectively were compared with MRI-guided biopsy,
p < 0.025 was considered significant for these comparisons.
In the analysis of complication rates,
three methods (VAB using a stereotaxis table,
an “upright”device and ultrasound) were compared with the MRI-guided method.
Consequently,
p < 0.017 is considered significant in these comparisons.
Sensitivity,
specificity,
negative (NPV) and positive (PPV) predictive values were presented as percentages with 95% Wilson confidence intervals.
Microsoft Excel 2010 (Microsoft Corporation,
Redmond,
Washington,
USA) was used for additional analyses and figures.