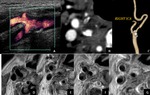
Patient selection and diagnosis.
Between January 2008 and December 2009,
102 patients (M/F 68/34) were referred to our institution for minor stroke or transient ischemic attack (TIA)risenup toonemonthbefore.The institutional review board at our institution gave full approval and waiver of informed consent for our retrospective study and approved our treatment protocol.Allpatientsenrolled wereat highsurgical risk.
At the hospitalization,
patients underwent epiaortic vessels duplex ultrasound imaging (DUI) and magnetic resonance (MR) scan of the brain.Neurologic symptoms were assessed by a neurologist,
who decided if patients should be considered symptomatic,
namely when a hemispheric or retinal transient ischemic attack or a non-disabling stroke or retinal infarct arose within 30 days before first evaluation.Stenoses greater than 50% were reported in 31 patients (30.4 %).
Patients affected by stenoses smaller than 50% or characterized by symptoms determined by a etiologyrelated to another cause from stenosis were excludedfrom the study.Patients presenting plaques determining stenoses greater than 50% underwent an instrumental in-depth evaluation with CT-angiography (31 patients,
100% of the whole patient cohort)in order to confirm stenosis degree and to obtain an objective and panoramic evaluation of the carotid plaque,
anatomy of Willis circle and aortic arch for a correct pre-procedural planning.Also a HR-MRI evaluation (31 patients,
100% of the whole patient cohort) was performed and only the T1-weighted before and after contrast media injection was considered.
Plaque morphology on CT-angiography was defined as mostly lipidic with an Hounsfield unit (HU) <50,
mostly fibrotic with an HU between 50 and 100 and mostly calcific with an HU >100.
Among these patients,
in 23 patients (71%) a mostly lipidic carotid plaque was diagnosed,
in 8 patients (29%) the carotid plaques were characterized by a mostly fibro-calcific component.In patients who underwent a HR-MR examination,
19 carotid plaque showed a hyperintense signal on after-contrast T1-weighted images,
while in 12 patients no significant contrast enhancement was pointed out.
CT acquisition protocol
CT exams were performed with a 64-slice CT scanner( LightSpeed VCT,
General Electric Medical System,
Milwaukee,
WI.
USA).
The acquisition stack extended from the ascending aorta to the intracranical circle in order to evaluate the anatomy and collateral vessels of the Willis circle for an eventual stop-flow protection device placement.
A second image stack was then acquired after intravenous administration of iodinated contrast material using a dual-head automated injector (Stellant,
MEDRAD,
Pittsburgh,
PA,
USA).
A dose of 80 ml of nonionic iodinated contrast material (Iomeron 400,
Bracco,
Milan,
Italy) was administered through an 18-gauge needle cannula placed in an antecubital vein,
followed by a 40 ml of saline solution injection,
both at a rate of 4 ml/s.
To synchronize the acquisition start with the arrival of the contrast agent in the coronary arteries,
the bolus-tracking technique was used.
Parameters for the contrast-enhanced scan were beam collimation 64×0.625 mm,
slice thickness 0.625 mm,
reconstruction increment 0.625 mm,
table feed 2.9 mm/rotation,
tube rotation 0.35 s,
tube voltage 120 kV,
dose modulation protocol (intensity 140–750 mA),
cranio-caudal scan direction.
Scan duration was 8-10 s; adsorbed dose was 8-10 mSv.
HR-MRI acquisition protocol
HR-MRI exams were performed with a 3T MRI (Philips Achieva,
Best,
Netherlands) equipped with a sinergy multichannel faced array head and neck coil,
using the following sequences:
-
T1 weighted black blood sequence on axial plane pre and post contrast administration (TR: 1000 ms,
TE: 20 ms,
matrix 372x442,
voxel size 0.43x0.43,
FOV 160x193 mm,
slice thickness 2 mm,
NEX 1) focus on brain and neck acquisition
-
T1 3D fast field echo (TR: 23 ms,
TE: 3.5 ms,
matrix 448x114,
voxel size 0.33x0.66,
flip angle 20°,
NEX 2)
-
Proton density weighted on axial plane (TR: 2000 ms,
TE: 20 ms,
matrix 264x258,
voxel size 0.53x0.54,
FOV 140x140 mm,
slice thickness 2 mm,
NEX 1)
-
T2 weighted black blood sequence on axial plane (TR: 2000 ms,
TE: 40 ms,
matrix 264x248,
voxel 0.53x0.56,
slice thickness 2 mm,
FOV 140x140 mm,
NEX 1)
In our retrospective study we considered only the pre- and after-contrast media injection T1-weigthed images,
in order to highlight plaques characterized by inflammation signs.
CAS Procedure
Thirty-one patients underwent to CAS procedure after the symptom onset.
Nineteen CAS procedures were performed in selected patients affected by magnetic resonance evidence of hyperintense plaque on after-contrast T1-weigthed images.
Patient cohort was characterized by 11 males (57.9 %) aged 75–90 years and 8 females (32.1%) aged 73–86 years.TwelveCASprocedures were performedinpatients who didn’t show any signs ofhyperintensity withinplaque on post-contrast T1-weighted ,
howeveraffected byrecent neurologic symptomsonset needing and a stenosis greater than 70%anendovasculartreatment.
They were 9 males (75%) aged 74-91 years and 3 females (25%) aged 77-84 years.
Treatment protocols
Once obtained a morphological classification of the carotid plaque,
patients were enrolled for an endovascular treatment.
Written patient informed consent was obtained from each patient prior to intervention.
Percutaneous treatment was performed in an endovascular suite.
Aspirin (100 mg/ day) and clopidogrel (75 mg/day) orticlopidin(500 mg/day) were administered to the patients during three days previous the procedure.
If this medical therapy couldn’t be administered,
patient received a loading dose of 500 mg aspirin and 300 mg clopidogrel before the intervention.
75 mg/day clopidogrel or 500 mg/dayticlopidinand 100 mg/day aspirin was continued for 45 days after stenting procedure.
At the procedure beginningadequateheparinheparin was given to elevate activated clotting time to 300 seconds.A right trans-femoral approach was obtained and a 6 Fr 10 cm long introducer sheath was placed.
To catheterize the right common carotid artery a 6 Fr 90 degree guide catheter(Boston Scientific,
Natick,
US )and an angle standard or stiff guidewire (Terumo,
Tokyo,
Japan ) were used,
while a 6 Fr 40 degree guide catheter allowed us to catheterize the left common carotid artery.
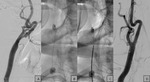
A diagnostic angiography was performed to highlight the stenosis localization and degree following NASCET classification.
In patients without any evidence of inflamed plaque an embolic protection device (EPD) (Epifilter,
boston scientific,
Natick,
U.S.; Spider fx,
Ev3; Emboshield,
Abbott Vascular,
Illinois,
U.S.) was positioned in the distal portion of internal carotid artery and a self-expandable closed-cells or multisegment design stent device was used.In all patients previously examined,
characterized by inflammation evidence,
and consequently highembolization risk as short-term complication,a Mo.Ma ultra protection device (Medtronic Invatec,
Roncadelle,
Italy) was positioned through a 9 Fr femoral approach.
The distal balloon of the device was inflated in external carotid artery with 5 ml contrast media and,
afterwards,
the proximal balloon in common carotid artery until flow was stopped.
A self-expandable closed-cells or multisegment design stent device (Wallstent,
Boston scientific,
Natick,
U.S.; Cristallo Ideale,
Medtronic Invatec,
Roncadelle,
Italy; X-Act,
Abbott Vascular,
Illinois,
U.S.
) was advanced over the 0,014 in guidewire.
Stent sizes were chosen on the basis of preprocedural CT evaluation.
At the end of the procedure the filter was collapsed and removed,
while,
in patients with Mo.Ma.
ultra device placement,
the blood in internal carotid was aspirate by three 20 cc syringes in order to remove accidental atheromatous debris; afterwards the distal balloon was deflated at first,
followed by the deflation of the proximal balloon.In all procedures the angioplasty wasn’t performed either before or after stent deployment to decrease embolization risk.Technical success was defined as stent deployment with residual stenosis not exceeding 30% of physiologic vessel diameter in absence of alteration of cerebral circulation.
Follow-up
All patients were examined by an expert neurologist before and after the procedure.
Main technical complications were defined as dissection,
vessel rupture,
thrombosis and occlusion.
Main clinical complications were defined as major stroke,
minor stroke,
TIA,
myocardial ischemia and death.
Neurologic complications were evaluated following National Institutes of Health Stroke Scale (NIHSS) (16)as clinical assessment and through the use of MRI with diffusion weighted sequences (DWI) and perfusion-weighted sequences (PWI) as instrumental evaluation.
Major stroke was characterized by an event characterized by NIHSS increase greater than 3 for a period longer than 24 hours; minor stroke was defined by an increase less than 3.
Hemodynamic parameters were monitorized during the post-procedural 4 hours.Patients were discharged after a mean period of five days after the endovascular procedure.DUI was performed at1,
3,
6 and 12 months during follow-up period to evaluate treatment results.
CT- or MRI-Angiography was indicated when DUI highlighted neointimal hyperplasia or hemodynamic alteration inside the stent.
Statistical analysis
All data were entered into a database for statistical processing.
Data were expressed as means plus one standard deviation (SD) or as percentages.
The comparison between groups was obtained using the Fisher exact test for categorical variables while t Student test was applied for continuous variables,
as appropriate.
Statistical significant was set at p<0.05.
All statistical analysis were performed using the software Epi Info 3.5.1 (CDC,
Atlanta USA).