Patient selection
A randomized prospective evaluation was performed on 60 Patients at high surgical risk who underwent CAS during a 14-month period. The institutional review board at our institution fully approved the treatment protocol.
All eligible patients were invited to participate in the study with an information letter.
On the basis of the written and spoken information,
the patient made the decision whether or not to join the study.
The informed consent was obtained from each participating patient.
Patients were informed of their right to withdraw from the study at any time.
The study was conducted in accordance with the current version of the Declaration of Helsinki.
Patient population
All patients were sorted using DUS evaluation as primary diagnostic tool.
Fifty-four patients (90%) were evaluated by the preprocedural Magnetic Resonance Angiography (MRA) integrated with HR-MR for plaque characterization; six (10%) patients underwent CTA forMRcontraindications. Patients presenting stenoses higher than 85% were excluded from the study,
as to avoid significant blood flow reduction to the brain during the IVUS catheter passage or thraumatism due to the catheter-to-plaque attritus.
Patients that presented carotid post-carotid
artery stenting (CAS) or post-carotid ewndarterectomy restenoses or stenosis determined by an aetiology different than atherosclerosis (e.g.
postattinic stenoses,
postsurgery stenoses,
stenoses determined by neoplasms),
were not enrolled in the study.Patients were matched by age,
risk factors and plaque pattern,
and divided in two groups: in group 1 VH-IVUS evaluation was performed during the CAS procedure,
before and after the stent deployment; in group 2 a conventional CAS procedure,
without the use of VHIVUS,
was carried out.
IVUS-assisted CAS was performed in 30 Patients (50%),
angiography-only CAS was performed
in 30 Patient (50%). Data about total procedure time,
fluoroscopy time and success rate were collected for all the 60 patients treated Table 1.
All Patients underwent a 5-day antiaggregation therapy with acetilsalicilic acid (100mg per day) and clopidogrel (75mg per day) or Ticlopidin (500 mg/die)
before the procedure.
Statistical methods
Analysis of the data gathered from this retrospective study was descriptive.
Simple descriptive statistics (n,
mean,
median,
SD,
minimum and maximum for continuous variables,
and n and percentage for discrete
variables),
graphs,
and patient listings were used to evaluate and summarize the data.
x2-test was used for the p-values calculation.
Carotid artery stenting procedure All procedures were carried out in an angiographic suite and performed by the same interventional radiologist.
Procedural and fluoroscopy times have been measured starting from the arterial puncture to the sheath removal. Common femoral artery access was obtained using Seldinger technique and guide catheter access into the
common carotid artery was obtained using a 7-Fr 10 cm cmlong introducer sheath (Terumo,
Tokyo,
Japan).
A diagnostic angiography was performed in order to confirm the carotid plaque location,
stenosis degree and to evaluate the anatomy and patency of the intracranial.
Five thousand international units of heparin were administered intravenously to maintain the active coagulation time between 200 and 250 s.
Distal embolism prevention was carried out using a cerebral protection device during both IVUS-guided and angiography-only procedures.
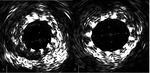
The Gray-Scale IVUS and VH-IVUS evaluations were performed using a 20-MHz IVUS probe (Eagle Eye,
Volcano Therapeutics,
Rancho Cordova,
California,
USA).
The IVUS catheter was washed with saline solution prior to its placement into the internal carotid artery through the guide catheter.
After the gain optimization,
the IVUS catheter was retrieved from the distal third of the internal carotid artery at a speed of 1.0mmper
s using a motorized pullback device.
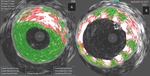
A 360 rotational and two-dimensional longitudinal ultrasound was performed for a plaque composition and stenosis degree assessment, the examination was recorded on a DVD.
This evaluation was also performed after stent deployment and compared during the procedure with the present one to assess wall integrity,
stent-wall apposition and stent expansion. Stent type and size were primarily chosen on the basis of the preprocedural MRA or CTA.
In the group 1 procedures, the prestenting IVUS findings were evaluated during the procedure in order to confirm or disprove the preprocedural stent choice.
Only two types of stents were used in this trial: an opencell nitinol one (Vivexx,
Bard Medical,
Covington,
USA) and a closed-cell medical steel one (Carotid Wallstent, Boston Scientific,
Natick,
USA).
The open-cell stent was
used for calcific-fibrous plaques,
the closed-cell one for mixed-lipidic ones.
The embolic protection device used was EPI Filter Wire EZ (Boston Scientific,
natick,
USA) in all the procedures but one,
in which a Spider FX
(Covidien,
Dublin,
Ireland) was used.
In both groups,
a diagnostic angiography was performed after the stent deployment and the embolic protection device retrieval,
as to evaluate the angiographic outcome of the procedure and the extra and intracranial vessels patency and blood flow.
All patients underwent a postprocedural neurological evaluation 24 h after the procedure and were discharged one day after the treatment,
with a 6 weeks double antiaggregation therapy.
Technical success was defined as a residual stenosis lower than 30%.
Follow-up
Neurological status and blood pressure of every patient were monitored for at least 24 h,
with a DUS examination before the discharge. Follow-up was performed at 1,
3,
6 months and each year after the procedure by DUS evaluation. Mean follow-up period was 235.3 months (minimum 15 – maximum 28).
A lifelong antiplatelet therapy was administered to each patient,
whereas clopidogrel or ticlopidin were administered for 6 weeks.