We retrospectively analyzed seven consecutive patients with eight renal artery aneurysms who underwent endovascular treatment from 2004 to 2009 using stent grafts in our institution.
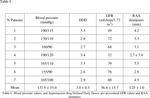
These patients were referred from Nephrology to Diagnostic Imaging Department to be examined due to arterial hypertension (137.9±35.0 mmHg) refractory to pharmacological treatment (Table I). Patients did not have a family history of hypertension,
and they suffered from headache and dizziness (2 patients),
macroscopic hematuria and backache (1 patient),
and abdominal bruits (3 patients).
In all patients have been previously examinated the following laboratory indexes: glomerular filtration rate (GFR),
creatinine,
nitrogen urea blood,
sodium,
potassium,
calcium,
phosphorus,
magnesium,
aldosterone,
renine,
angiotensin II,
ACTH,
cortisol,
adrenalin.
The GFR was calculated using the formula of the Modification of Diet in Renal Disease Study Group (17) while the drug therapy was quantified in Defined Daily Doses (Defined Daily Doses: DDD) (18).
Patients showed a GFR decrease (56.6±15.7 mL/min/1.73m2 ) (table I) associated with creatinine (4.6±3.1 mg/dl) and nitrogen urea blood (60±4.5 mg/dl) increase,
a slight increase of sodium
(NA+1=151±2.7 mEq/dl),
a potassium decrease (K+1=2±2.3 mEq/dl); the other blood parameters were within the normal range.
The diagnostic step involved Duplex UltraSound evaluation which has diagnosed the presence of the aneurismatic lesion on renal artery in six patients (86 %),
in three cases a hemodynamically significant stenosis was found,
in one (14 %) there were two bilateral aneurysms.
In one patient because the abdominal fat and meteorism,
ultrasound was not exhaustive and only morphologic and flowmeter parameters were evaluated.
Parameters showed a slight renal dimension decrease (maximum longitudinal length range between 7.5 and 9 cm) with a thinner cortical area.
The Duplex evaluation demonstrated in all patients a blood flow turbulence inside the lesion,
and, moreover,
in three patients pathological resistance index (RI= 0.9±0.1) and systolic velocity pick
(svp=270±16 cm/s) were detected.
The following diagnostic step has been a Computed Tomography-Angiography (CTA),
to obtain morphologic imagines in order to plan a correct procedure.
Patients with low GFR level (GFR<59 mL/min/1.73m2) were studied with an Magnetic Resonance-Angiography (MRA).
In both examinations we evaluated the side and the precise dimensions of the aneurysm,
presence of stenosis,
shape of the aneurysmatic sac,
length of the artery before and after the aneurysm neck, length and diameter of the neck,
components of the wall as fibrous or calcified tissue,
presence of
thrombosis within the sac and signs of active or recent bleedings.
Indications for treatment were: symptomatic lesions,
diameters of the aneurysm >1.5 cm,
rupture or dissection,
growth or increasing size with serial observations,
asymptomatic lesions in high risk patient ( e.g.
pregnancy,
one only kidney) (03,
04).
All treated aneurysms were type 1 following Rundback classification,
namely aneurysms arisen from the middle segment of the renal artery. Fusiform RAAs (type 2) located near a bifurcation of the main trunk and distal or intralobar RAAs (type 3) were excluded from the study (12). The institutional review board at our institution gave full approval and waiver of informed consent for our retrospective study and approved our treatment protocol.
Written patient informed consent was obtained from each patient prior to intervention.
The procedures were performed in the angiographic room with the control of patient’s parameters. After a local anestesia using lidocaine 2%,
a right transfemoral approach was obtained and a 6 Fr 10 cm long introducer sheath (Radiofocus Terumo,
Tokyo,
Japan) was positioned.
In only one
patient the transbrachial approach was preferred due to an acute angled of renal artery origin and the Shuttle introducer sheath (Cook Medical,
Bloomington,
US) was used.
In two cases we used a 7 Fr Pinnacle Destination guiding sheath (Terumo,
Tokyo,
Japan),
with a renal curve,
in order to achieve a good stability and to avoid a high diameter shaft in femoral access.
In one patient a 7 Fr 10 cm long introducer sheath was placed.
5000 IU heparin were administered to the patients.
In one patient
two 0,014 inches guidewires were used,
one in order to catheterize the artery and to give support in the complex anatomy vessel,
the other one to advance and deploy the device.
In two patients there were severe stenosis and,
in one case,
we used a monorail balloon catheter for a predilatation as Gazzelle 5 x 20 mm (Boston Scientific,
Natwick,
US) while in the other patient an Advanta balloon expandable covered stent was placed (Fig.
2).
Once obtained a stable access to the aneurysm,
the
device was advanced until the neck of the aneurysm and was deployed in the vessel to exclude the lesion from the blood flow.
The deployed stents had several sizes,
diameter size between 4 and 6
mm and length size ranged from 25 to 48 mm.
We deployed two Jostent peripheral stent-graft,
four Symbiot,
one AdvantaV12,
and two Direct-stent stent-graft.
In one case we used two Symbiot (Boston Scientific,
Natick,
US) in overlapping in order to cover completely the large neck of the aneurysm and to improve the strength of the stent structure,
while in one patient two Symbiot stents 5 x 45 mm were placed in both renal arteries due to the presence of bilateral aneurysms.
Two Jostent peripheral stent-grafts (Abbott,
Illinois,
US) were placed in two patients,
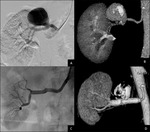
which dimensions were respectively 4-9 x 28 mm and 4-9 x 48 mm (Fig.
1),
respectively mounted on a 5 x 30 mm and 5 x 60 mm monorail balloon Ultrasoft (Boston Scientific,
Natick,
US).
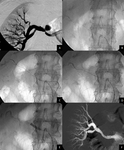
An Advanta V12 (Atrium, Hudson,
US) was deployed in one patient and it was 6 x 38 mm as measurement (Fig.
2).
We deployed two Direct-stent stent-graft (Minneapolis,
Minnesota,
USA) in two patients; they were 5 x 13 mm and 6 x 19 mm as measurement. The pre-procedural drug therapy has been based on double anti-aggregation treatment with aspirin (100 mg/die) and ticlopidin (500 mg/day) or clopidogrel (75 mg/day) for three days.
This therapy was administered to patients also in the after-procedural period for 6 weeks promoting re- endothelization; at the end of the period a life-long assumption of aspirin was kept.
Moreover a broad-spectrum antibiotic therapy was included,
based on penicillin (2g/day) per os for one day,
as
prophylactic for infection to stent-graft or to necrotic tissue in the case of embolic infarcts. The day after,
a Duplex ultrasound was performed to demonstrate patency of renal artery and exclusion of RAA and 2 days after procedure patients were discharged. Our follow-up consisted on clinical and instrumental examination which involved: laboratories indexes,
especially the GFR and blood pressure,
the administered pharmacological therapy before treatment and during follow-up period,
and CTA at 1-6-12 months and once a year after the last examination,
and Duplex US evaluation at 3-9 months after the procedure.
The treatment was considered successful when aneurysmatic sac exclusion,
patency of the placed stent-grafts and absence of complications related to device deployment can be observed on the CTA or MRA examination.
It represents the primary endpoint of our study. Secondary endpoint was represented by clinical and laboratory parameters improvement during the follow-up period evaluated by an expertise clinician. All data are expressed as mean ± standard deviation ( SD ).
The categorical data are expressed in percentages.
Statistical significance of differences between the data pre- and post-treatment was defined as P value <0.05.
We used the Student t test for continuous variables.
All statistical analysis were performed using the software Epi Info 3.5.1 (CDC,
Atlanta USA).