From January 2007 to April 2011,
26 Patients underwent to a post-EVAR endoleak treatment. The Institutional Review Board at our institution gave full approval and waiver of informed consent for our retrospective study and approved our treatment protocol.
Written patient informed consent was obtained from each patient prior to intervention.
Diagnosis of type II endoleak was performed with a follow-up computed tomography angiography (CTA) before the enrollment in the study. The indications for treatment were the same for all Patients: evidence of type II endoleak at 12-month follow up in large aneurysm (>5 cm) with no evidence of significant sac enlargement or a significant increase in aneurysm sac diameter (> 5 mm compared with the diameter highlighted at a previous examination) after at least 6 months of follow up. TCEE was proposed as treatment of type II endoleak. The study population consisted of 18 men and 8 women.
The mean age ± standard deviation was 73.6 ± 4.5 years old (range,
68 to 82 years). Time between initial AAA repair and endoleak treatment was 12.3 ± 4.2 months.
The mean increase of aneurysm sac diameter was 5.7 ± 4.2 mm. All data for each patient,
procedure,
and follow up were analyzed retrospectively.
Each of the 26 patients had concomitant comorbid medical condition (Table I).
PREOPERATIVE MANAGEMENT
Preoperative assessment included helical computed tomography angiography (CTA), performed by 64 -stations scanner ( LightSpeed VCT,
General Electric Medical System,
Milwaukee,
WI.
USA).
Helical images were obtained from the celiac artery to the common femoral arteries,
both before and after intravenous administration of 120 mL of nonionic contrast medium with a flow rate of 3 mL/s.
before the injection of contrast medium,
acquisition parameters for spiral CT were 8-mm collimation,
5-mm reconstruction,
and 1,5 pitch.
After contrast medium injection,
were used 5-mm collimation,
3 mm reconstruction and 1.5 pitch.
Images were obtained in both arterial and venous phases.
The scanning delay for the arterial phase was usually set at 30 seconds and the venous/delayed phase was set at 120 seconds so that late endoleaks could be identified.
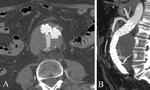
An experienced radiologist reviewed the images and evaluated the maximum transverse aneurysm diameter,
the presence and origin of endoleaks,
defined as the evidence of contrast enhancement within the aneurysm sac,
a tight adhesion between the aortic aneurysm and the caval walls in order to evaluate the transcaval approach feasibility,
and the “safety space”,
defined as the area between the aortic aneurism and endoprostesis walls to avoid device damage (Fig.1 A,B).
The procedures were performed into angiographic suite (Allura Philips Healthcare,
Best,
the Netherlands). We used TCEE technique in 26 Patient (100%).
TECHNIQUE
All procedures were performed in a dedicated angiography suite with the patient in a supine position.
A broad-spectrum antibiotic therapy was included in the immediate preprocedural period,
based on penicillin (2g/day) per os for one day,
as prophylactic for infection to stentgraft
or intrasac necrotic tissue. A percutaneous transfemoral vein approach was performed in all patients. The common femoral vein was punctured previous a local anesthesia (2% lidocaine) and a standard 0.035” J tipped 180 cm long hydrophilic guidewire (Radiofocus,
Terumo,
Tokyo, Japan) was advanced into the inferior vena cava (IVC). A 10F introducer sheath 40 cm long was placed and advanced into the IVC to give stability and allow a safe puncture.
Transcaval puncture of the aneurysm sac was performed with the same technique for all patients,
using a dedicated kit for trans-jugular porto-sistemic shunt (Angiodynamics,
Queensbury NY,
US).
The angle of the metallic needle has been handmade modified from 40 degree to 60/70 degree to ensure the correct puncture of the abdominal aortic aneurysm and to reduce bleeding risk due to the more orthogonal access obtained on caval wall.
The 8 French transjugular curved catheter and the 5F curved guiding cannula was advanced through the introducer sheath over the guidewire.
The system direction is verified correlating the fluoroscopic images with the pre-procedural CT examination.
The stent graft,
vertebrae and,
when present,
aortic wall calcifications help to orienting of the aneurysm and the introducer assembly.
When there are no calcified walls, ultrasonographic guidance can be helpful.
The shunt set must be completely wedged along the caval wall at a site in which there is a tight adhesion between the caval and aneurysm walls.
After checking for the system’s wedging against the caval wall with a contrast injection,
the Colapinto needle assembly with the 5F curved guiding cannula was advanced through the system and finally the aneurysm sac was punctured according to the landmarks gathered
from the pre-procedural CT scan. The 5 Fr cannula with its catheter was introduced under fluoroscopic guidance through the Colapinto needle.
The flexible puncture needle was removed,
and a 0.035” standard J tipped 180cm long hydrophilic guidewire was slowly advanced into the aneurysm sac until a low resistance path was engaged.
This was considered as an indirect sign of the passage of the guidewire into an unthrombosed area,
suggestive of the site of the endoleak.
The 4 Fr Cobra catheter was then advanced over the guidewire into the area of the presumed endoleak.
This
diagnostic catheter allowed us to improve the guidewire control within the thrombus in the aneurysm sac.
The backflow of blood through the catheter,
after removal of the guidewire,
confirmed its position within the endoleak.
The placement of the needle/sheath system within the endoleak nidus is not always possible, but the thrombus can be broken into small clots by the guidewire as it enrolls inside the sac.
The contrast media was manually injected with low pressure to visualize the endoleak. It was performed a latero-lateral projection to highlight the lumbar arteries,
a right oblique projection for the inferior mesenteric artery and a postero-anterior one for the sacral artery. In this setting an optimal visualization of the feeding vessels was obtained to make the feeding arteries catheterization manageable. Intrasac pressure measurements were performed during procedure,
by using a pressure transducer for invasive pressure monitoring the ACIST System.
(Bracco,
Milan,
Italy).
After connection of the 5F catheter to the transducer and before glue injection,
intrasac pressures was recorded in all patients,
indicating that blood flow was present inside the sac. Intrasac pressure was recorded after the performance of each glue injection,
usually 3 minutes after the injection.
Both before and after embolization,
pressure measurements was
recorded in different places,
even though the range of positions for the catheter tip was small.
A stable intrasac pressure in an aneurysm with a previous systolic-diastolic waveform pressure or a reduction of at least 50 mm Hg,
or both,
were considered as signs of thrombosis of endoleak,
signifying the abolition of arterial blood flow inside the endoleak itself.
Initially,
in nine cases (34.6 %),
we performed an unselective embolization of aneurysm sac using a combination of 7-15 platinum fibered coils (diameter range: 6-9 mm),
1ml of Glubran 2 acrylic glue (GEM,
Viareggio,
Italy) mixed to Lipiodol (volume ratio1:3),
in order to allow its fluoroscopic visualization,
and thrombin.
This procedure was considered in our study an unselective transcaval endoleak embolization (uTCEE).
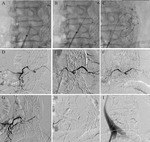
In 20 cases (76.9 %),
during the procedure,
the embolization of the feeding branch with fibered coil was performed obtaining a selective occlusion of the in-flow and out-flow vessels,
defined as selective TCEE (sTCEE) (Fig.
2 A-I).
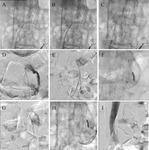
In only three patients embolization of the sac with acrylic glue mixed to Lipiodol and concomitant coils in the feeding vessel
was performed due to the dimension of the aneurysm sac (Fig.4 A-I).
Intrasac embolization is stopped only when there is no more evidence of flow inside the aneurysm sac.
This was defined as no evidence of blood dripping from the 5F catheter and that contrast is stable inside the sac.
Stable intrasac pressure within an aneurysm that had a previous systolic-diastolic waveform or its reduction by a minimum of 50 mm Hg,
or both,
was considered as further evidence of successful embolization.
At the end of all procedures,
we check for any lesions at the site of caval puncture by performing a cavography through the sheath before removing the venous sheath.
In the first 24 hours after endovascular treatment,
all patients were observed in the our Department during which central venous blood pressure,
arterial pressure,
heart rate,
and peripheral oxygen saturation were monitored.
They underwent unenhanced CT scan at 24 hours.
They were all discharged at 24 hours from the procedure.
A broad-spectrum antibiotic therapy,
based on amoxicillin clavulanate (Augmentin; GlaxoSmithKline,
Brentford,
Middlesex,
United Kingdom) at a dose of 2 g/day per os,
was administered one day before the procedure as prophylactic for infection to stent-graft or intrasac necrotic tissue.
FOLLOW UP
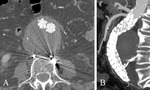
Follow-up was performed by clinical examination and CTA evaluation at 1,
3,
6,
12 and each following year,
by use the same standard acquisition protocol followed as for the preoperative Imaging,
in case of no pathology recurrence (Fig.
3 A,B; Fig.
5 A-D).