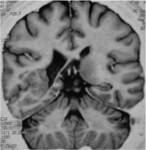
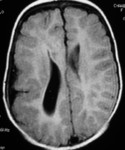
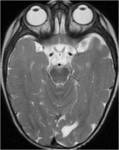
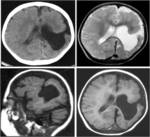
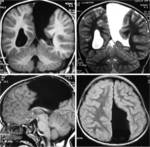
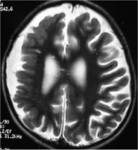
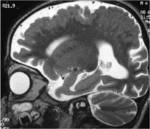
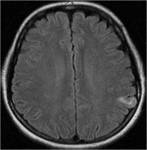
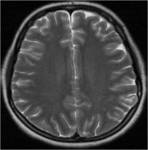
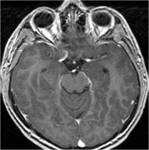
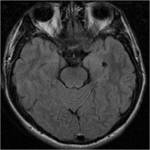
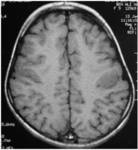
MRI analysis evaluates particularly the cortical thickness,
sulcal and cortical morphology,
gray-white matter junction,
and looks for gray matter in abnormal location.
These data coupled with the familial history,
the seizure characteristics and genetic findings should allow an appropriate classification of the lesions.MRI results: Hemimegalencephaly (n=2),
cortical dysplasia (n=2),
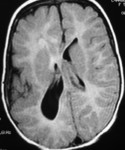
polymicrogyria (n=5),
schizencephaly (n=6) (5 patients with open-lip schizencephaly,
and 1 patients with closed-lip schizencephaly),
lissencephaly (n=1),
dysembryoplastic neuroepithelial tumors (DNTs) (n=2),
and nodular heterotopias (n=2).
DISCUSSION
Introduction
Malformations of cortical development (MCD) are a group of disorders characterized by a congenital abnormal structure of the cerebral cortex.
In general,
malformations are defined as structural abnormalities caused by a disturbance in cell organization or function within a tissue type.
When a disturbance results in an abnormal structure of the cerebral cortex we call this: malformations of cortical development.
MCD are increasingly recognized as important causes of epilepsy,
developmental delay and other neurological disorders.
MR analysis evaluates particularly the cortical thickness,
sulcal and cortical morphology,
gray-white matter junction,
and looks for gray matter in abnormal location.
These data coupled with the familial history,
the seizure characteristics and genetic findings should allow an appropriate classification of the lesions.
Classification
Malformations of cortical development can be classified into several subgroups,
based on the MRI.
The revised Barkovich classification is most widely accepted,
and is based on the proposed underlying mechanisms with a mixture of genetic and imaging criteria.
Malformations of cortical development
At all stages of cortical development trouble may arise,
and abnormal development can result in abnormal anatomy.
The gold standard of the diagnosis is microscopic neuropathology,
but improvements in neuroimaging techniques enable the diagnosis of many malformations of cortical development during life.
The investigation of choice to visualize the central nervous system is MRI imaging.
The classification system in broadly based on the following categories:
1.
Malformations due to abnormal neuronal and glial proliferation or apoptosis
1.1 Decreased proliferation/increased apoptosis: Microcephaly with/without normal cortex.
1.2 Increased proliferation/decreased apoptosis: Megalencephaly with/without normal cortex.
1.3 Abnormal proliferation (abnormal cell types)
2.
Malformations due to abnormal neuronal migration
2.1 Lissencephaly/subcortical band heterotopia spectrum
2.2 Cobblestone complex
2.3 Heterotopia
3.
Malformations due to abnormal cortical organization (including late neuronal migration)
3.1 Polymicrogyria and schizencephaly.
1- Disorders of proliferation
1.1- Microcephaly with simplified gyration (MSG)
MRI features of MSG
Fewer glia and/or neurons result in a small brain (microcephaly).
This is generally associated with a more or less simplified gyral pattern.
Gyration is less complex and the number of gyri is reduced.
There is a continuum between normal gyration and a simplified pattern however,
and a clear distinction is not always possible.
It is more informative to look at the complexity of the sulci formation,
which can give an indication of the maturity of the gyral pattern.
Clinical features of MSG
In children with autosomal recessive microcephaly with simplified gyral pattern,
head circumference is more than 3 SD below the mean.
Children are generally mentally retarded,
but show no specific neurological abnormalities such as epilepsy or spasticity.
Syndromal variants of MSG,
such as Amish lethal microcephaly or Nijmegen breakage syndrome usually do show more severe neurological deficits and a reduced life expectancy.
Causes of MSG
MSG can be a consequence of fewer proliferative cell cycles in early development,
or of loss of progenitor cells or neurons by apoptosis.
Classic microcephaly with simplified gyral pattern is also known as ‘microcephalia vera’,
and several causative genes have been identified that are all involved in mitosis organization (see appendix).
Mutations cause a reduction in the number of times that neuroblasts are able to divide,
and therefore a reduction in the resulting total number of neurons and glial cells.
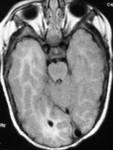
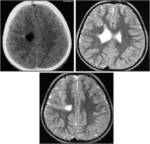
1.2- Hemimegalencephaly
Hemimegalencephaly or unilateral megalencephaly is a rare but well-known congenital malformation with ipsilateral enlargement of the hemicerebrum.
Hemimegalencephaly is characterized by enlargement of all or parts of a cerebral hemisphere.
The affected hemisphere may have focal or diffuse neuronal migration defects,
with areas of polymicrogyria pachygyria,
and heterotopia.
The etiology of this condition is unknown,
though it is speculated that it involves abnormalities of neuronal differentiation and cell migration in a single hemisphere.
MRI features of MSG
MR imaging is the imaging technique of choice for diagnosis of this condition.
In addition to excessive growth limited to 1 cerebral hemisphere,
MR imaging studies have revealed various other abnormalities accompanying this condition,
such as enlargement of the lateral ventricle,
an abnormal gyral pattern with a thick cortex,
gliosis in the white matter on the affected side,
and abnormal myelination.
Clinical features of MSG
Symptoms of megalencephaly may include delayed development,
convulsive disorders,
corticospinal (brain cortex and  spinal cord) dysfunction,
and seizures.
spinal cord) dysfunction,
and seizures.
2- Disorders of differentiation
Local abnormalities of the cellular structure of the cortex are called focal cortical dysplasias.
Cortical dysplasias are thought to either result from abnormal maturation or differentiation of neurons or from abnormal migration (based on ‘migration lines’ of neuronal tissue in the white matter proximal to the dysplasia). Cortical dysplasias consisting of purely heterotopic ‘normal’ neurons (type 1 cortical dysplasia) should be considered ‘true’ migration disorders,
but are also included in this paragraph for the sake of clarity.
Clinical features of cortical dysplasia
Cortical dysplasias are highly epileptogenic,
and are rigorously sought for in children with intractable focal epilepsy,
as they are the most frequent cause of focal refractory seizures in this age group.
Epilepsy surgery has a high chance of success in patients with cortical dysplasia,
but higher in type II than in type I cortical dysplasia (see below).
MRI features of cortical dysplasia
Clues to look for are focal cortical thickening,
blurred gray-white matter junction,
and related ‘migration lines’.
These are gray matter lines in the subcortical white matter.
If these are visible and extend to the lateral ventricle,
the abnormality is called a transmantle dysplasia.
MRI features suggestive of a type II cortical dysplasia (with abnormal neurons) are increased cortical thickness,
transmantle signs,
and FLAIR or T2 weighted signal change.
Migration lines and the gray-white matter junction are best appreciated on T1 weighted imaging.
T2 weighted images or FLAIR images show associated white matter hyperintensity or gliosis.
Causes of cortical dysplasia
One of the most important causes of focal cortical dysplasia due to abnormal proliferation is tuberous sclerosis complex.
Growth regulation is impaired in this neurocutaneous disease,
not only leading to increased number of neurons,
but also the formation of abnormally large neurons that form the characteristic cortical ‘tubers’ or hamartomas of the disease.
It is a multi-organ disease and is not further explored in this thesis.
Pathology of cortical dysplasia
Histopathologically,
cortical dysplasias are graded by the Palmini grading system. Dysmorphic neurons are misshapen cells with abnormal orientation,
size,
cytoskeletal structure,
and atypical dendritic processes.
Balloon cells are abnormal cells with pale,
eosinophilic cytoplasm and eccentric nucleus or nuclei,
as some are multinucleated.
3-Disorders of migration
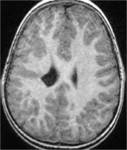
3.1 Lissencephaly
MRI features
In the lissencephaly-pachygyria-subcortical band heterotopia spectrum,
MRI features can distinguish well between the different types of lissencephaly and give direction to additional genetic tests.
In type 1 or classic lissencephaly the cortex is smooth and thickened (>10mm) with a cell sparse zone.
Milder types of lissencephaly show thickened gyri (pachygyria) or fairly normal gyri with a band of gray matter below,
the subcortical band heterotopia,
consisting of neurons arrested during migration.
Taking note of which areas of the brain are most affected aids investigations into the cause.
This also goes for associated abnormalities of the corpus callosum or cerebellum.
Clinical feature of lissencephaly
Lissencephaly type I was the earliest recognized MCD,
and consequently much is known about the clinical features.
Signs and symptoms are very diverse and range from normal intelligence to severe mental retardation.
There is a relationship between the severity of the lissencephaly/pachygyria grading based on neuroimaging and outcome with respect to the severity of psychomotor retardation,
motor symptoms and epilepsy.
Most to all patients have epilepsy.
Lissencephaly type I is associated with facial dysmorphic features when part of the Miller-Dieker syndrome.
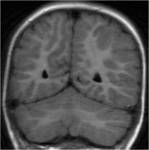
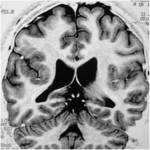
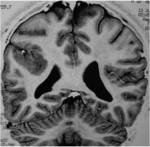
3.2 Heterotopia
MRI features of heterotopia
Heterotopia are formed of neurons that have not migrated to their destination in the cortex.
As they are otherwise normal neurons,
they have all characteristics of gray matter on MRI.
Heterotopia can be unilateral or bilateral,
and isolated versus continuous.
Bilateral continuous periventricular heterotopia with an otherwise normal brain,
except for an enlarged retrocerebellar space,
is the classical phenotype of FLNA mutations.
Distinctly different types of heterotopia are those found in the cerebral white matter.
Clinical features of heterotopia
Clinical features are very diverse,
may give no neurological complaints and can be a chance finding.
Psychomotor development may be completely normal or delayed.
Periventricular nodular heterotopias give a high risk of epilepsy,
but seizures may be delayed until adulthood.
Associated congenital anomalies may give a clue to the underlying cause,
e.g.
cardiac malformations are associated with FLNA mutations.
Causes of heterotopia
The most common monogenetic cause are loss-of-function mutations in X-linked FLNA.
This causes bilateral periventricular nodular heterotopia,
but also cardiac malformations in the outflow tract and Ehlers-Danlos like skin and joint anomalies.
Rarely bilateral PNH are caused by autosomal recessive mutations in ARFGEF2,
also associated with microcephaly,
severe mental retardation,
movement disorder and epilepsy.
Pathology of heterotopia
In bilateral periventricular nodular heterotopia due to FLNA mutations the heterotopias contains normal appearing neurons.
Apart from the heterotopia,
glomeroid vascular abnormalities have been reported in autopsy material.
Interestingly,
nodular heterotopia show an intrinsic laminal specificity that is similar to that of the cortex.
This suggests that the neurons in the heterotopia fail to migrate,
but do attempt to differentiate normally.
4.
Disorder of cortical organization:
Polymicrogyria is the most heterogeneous malformation of cortical development,
both in appearance as in causes.
It can be seen as an isolated malformation,
but also in combination with periventricular nodular heterotopia or other brain malformations such as corpus callosum agenesis or cerebellar abnormalities.
4.1 Polymicrogyria
MRI features of polymicrogyria
Polymicrogyria is probably the most common MCD and also probably the most difficult malformation to recognize on MRI imaging.
This is partly due to the marked change in its appearance during the first years of life,
and partly to its heterogeneous nature.
In newborns the cortex is thin and due to a lack of myelination the gray-white matter border is harder to appreciate.
In these infants the polymicrogyric cortex shows numerous,
very small ripples.
After myelination the polymicrogyric cortex appears thickened with an unclear white-matter junction.
The surface of the cortex may appear lumpy-bumpy or may seem smooth due to fusion of the upper layer of the cortex across adjacent microgyri.
Clinical features of polymicrogyria
Bilateral perisylvian polymicrogyria results in the so-called perisylvian syndrome.
Motor function of the oral musculature is impaired,
resulting in speech delay,
dysarthria and dysphagia.
This clinical syndrome has also been described as Worster-Drought syndrome,
however also children without PMG can have this syndrome.
PMG can be unilateral or bilateral.
Causes of polymicrogyria
Polymicrogyria is the probably the MCD with the most heterogeneous causative mechanisms.
Several non-genetic causes have been described,
such as congenital cytomegalovirus infection or prenatal ischemia.
Several genes and microdeletion syndromes have been associated with PMG,
but each seems to be responsible for only a small group of patients.
The most important established genetic cause is the 22q11 microdeletion syndrome.
Pathology of polymicrogyria
PMG is characterized by an excessively folded cortical ribbon of miniature,
individually thin gyri,
which may be fused together or piled on top of each other.
Predilection areas are around the insula and the central fissure.
The normal layered cortex can be reduced to four layers or no clear layers may be seen at all.
The four layers consist of a molecular layer and two layers of neurons separated by an intermediate layer of few cells.
Pathology can be the only way to distinguish PMG from cobblestone complex,
as brain MRI may be ambiguous.