We retrospectively reviewed 12 cases of inferior mesocaval shunts (IMS) identified in our tertiary referral hospital between 2004 and 2010,
including 6 men and 6 women aged between 41-82 years (mean age 64’7 years),
who underwent abdominal MDCT for different purposes,
the most frequent one for evaluating liver disease. This is,
to the best of our knowledge,
the largest serie reported (Table 3).
10 of 12 patients had chronic hepatopathy,
including Hepatitis B Virus,
alcoholic diseases and unknown cause,
and none of them had a history of abdominal trauma or surgery.
Studies were performed with a 4-row (n=3) or 16-row (n=9) MDCT scanner.
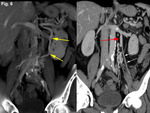
We revised imaging findings paying special attention to venous phase in axial plane and also in coronal multiplanar reconstruction (MPR).
Maximum intensity projection (MIP) and volume rendering (VR) were systematically performed on a workstation to analyze portosystemic collateral vessels.
Studies were analyzed by three different radiologists,
assessing the pathway,
the drainage level and the size of the shunt,
as well as the presence of other portosystemic shunts that can lead to encephalopathy.
ANALYSIS OF RESULTS
IMV-systemic shunts are rarely encountered,
although they have been reported with a frequency of about 13% in portal hypertension (including via the rectal vein,
a type of shunt not depicted in our serie).
Mesenteric collateral vessels may arise from the inferior mesenteric veins,
and may ultimately drain into the systemic venous system via the retroperitoneal or pelvic veins.
It is usually difficult to trace their drainage due to the tortuous,
cirsoid or dilated course.
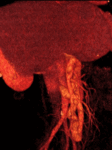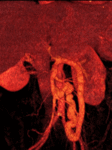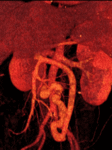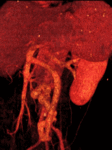
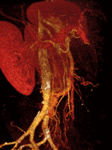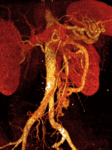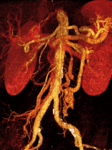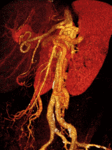
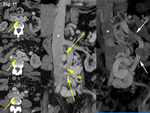
We identified 17 shunts in 12 patients,
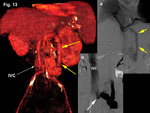
being most of them (9/12)formed by a single drainage vein (Figures 1,
2,
3,
5,
6,
7,
8,
9,
12,
13,
and 14),
mainly to the IVC.
However,
several drainage veins have been reported,
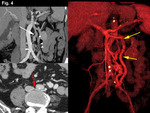
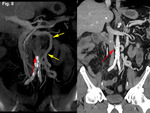
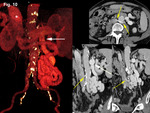
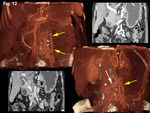
being observed in our series one patient with two (Figure 4) and two patients with three portosystemic connections (Figures 10 and 11) in most cases draining directly into the IVC.
Recognition of these drainage pathways is important to guide the treatment.
The mean size of the shunts was 7’2 mm (ranging from 4 to 13 mm).
Noteworthy that the largest sizes (12 and 13 mm) were observed in those who had until 3 shunts.
The lowest sizes (4 and 5 mm) were visualized in those patients without liver cirrhosis or portal hypertension, and are considered to be congenital,
particularly when the shunt vessel is single,
and occurred in one young patient.
Several different pathways from IMV toward IVC have been described.
Despite of the small percentage of cases described in literature of direct flow from the IMV toward the IVC and directly to the IVC (mesenteric-caval),
we have observed it in 12 of 17 (70 %) of the portosystemic shunts,
passing the aorta indistinctly anteriorly (n=6) (Figure 1,
2,
8,
12 and 13) or posteriorly (n=6) (Figures 4,
10,
11).
We have noticed that the 3 patients who presented with encephalopathy had developed this last type of drainage,
with mean size of the shunt of 9’3 mm (8,
8 and 12 mm),
slightly higher than de mean size of all the shunts (7’2 mm).
Two cases presented with drainage through the left renal vein,
(mesenteric-renal) (Figures 5, and 10), having both of them a large diameter (12 and 13 mm).
The mesenteric-gonadal shunt are often due to ileocolic veins from the superior mesenteric vein,
anastomosed with right gonadal vein,
while the anastomosis between the left gonadal vein with a venous network developed from the IMV is exceedingly rare,
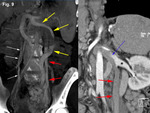
althogh we have identified this type of pathway in 3 cases (Figures 4,
6,
and 9).
In such cases,
a variceal plexus around the uterus may develop (Figure 9),
affect the opposite gonadal vein and even produce ileocecal varices.
Another type of drainage pathway described is toward the internal iliac veins (mesenteric-iliac),
not identified in our series.
Two patients presented 3 drainage pathways,
one of them with all connections toward the IVC,
meanwhile the other patient with two shunts to the IVC and the other one to the left renal vein.
In 7 cases the IMV was larger (12’5 mm of mean size) than the trunk of the portal vein (10’8 mm of mean size).
Although our studies are not dynamic studies for evaluating vascular flows,
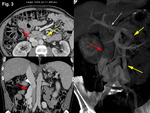
we observed in 4 patients a jet within the IVC at the shunt level (representing inflow from an opacified drainage pathway toward an unenhanced IVC) (Figure 3) and a “flow artifact” in left renal vein at the shunt level during the arterial phase (representing inflow from unenhanced collateral vessel toward a high opacified left renal vein) (Figure 9).
SYMPTOMS RESULTING FROM THE SHUNT
The clinical manifestations of IMV-IVS shunts are the result of the retrograde direction of flow from the IMV and the size of the shunt.
In cirrhotic patients,
the development of hepatic encephalopathy depends largely on the function of residual hepatocytes and the portal blood diverted to the systemic circulation.
In our serie 3 patients presented encephalopathy (disorientation and/or disturbance of consciousness),
corresponding all of them to shunts toward the IVC and with a mean size of 9’3 mm,
slightly higher than the mean size of all the shunts (7’3 mm).
The IMV-IVC shunt is not associated with gastrointestinal bleeding,
although exceptional ruptures into the retroperitoneal cavity have a severe prognosis.
TREATMENT
In some series it’s been observed that with treatment there’s usually improve of symptoms secondary to portosystemic encephalopathy,
coinciding with decrease of the serum ammonia levels.
The treatment of symptomatic shunts includes conservative therapy in most cases,
related to the symptomatolgy (encephalopathy),
but surgical ligation,
and transcatheter embolization (percutaneous transhepatic obliteration or retrograde transcaval obliteration) have also been described.
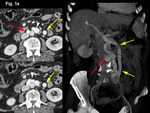
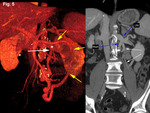
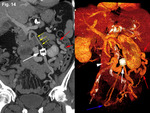
One case of our serie was treated with embolization,
placing an Amplatzer Vascular Plug (AVP) in the IMV. A follow-up MDCT demonstrated an important decrease of IMV size,
although persistence of the portosystemic shunt was detected through the left colic vein,
as well as dilated superior haemorrhoidal veins,
not present before Amplatzer placement (Figure 14).
However,
the therapeutic blockade in some cases can lead to overload of the portal venous system,
increasing the risk for ascites formation and variceal bleeding.
This is the reason why it’s widely believed that the management of these patients should be judged according to their characteristic risk/benefit ratio.
Some recent studies have shown that proangiogenic molecules like VEGF or PDGF may have an important role in splanchnic neovascularization.
According to this,
antiangiogenic therapies like sorafenib® could be useful to reduce portosystemic collateral circulation,
improve splanchnic hyperdynamics and decrease portal pressure.