IMAGING MANIFESTATIONS:
Paraduodenal pancreatitis manifests as either cystic lesions (cystic variant) or a solid-mass occurring predominantly in and around the minor papilla (solid variant) (Fig-7).
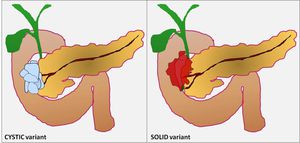
Fig. 7: Adsay and Zamboni recognized two types of Paraduodenal Pancreatitis: one characterized by cystic changes and the other characterized by solid lesions.
IMAGING MODALITIES:
Endoscopic ultrasongraphy (EUS) and MRCP are considered the preferred imaging modalities amongst various options available including magnetic resonance imaging (MRI),
computed tomography (CT) and trans-abdominal ultrasonography.
ABDOMINAL ULTRASONOGRAPHY may be able to sometines demonstrate:
- A hypoechoic mass adjacent to the pancreatic head with
- Thickening of the second part of the duodenum.
CT SCAN can reflect the histological characteristics of the disease.
At CT examination,
paraduodenal pancreatitis is seen as:
- A hypoattenuating poorly enhancing soft tissue mass in the pancreatico-duodenal groove (solid variant).
Duodenal wall is frequently involved and thickened.
- This may be accompanied with cysts in the duodenal wall and/or the groove (cystic variant).
Patient-1: A 35-year old alcoholic male with gradually progressive abdominal discomfort (Fig: 8-10).
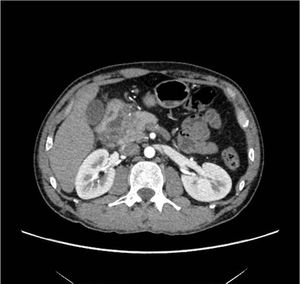
Fig. 8: Axial CECT reveals a hypoenhancing inflammatory mass within the pancreaticoduodenal groove.
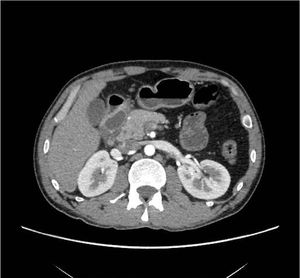
Fig. 9: Axial CT section in the same patient reveals cystic component indenting and displacing the duodenum. Also seen is the leftward displacement of a normal appearing gastroduodenal artery (GDA), a key imaging finding to differentiate it from groove pancreatic carcinoma.
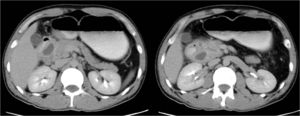
Fig. 10: Follow-up CT of the same patient with oral contrast adminstration shows predominantly cystic nature of the pathology.
Patient-2: A 30-year-old male with CYSTIC variant of the disease (Fig-11,
12).
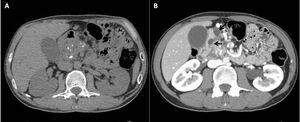
Fig. 11: (A) Plain CT abdomen reveals multiple calcific specks in the pancreatic parenchyma consistent with chronic calcific pancreatitis. (B) CT depicts hypoattenuating cysts (arrows) along the medial wall of duodenum with mild duodenal wall thickening.
References: Image reproduced from: Arora A, Mukund A, Thapar S, Jain D (2011) Cystic dystrophy of the duodenal wall. 10.1594/EURORAD/CASE.9429
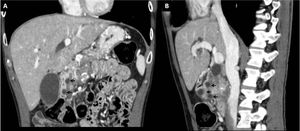
Fig. 12: Coronal CT of the same patient (A) displays cysts embedded in the medial wall of duodenum causing intrinsic contour bulge and medial displacement of the enhancing duodenal mucosa (arrows). (B) Intraluminal projection of the cysts and mucosal indentation can be well appreciated on the sagittal image.
References: Image reproduced from: Arora A, Mukund A, Thapar S, Jain D (2011) Cystic dystrophy of the duodenal wall. 10.1594/EURORAD/CASE.9429.
Patient-3: A 51-year old alcoholic patient with SOLID variant of the disease (Fig-13,
14).
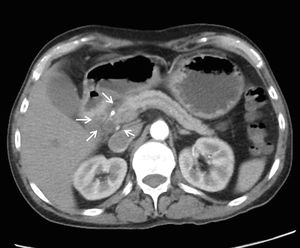
Fig. 13: The normal fat-attenuation of the groove between the pancreas and duodenum is obliterated. Instead an ill-defined hypodense soft tissue is seen within the pancreatico-duodenal groove.
References: Image reproduced from: Arora A, Mukund A, Thapar S, Jain D (2011) Groove pancreatitis: CT, MRI & MRCP findings. 10.1594/EURORAD/CASE.9404
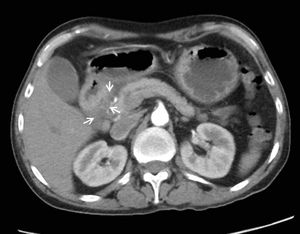
Fig. 14: The lesion is hypoenhancing as compared to the adjacent pancreatic parenchyma.
References: Image reproduced from: Arora A, Mukund A, Thapar S, Jain D (2011) Groove pancreatitis: CT, MRI & MRCP findings. 10.1594/EURORAD/CASE.9404.
Patient-4: A 32-year old chronic alcoholic male with recurrent abdominal pain and obstructive jaundice (Fig: 15-17).
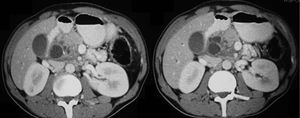
Fig. 15: Axial CECT reveals cystic changes along the pancreaticoduodenal groove indenting the duodenum with suggestion of ductal dilatation within the pancreas.
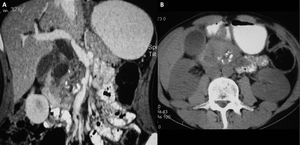
Fig. 16: Paraduodenal cystic changes are associated with changes of chronic calcific pancreatitis.
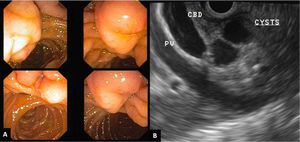
Fig. 17: Upper GI endoscopy (A) confirms submucosal cystic changes in the duodenum causing mucosal contour bulges. Endoscopic US shows clear cystic (anechoic) lesions compressing the common duct with upstream duct dilatation.
MR IMAGING can also readily demonstrate the pathologic features characteristic of the disease i.e.
- The fibrous tissue or cysts in the pancreaticoduodenal groove,
thickened and inflamed duodenal wall,
and/or duodenal wall cyst formation.
- The most characteristic finding on MRI is sheet-like mass (solid subtype) corresponding to the fibrous scar in the groove.
- This exhibits hypointense signal relative to the pancreatic parenchyma on T1-weighted images with isointense/ hypointense/ or slightly hyperintense signal on T2-weighted images.
- On contrast enhanced imaging the mass shows delayed enhancement reflecting its fibrous nature.
- Cystic lesions of the groove and/ or the duodenal wall are well displayed on T2-weighted images.
Patient-1: A 44-year old chronic alcoholic male with CYSTIC variant of the disease diagnosed on MRI (Fig-18,
19).
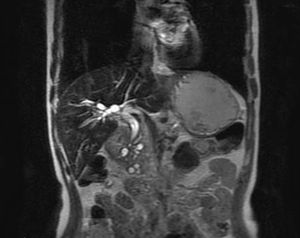
Fig. 18: A 44-year-old chronic alcoholic male. Paraduodenal cystic lesions are well seen on this coronal T2-weighted MR image. Smooth distal CBD stricture is also seen along with dilated pancreatic duct.
References: Image reproduced from: Arora A, Mukund A, Thapar S, Jain D (2011) Paraduodenal pancreatitis. 10.1594/EURORAD/CASE.9436.
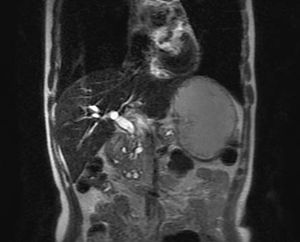
Fig. 19: Multiple paraduodenal cystic lesions are present along the pancreatic head and pancreatico-duodenal groove.
References: Image reproduced from: Arora A, Mukund A, Thapar S, Jain D (2011) Paraduodenal pancreatitis. 10.1594/EURORAD/CASE.9436.
Patient-2: A 51-year old chronic alcoholic male with recurrent abdominal pain showing SOLID variant of the disease on MRI (Fig: 20-22).
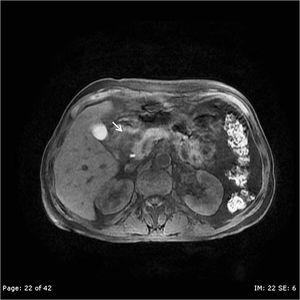
Fig. 20: A 51-year-old chronic alcoholic with recurrent episodes of upper abdominal pain. Axial T1-weigted MR image depicts an ill-defined soft tissue within the pancreatico-duodenal groove closely abutting the pancreatic head consistent with SOLID subtype of the disease.
References: Image reproduced from: Arora A, Mukund A, Thapar S, Jain D (2011) Groove pancreatitis: CT, MRI & MRCP findings. 10.1594/EURORAD/CASE.9404.
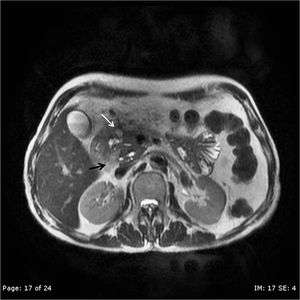
Fig. 21: The soft tissue in the groove is of intermediate signal intensity (black arrow) on T2-weighted MRI. It also shows cystic areas (white arrow) and is closely abutting the duodenum. Also noted is GB sludge.
References: Image reproduced from: Arora A, Mukund A, Thapar S, Jain D (2011) Groove pancreatitis: CT, MRI & MRCP findings. 10.1594/EURORAD/CASE.9404.
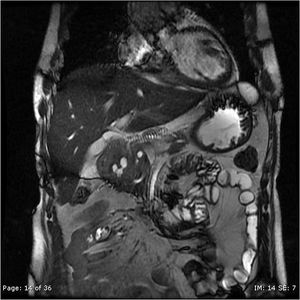
Fig. 22: Cystic lesions in the groove are very well delineated on this coronal scan.
References: Image reproduced from: Arora A, Mukund A, Thapar S, Jain D (2011) Groove pancreatitis: CT, MRI & MRCP findings. 10.1594/EURORAD/CASE.9404.
MRCP can show:
- Widening of the space between the duodenal lumen and the distal CBD and pancreatic ducts.
- A long segmental smooth distal CBD stenosis may be present as opposed to an irregular stricture with shouldering in groove carcinomas.
Patient-1: A 51-year old chronic alcoholic male with characteristic imaging findings on MRCP (Fig-23).
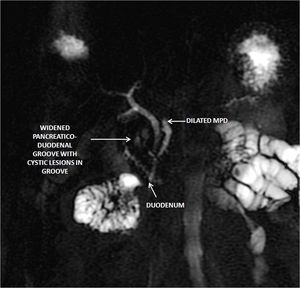
Fig. 23: There is widening of the space between the duodenum and the distal common bile duct and pancreatic ducts with presence of cystic lesions in the pancreatico-duodenal groove.
References: Image reproduced from: Arora A, Mukund A, Thapar S, Jain D (2011) Groove pancreatitis: CT, MRI & MRCP findings. 10.1594/EURORAD/CASE.9404.
Patient-2: A 44-year old chronic alcoholic male with cystic paraduodenal pancreatitis (Fig-24).
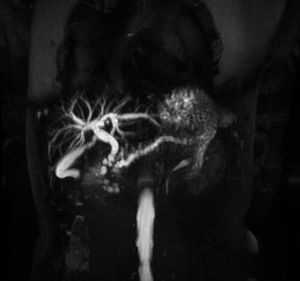
Fig. 24: A 44-year alcoholic with paraduodenal pancreatitis. MRCP reveals dilated pancreatic duct and its side branches. Associated smooth distal CBD stricture with upstream biliary dilatation is present. Multiple paraduodenal cystic lesions are also seen along the pancreatic head.
References: Image reproduced from: Arora A, Mukund A, Thapar S, Jain D (2011) Paraduodenal pancreatitis. 10.1594/EURORAD/CASE.9436.
Endoscopic retrograde cholangiopancreatography (ERCP) can demonstrate:
- Dilatation of the Santorini's duct and its branches,
depicting intraductal stones or protein plugs.
At present,
ERCP is used mainly for endoscopic therapy.
ENDOSCOPIC ULTRASONOGRAPHY (EUS) is considered the preferred imaging modality as it can easily demonstrate:
- Hypoechoic area between the duodenal wall and the pancreatic parenchyma,
- Thickening and narrowing of the duodenal lumen,
- Associated pancreatic calcifications and pseudocysts,
- Stenosis of the CBD and/or dilatation of the pancreatic duct.
- Furthermore,
the diagnosis can be confirmed by EUS-guided fine-needle aspiration in case the imaging findings overlap with groove pancreatic carcinoma.
Patient-1 (Fig-25):
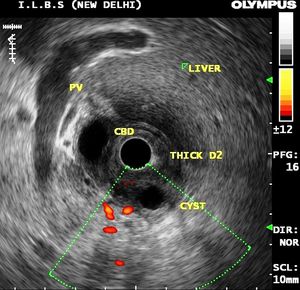
Fig. 25: Endoscopic Ultrasonogram of a 32-year old alcoholic male patient reveals marked submucosal thickening of the duodenum with cystic dystrophy of the duodenal wall.
Patient-2 (Fig: 26-31):
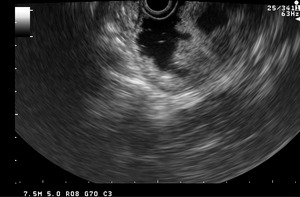
Fig. 26: Endoscopic US reveals preferrential thickening of the medial wall of second part of duodenum. Also, noted is a paraduodenal cystic lesion.
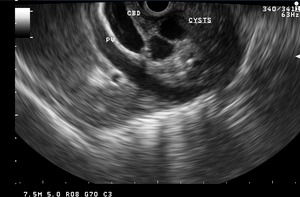
Fig. 27: Multiple paraduodenal cysts are well visualized on this sonographic image.
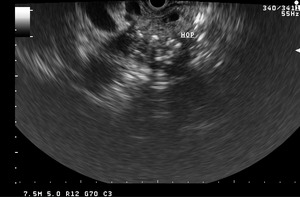
Fig. 28: Associated multiple calcific specks are seen throughout the head of the pancreas.
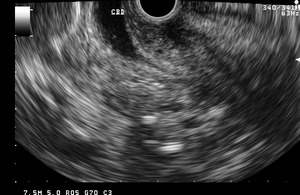
Fig. 29: The disease is associated with smooth distal tapering of the common bile duct (CBD) due to circumferential smooth reactive mual thickening of the distal CBD.
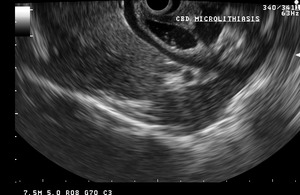
Fig. 30: Multiple microliths are seen in the dilated CBD secondary to biliary stasis.
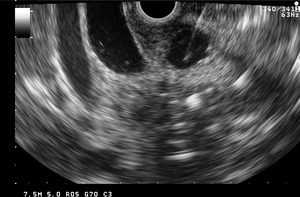
Fig. 31: In doubtful cases, the lesions can be subjected to Endoscopic US guided fine needle aspiration (FNA) to exclude malignancy.
DIFFERENTIAL DIAGNOSES:
- The differential diagnosis of the solid form of paraduodenal pancreatitis includes pancreatic carcinoma,
cholangiocarcinoma,
duodenal carcinoma or acute pancreatitis with phlegmon along the groove.
- Cystic pancreatic lesion and pseudocyst along the groove are important diagnostic considerations for cystic paraduodenal pancreatitis.
- The most important differential diagnosis is pancreatic carcinoma arising in the groove region.
The distinction between fibrous tissue in solid paraduodenal (groove) pancreatitis and scirrhous carcinoma of the pancreatic head invading the groove is often difficult on CT and MR imaging.
- The distinction between the two although difficult,
is crucial as the appropriate management of the two conditions differs significantly.
It is particularly challenging in those cases of pancreatic carcinoma which have a significant fibrous component and therefore may display delayed enhancement similar to that seen with groove pancreatitis.
- The clinical and radiological differential diagnostic clues are enlisted here (Fig-32):
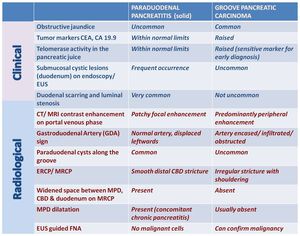
Fig. 32: Clinical and radiological diagnostic clues to differentiate Groove carcinoma from Solid variant of Paraduodenal Pancreatitis
Example-1:
An important differentiating clue is the normal appearance of the peripancreatic vessels (GDA) that are displaced leftward in paraduodenal pancreatitis (Fig-33A),
but they do not show signs of obstruction or encasement.
In contrast,
groove pancreatic carcinoma obstructs or encases the peripancreatic vessels (Fig-33B).
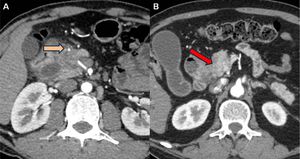
Fig. 33: A) CT in a patient with paraduodenal pancreatitis reveals normal appearing GDA displaced to the left by the fibrous inflammatory mass. (B) In contrast, a axial section in a patient with groove pancreatic carcinoma shows encasemenet and marked attenuation of the GDA.
Example-2:
MRCP is useful for differentiating paraduodenal pancreatitis from groove cancer,
since the former shows a smooth CBD stenosis with frequent involvement of the pancreatic duct due to concomitant chronic pancreatitis.
In contrast,
groove carcinoma shows an irregular distal CBD stricture usually without abnormality of the main pancreatic duct (Fig-34).
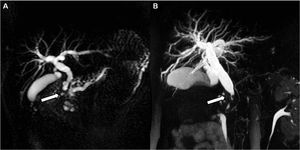
Fig. 34: (A) MRCP in a patient with paraduodenal pancreatitis reveals smooth tapering of the distal bile duct with a dilated MPD and its side branches. (B) In contrast, MRCP in a patient with groove pancreatic carcinoma shows irregular distal CBD stricture with a normal sized MPD.
CLINICAL MANAGEMENT:
Although preoperative recognition is important to avoid unnecessary procedures,
pancreaticoduodenectomy still may be required to relieve obstructive symptoms.
Surgical resection in the form of pancreatico-duodenectomy (using either the Whipple procedure or a pylorus preserving pancreaticoduodenectomy) is the preferred therapeutic option in symptomatic cases,
and in confounding cases to rule out malignancy.
Conservative medical management has been suggested in the acute phase of paraduodenal pancreatitis and endoscopic cyst drainage or stenting has also been attempted in cases with biliary or duodenal obstruction.





























