PARADUODENAL PANCREATITIS
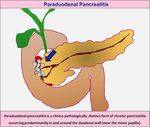
Paraduodenal pancreatitis recently described in 2004 by Adsay and Zamboni refers to a distinct form of chronic pancreatitis occurring predominantly in and around the duodenum along the minor-papilla
(Fig-1).
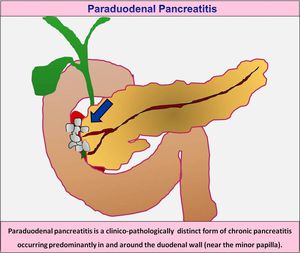
Fig. 1: Paraduodenal pancreatitis is a distinct form of chronic pancreatitis occurring predominantly in and around the duodenal wall (near the minor papilla).
This is an umbrella-term used for various conditions which previously had a variety of names,
including cystic dystrophy of pancreas,
para-duodenal wall cyst,
groove pancreatitis,
pancreatic duodenal hamartoma,
and myoadenomatosis (Fig-2).
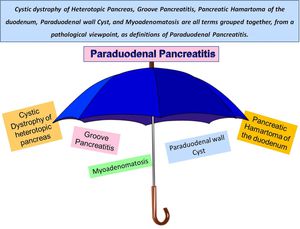
Fig. 2: Previously known under various names such as groove pancreatitis, cystic dystrophy of the duodenal wall, cystic dystrophy of heterotopic pancreas, pancreatic hamartoma of duodenum, myoadenomatosis and myoepithelial hamartoma, the current unified term proposed by Adsay and Zamboni is paraduodenal pancreatitis.
ETIOPATHOGENESIS:
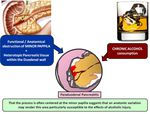
A disturbance of the flow of pancreatic juice through the Santorini duct and presence of heterotopic pancreatic tissue within the duodenal wall has been implicated as important contributory factors.
Pathogenetically,
key factors are anatomical or functional obstruction of the minor papilla and chronic alcohol consumption.
That the process is often centered in the region of minor papilla suggests that an anatomic variation of the ductal system may render this area particularly susceptible to the effects of alcoholic injury (Fig-3).
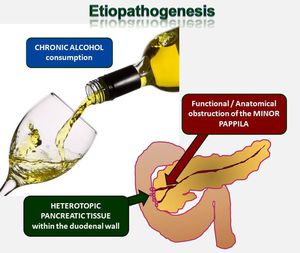
Fig. 3: Paraduodenal pancreatitis is believed to result from focal chronic pancreatitis which affects the minor papilla of the duodenum. Pathogenetically, key factors are anatomical or functional obstruction of the minor papilla, heterotopic pancreatic parenchyma in the duodenum and chronic alcohol consumption.
- One of the proposed theories is that chronic alcohol consumption probably increases the viscosity of the pancreatic juice and leads to Brunner gland hyperplasia,
causing occlusion or dysfunction of the minor papilla.
- Another likely explanation is that when a portion of the pancreas becomes trapped in the duodenal wall during organogenesis,
the resulting intraduodenal pancreas may render an anatomically aberrant minor papilla and duct of Santorini vulnerable to the effects of pancreatitis,
particularly in patients with a history of significant alcohol abuse.
HISTOPATHOLOGICAL HALLMARK:
Gross examination: of the surgical specimen usually shows an abundant whitish firm fibrous mass in the groove area stenosing the duodenum and/ or the distal common bile duct.
This is frequently associated with cystic lesions in the thickened wall of the second portion of duodenum.
Histopathology: these lesions have the following common characteristics (Fig-4):
- Intramural cystic changes within the duodenal wall
- Brunner's gland hyperplasia & dense myoid stromal proliferation
- Spillover of fibrosis into the 'pancreaticoduodenal groove' area
- Because of marked scarring & ill-defined borders of the process it simulates pancreatic or periampullary schirrous tumors
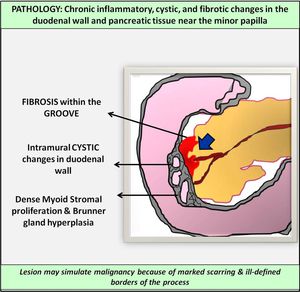
Fig. 4: Adsay and Zamboni were able to show that all these conditions have uniform histological findings: scarring in the plane between the duodenum and pancreatic head, dilated ducts and cysts within the duodenal wall, hyperplasia of the Brunner glands, dense myoid stromal proliferation resembling pancreatic hamartoma, myoadenomatosis or even leiomyoma.
CLINICAL SCENARIO:
Paraduodenal pancreatitis is predominantly seen in male patients,
40-50 years of age,
with a history of chronic alcohol abuse and/or smoking (Fig-5).
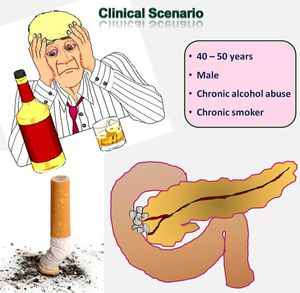
Fig. 5: The classic clinical presentation is upper abdominal pain, vomiting, and weight loss in a 40- to 50-year-old man with a history of chronic alcohol abuse and/ or smoking.
Clinical presentation: Patients usually seek medical attention for complaints pertaining to chronic pancreatitis with post prandial abdominal pain of varying degrees.
Progressive scarring fibrosis and luminal compromise of the duodenum can present with gastric outlet obstruction.
Obstructive jaundice due to distal common bile duct stricture or compression has also been reported.
Laboratory parameters: Blood tests often show a slight elevation of serum pancreatic enzymes and occasionally of liver function tests.
Tumor markers,
carcinoembryonic antigens and carbohydrate antigen 19-9,
are rarely elevated.
MORPHOLOGICAL/ IMAGING CLASSIFICATION:
Paraduodenal pancreatitis can be classified morphologically as either a solid-mass occurring predominantly in and around the minor papilla (solid variant) or cystic lesions (cystic variant) within the pancreaticoduodenal groove or intramurally within the duodenal wall (Fig-6).
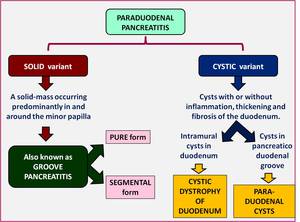
Fig. 6: Adsay and Zamboni recognized two types of Paraduodenal Pancreatitis: one characterized by cystic changes and the other characterized by solid lesions.
The SOLID variant is characterised by the presence of a sheet-like mass corresponding to the fibrous-scar predominantly located in the pancreaticoduodenal groove.
- This variant of paraduodenal pancreatitis has been traditionally described as ‘Groove Pancreatitis’ in literature.
- Groove pancreatitis was first described in 1973 by Becker and Bauchspeichel,
however,
the term ‘Groove Pancreatitis’ was coined by Stolte et al.
in 1982.
- Becker and Mischke classified it into 2 subtypes: pure and segmental form.
- In the ‘pure’ form only the pancreatico-duodenal groove is involved,
with sparing of the pancreatic parenchyma; while in its ‘segmental’form the head of the pancreas and the main pancreatic duct get involved.
CYSTIC variant of paraduodenal pancreatitis is characterised by the presence of cysts in the duodenal wall (cystic dystrophy of the duodenum) and cysts within the pancreatico-duodenal groove (paraduodenal cysts) with or without inflammation,
thickening and fibrosis of the duodenum.
- Cystic dystrophy of duodenum is characterised by the development of true cysts within the heterotopic pancreas located in the gut wall.
- The cysts are usually lined with cuboid epithelium similar to those lining the pancreatic ducts.
- This heterotopic pancreatic tissue is most frequently seen along the duodenum in close vicinity to the pancreas.
It is still unclear and contentious whether groove pancreatitis and cystic dystrophy are distinct entities or part of the same spectrum.
Hence,
a broad category categorized ‘Paraduodenal Pancreatitis’ has been proposed to include groove pancreatitis (solid subtype),
cystic dystrophy of the duodenum and paraduodenal cysts (cystic subtype).