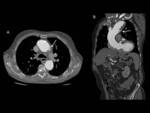
Endoleaks are diagnosed when contrast material fills the excluded aneurysmal sac.
Imaging studies should be able to define the endoleak and its cause. The imaging method of choice to detect,
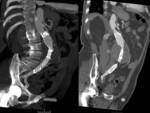
classify and follow-up of endoleaks after endovascular aortic aneurysm repair is multidetector CT angiography with multiplanar reconstructions (Fig 3) [5,
6,
7].
Digital subtraction angiography (DSA) is not a first line technique in diagnosing endoleaks but could be used for characterization of type I leaks or in problematic cases,
as well as in the treatment of some types of leaks.
Due to the variable flow rates of the endoleaks,
they can be better depicted at variable times after IV contrast material injection.
To optimize the detection,
the classical protocol in a CT of a single line of detectors included a nonenhanced CT acquisition and then another two series acquired during the arterial and the venous phases of enhancement [1].
However,
due to periodic CT studies,
these patients will be exposed to a cumulative amount of ionizing radiation,
and in an attempt to decrease this exposure and with the current use of multidetector CT; many authors consider the option of removing parts of this multiphasic CT study.
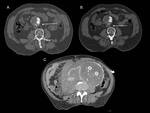
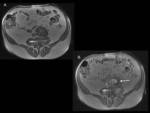
The nonenhanced scan helps to distinguish endoleaks from calcifications [6,
8] and from residual endosac contrast material from the stent placement procedure,
when early CT follow up is obtained (Fig 4) [5].
This basal acquisition is necessary only in the first CT examination [8].
Images obtained during the arterial phase (using bolus track technique) are useful to detect high flow leaks and to plan embolization procedures in order to treat endoleaks [2,
7].
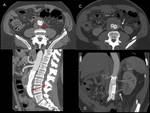
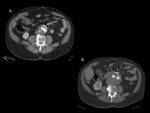
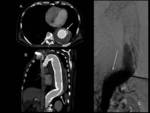
The delayed phase (60 seconds after starting contrast administration) depicts some endoleaks with higher sensitivity than the arterial phase [2] (Fig 5).
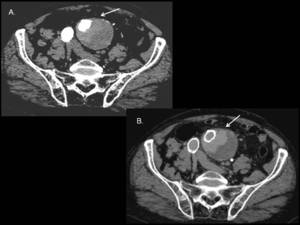
Fig. 5: Importance of a dual CT acquisition (arterial and delayed phases) in the evaluation of AAA stent-graft repair. (A) On an axial arterial phase CT image an endoleak is barely visible (arrow). (B) On an axial venous phase image, the leak (arrow) becomes much more evident.
Some investigators have proposed to eliminate the arterial phase acquisition as part of the routine follow-up,
taking it just at the first CT examination after the procedure or during the follow up in patients with aneurismal sac growth in which leaks are not detectable in the venous phase study or in those with symptoms suggesting femoral arteriovenous fistulas or inguinal pseudoaneurysm [1].
We consider that the biphasic enhanced CT study provides important information to detect and classify endoleaks and in some cases also to drive the treatment.
For these reasons we acquire the two phases during follow-up.
Endoleak Classification:
-Type I endoleaks result from a separation between the endograft and the arterial wall,
allowing high pressure aortic flow entering into the aneurismal sac [9].
This separation may occur at either the proximal (Ia) or the distal (Ib)attachment sites (Fig 6,
7).
This type of leak is the most frequently reported in the repaired thoracic aorta aneurysms (Fig 8).
In cases where an aorto-uni-iliac stent-graft has been deployed,
in conjunction with a femoral-femoral bypass graft,
an occluder device is placed in the contralateral common iliac artery to prevent back-filling of the aortic aneurysm.
If contrast comes from the excluded common iliac artery a Ic leak is diagnosed.
Type I endoleaks have been identified as a clear risk factor for late aneurysm rupture and they should be repaired when detected.
The endovascular treatment of type Ia and Ib endoleaks is usually done with a balloon expandable stent.
When this procedure is unsuccessful,
the insertion of an overlapping stent graft in the nonadherent portion of the original stent graft can be performed [2,
10] (Fig 7,
9).
If these procedures fails,
open surgery may be needed,
as well as in type Ia abdominal endoleaks due to the placing a proximal overlapping stent will usually cover the renal arteries [10].
The treatment of type Ic endoleaks requires completion of the intended thrombosis of the common iliac artery [5].
Embolization of type I endoleaks has also been described [2,
9,
10].
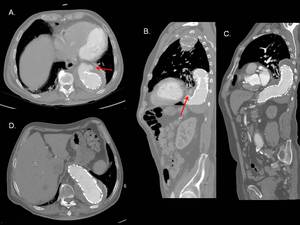
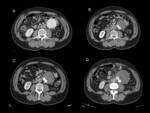
Fig. 9: Type Ib endoleak in thoracoabdominal aorta. Axial (A)and sagittal MIP (B) images from contrast-enhanced CT showing a type Ib endoleak (arrows) caused by ineffective sealing between the stent-graft and the aortic wall at the distal attachment. (C and D) Images at the same planes showing sucessful endoleak sealing after the endovascular insertion of a stent-graft extension.
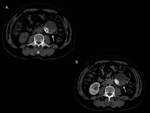
-Type II endoleaks result from retrograde blood flow from aortic branches,
such as inferior mesenteric or lumbar arteries [9] in the abdominal aorta,
whereas in thoracic endografts,
type II endoleaks depend on the bronchial and intercostal arteries.
Usually,
these leaks are located in the aneurysmal sac periphery without contact with the stent [2] (Fig 10,
11) and they are the most frequent type in abdominal aorta presenting in up to 25% of endovascular repaired aneurisms [10].
Increased blood flow into the aneurysm can cause enlargement of the sac and increasing pressure could cause rupture [10],
although this does not occur in all cases.
If a type II endoleak is present without an associated increase in size of the aneurysm sac intervention is not needed and this endoleak can spontaneously thrombose [2,
10].
When treatment is required,
endoleaks type II reparation could be done via a transarterial or translumbar approach [10].
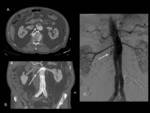
With the transarterial technique,
a catheter is placed in the superior mesenteric artery or the internal iliac artery.
Microcatheters are then manipulated through collateral vessels into the vessel communicating with the aneurysmal sac,
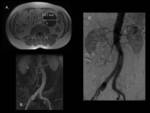
usually the inferior mesenteric artery or a lumbar artery [2,
9] (Fig 12).
However,
success rates of the single-vessel transarterial approach is poor because when one artery supplying the endoleak is embolized,
other communicating vessels can still supply it.
For this reason,
a modification of the transarterial method is to place the microcatheter into the aneurysm sac,
embolize with coils the sac itself and then embolize the feeding vessels when the microcatheter is withdrawn,
trying to thrombose the nidus or sac of the endoleak as well as the major feeding artery [2,
10].
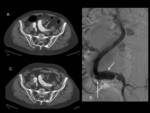
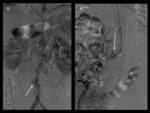
In the translumbar approach,
the aorta can be punctured under CT or fluoroscopic guidance (Fig 13). The proper positioning of the catheter within the endoleak is usually signaled by a free return of blood and opacification of lumbar arteries or the inferior mesenteric artery on manual injection of contrast material.
The most commonly used embolic agents are stainless steel or platinum coils [2,
10].
The use of several other agents has been successfully reported with translumbar treatment of type II endoleaks,
including Onyx,
Ethibloc,
Thrombin,
and Cyano-acrylate [5].
In our institution we use thrombin injected directly into the sac.
Translumbar embolization has been shown to be more durable than single-vessel transarterial endoleak embolization [10].
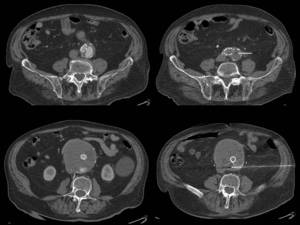
Fig. 13: Translumbar fine-needle thrombin embolization of type II leak. Pacient with two type II endoleaks with progressive growth of the aneurysmal sac (left). Translumabr approach under CT guidance for thrombin embolization of the two leaks (right).
-Type III endoleaks are usually caused by a defect within the graft material or are due to structural failures causing separation between the components or inadequate overlap (Fig 14).
These leaks are believed to be the most dangerous due to the fast repressurization of the aneurysm sac [2].
Repair of type III endoleaks involves placement of a new stent-graft component across the defect or junctional separation [10].
-Type IV endoleaks are uncommon with today's grafts but appear as aneurysm blush during immediate postdeployment angiography,
caused by graft wall porosity while the patients are fully anticoagulated.
These leaks are self-limited,
requiring no treatment and resolving spontaneously once the patient's coagulation status is normalized [2,
10].
-Type V endoleaks as known as endotension (increasing pressure in the sac) are classified as an enlarging aneurysm sac (increasing more than 5 mm in diameter) without a visible endoleak.
They might be due to undiagnosed endoleak,
presumably with very slow flow and suboptimal imaging [5].
Some authors suggest that the reason behind endotension is intrasaccular hygroma,
which causes pressure and sac enlargement.
Other believes that endotension may also be related to the graft material allowing transudation of fluid throught the graft fabric.
Some others suggest that the graft infection may cause endotension [11].
These patients can require conversion to open surgery with removal of the stent-graft and placement bypass [10].
If endotension is identified with CT,
it is recommended to confirm the absence of endoleaks by other techniques (Fig 15,
16,
17) [11].
Other imaging techniques in classifying endoleaks and therapy:
MR angiography: Gadolinium-enhanced MR angiography is capable of depicting endoleaks,
but its performance is dependent on the stent-graft composition [2] (Fig 18).
Stents composed of nitinol are generally more suitable for MR imaging; elgiloy stents can obscure the lumen,
and stainless steel stents cause extensive artifacts that render the study nondiagnostic [2].
New blood pool MR contrast agents may improve the detection of endoleaks with slow flow rates that are occult on CT angiograms or catheter angiography [2,
5,
12],
although the clinical importance of these endoleaks is unknown.
MR angiographys main current use is in patients with allergy to iodinated contrast material.
US and contrast agent-enhanced US: Color Doppler US is the noninvasive alternative for follow-up,
but the reported sensitivity in endoleak detection ranges from 42% to 97% [13].
Second-generation of ultrasonography contrast agents consist of gas microbubbles (perfluorcarbon or sulfur hexafluoride) encapsulated by a phospholipid shell that have a nonlinear behavior when interrogated with a low-mechanical-index ultrasound beam,
resulting in the emission of harmonics that can be detected with specific contrast software.
Endoleaks are diagnosed when identifying a hyperechogenic flowing region that was absent on the baseline unenhanced images outside the endograft lumen but within the aneurysm sac [13].
Limitations of US are related to incomplete examinations due to patient’s obesity or bowel gas obscuring parts of the aorta and the endograft,
and the variability of the technique.
The role of contrast agent-enhanced US is not to be a substitute for CT angiography but to be integrated as a complementary tool in endovascular abdominal aneurysm repair follow-up.
In patients with stable or decreasing aneurysmal sac a year after procedure,
contrast agent-enhanced US could replace CT angiography in the follow-up [13].
Digital subtraction angiography (DSA): DSA could characterize some types of endoleaks and is useful for their endovascular treatment.
Angiographic examination should include a global map contrast injection into the aorta up to the level of renal arteries and inside stent-graft.
Adquisition time must be long enough to allow filling of type II endoleaks (Fig 19) [5].
Anteroposterior and lateral aortograms should be obtained at the proximal attachment site with a flush catheter to evaluate for type I endoleaks.
The catheter is then repositioned into the stent-graft just above the flow divider for another aortogram for evaluating type III endoleaks or distal type I endoleaks [2]. For type II endoleaks,
the superior mesenteric artery and each internal iliac artery must be injected in order for detect retrograde filling of the aneurismal sac from the inferior mesenteric artery and/or the ilio-lumbar arteries [5].