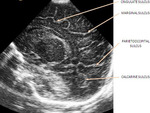
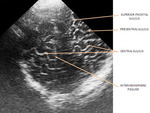
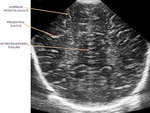
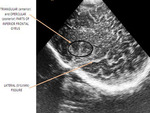
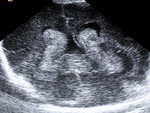
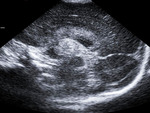
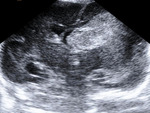
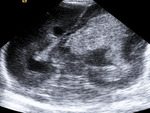
We reviewed the ultrasound (US) imaging studies obtained in a group of premature and term neonates.
The studies were performed with high frequency linear array transducer (10 mHz),
by direct contact with the anterior fontanelle,
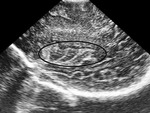
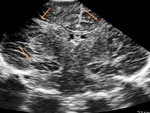
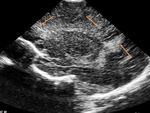
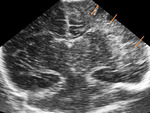
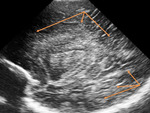
using a standardized protocol with sagittal and coronal images of the whole brain.
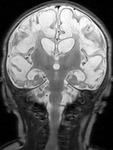
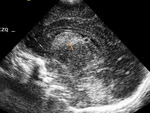
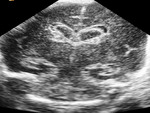
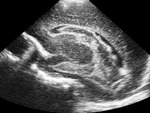
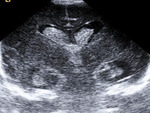
We evaluated the anatomy in premature and term infants (Fig. 1 - Fig. 5).
Among the group of patients with pathology we classified brain injuries according to the two different groups:
I-Hypoxic-ischemic brain injury in the premature infants (Fig. 6 - Fig. 9):
- White matter injury
- Germinal matrix hemorrhage
- Intraventricular hemorrhage
- Periventricular (PV) hemorrhage
- Infarction
- Posterior fossa injury
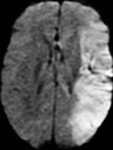
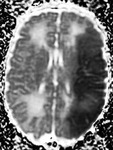
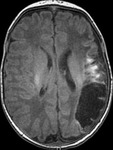
II-Hypoxic-ischemic brain injury in the term infants (Fig. 10 - Fig. 15):
- Cerebral cortex injury
- Boundary zone injury
- Basal ganglia injury.
The hypoxic-ischemic brain injury is due to decreased cerebral blood flow or systemic hypoxemia [2].
It can have various stages.
Mild hypoxia affects the boundary regions of vascular territories (watershed lesions).
Severe hypoxia leads to a deeper involvement: the basal ganglia and the perirolandic cortex [2].
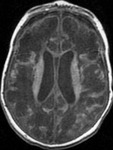
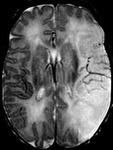
We analyzed the findings and correlated them with follow up MRI images.
Indications for routine US screening:
- birth weight less than 1500 g
- gestational age less than 35 weeks
- suspicion of intracranial hemorrhage or hypoxic-ischemic brain injury [2,
3].
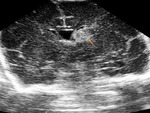
US of the neonatal brain:
-efficient tool for identifying hypoxic-ischemic injuries (Table 1) and hemorrhagic lesions (Table 2)
-the normal PV white matter is less bright than the choroid plexus
-in the ischemic lesions and also in the hemorrhagic ones it shows increase of echogenicity
-depicts between 28% and 80% of histologically demonstrated periventricular leukomalacia (PVL)
-difficulty in depiction of mild homogeneous densities (that are associated with transient signal anomalies or even normal findings on follow-up MRI and with no effect on the patient outcome)
-irregular and inhomogeneous US densities are associated with more severe MRI findings [1-3].
MRI utility (Table 3):
-in 2/3 of the infants with hypoxic-ischemic injuries provides additional information over US
-PV or cortico-subcortical areas of T2 hyperintensities in ischemic lesions
-better characterization of the cysts (showing more numerous or more extensive cysts than the US)
-better depiction of hemorrhagic lesions: increased signal intensity on T1WI and decreased on T2WI (in areas not normally myelinated at this age,
as myelination gives similar intensities)
-important role in the group with heterogeneous echodensities (superior evaluation of the hypoxic-ischemic damage)
-extensive hemorrhages and intensities on MRI can predict the development of cystic leukomalacia
-when no dilated ventricles,
MRI could relieve small intraventricular hemorrhages not detected by US
-for subarachnoid bleeding,
MRI or CT is preferable to US [2].
Periventricular leukomalacia - stages:
-acute phase: increased echogenicity of the PV white matter,
appearing within 24 to 48 hours after a hypoxic-ischemic event (Fig. 6, Fig. 7)
-late subacute phase: 2-4 weeks,
cysts can arise in the hyperechogenic areas
-chronic phase: the cysts resolve,
with development of ventricular enlargement [1,
2].
Hemorrhagic lesions:
-in 1/3-1/2 of all infants of less than 35 weeks gestational age or weighing less than 1,
500 g
-in 2/3 of the infants which require assisted ventilation
-the spectrum of the hemorrhagic lesions: petechial,
focal and extensive (at least three lobes involved in the last one); the higher the grade,
the greater the tendency to cystic degeneration
-the frontoparietooccipital PV regions are the more frequently involved,
but also frequently found in the subcortical white matter [3-5]
-90% of hemorrhages occur in the germinal matrix overlying the caudate head and 10% in the vicinity of the body of the caudate nucleus
-highly echogenic region of convex borders
-best seen on coronal sections: inferolateral to the lateral ventricle,
just posterior to the foramen of Monro
-on sagittal scans: anteriorly and laterally to the choroid plexus (also echogenic); an useful anatomic landmark - the thalmocaudate notch
-tendency to develop cystic spaces [4,
5]
-complicates about 80% of subependymal hemorrhages
-echogenic intraventricular densities
-best diagnostic clue: search the frontal and occipital horns on sagittal scans
-anterior to the caudate nucleus in the frontal horns (be aware that the choroid plexus shouldn’t be present anterior to the foramen of Monro)
-in preterm neonates: extension pattern from the thalamocaudate notch (germinal matrix hemorrhage) within the lateral ventricle to its occipital horns
-in term infants: intraventricular bleeding is rare; the choroid plexus is the major source of the intraventricular hemorrhage [4,
5]
- Other hemorrhage locations
-cerebellar,
subarachnoid and small extraaxial hematomas
-the incidence of intracerebellar hemorrhage is up to 25% of preterm neonates less than 34 weeks of gestational age
-the cerebellar vermis,
inherently echogenic,
shouldn’t be confused with blood
-they are unreliably visualized by US; consequently,
the study should be completed by MRI or CT [4,
5].
Complications:
- Of hypoxic-ischemic lesions
-cystic transformation,
multicystic leukomalacia
-basal ganglia necrosis
-PVL: white matter loss,
ventricular dilatation,
abnormal T2 hyperintensity in the periventricular region [2]
-hydrocephalus,
early or late (up to 1/3 of the hemorrhages,
resulting from obstruction at any level – foramen of Monro,
aqueduct of Sylvius or foramen of IV ventricle – or by inflammatory mechanisms) [4,
5]
-significant neurologic sequelae,
some of long-term morbidity or even incapacitating
-mortality (up to 1/3 of the hemorrhages)
-generally proportional to the extension [2,
5].