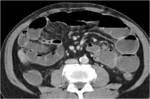
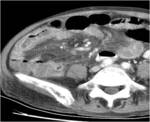
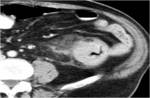
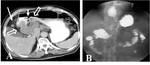
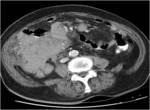
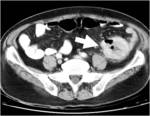
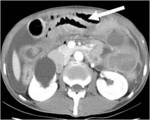
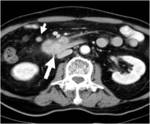
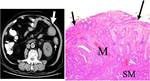
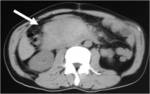
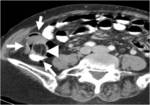
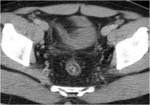
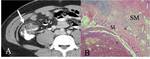
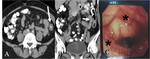
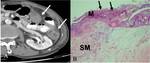
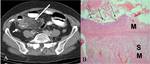
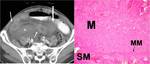
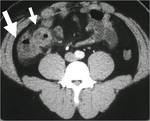
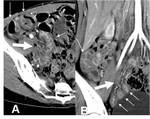
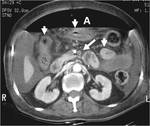
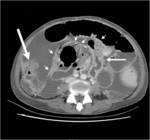
CT Image of Normal Bowel Wall
When optimally distended,
normal bowel wall is barely visible at CT.
The thickness of small bowel and colon measures not greater than 1.5 mm and 2-3 mm,
respectively.
When the thickness of distended bowel is greater than 3 mm,
bowel wall thickening is defined.
After intravenous contrast material administration,
the visible wall usually manifests modest enhancement at CT.
The mean enhancement measures about 118-120 HU and 107-111 HU at arterial and portal phases,
respectively.
Normal perienteric fat should appear as clear as subcutaneous or retroperitoneal fat,
without evidence of stranding,
haziness or fluid (Fig 1).
Symmetry and Degree of Wall Thickness
The majority of gastrointestinal diseases present wall thickening of involved segment.
In general,
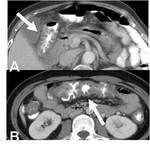
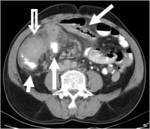
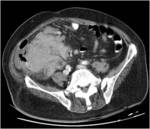
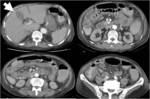
non-neoplastic diseased bowel wall is concentrically and smoothly thickened with long transition in most cases (Fig 2-5),
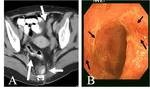
while neoplastic bowel wall is often either asymmetrically and irregularly thickened or polypoid mass-like with abrupt shouldering (Fig 6-8).
Nevertheless,
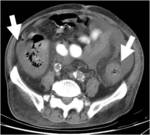
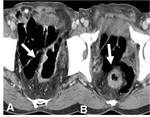
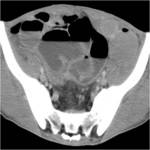
some specific inflammatory diseases such as acute appendicitis and colonic diverticulitis may also cause asymmetric focal wall thickening of contiguous colon by extending from the inflammation origin (ie.
vermiform appendix or diverticulum).
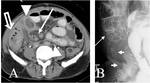
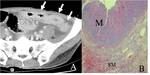
Such asymmetrically thickened juxta-lesional colon wall may indent its lumen,
thereby creating so-called “arrowhead sign” (Fig 9,
10).
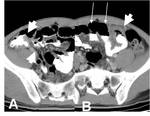
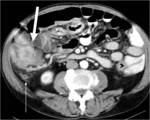
Smooth mucosal surface of bowel tumor may suggest submucosal,
intramural or subserosal neogrowth such as gastrointestinal stromal tumors (GISTs) (Fig 11).
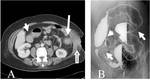
The degree of wall thickness varies markedly among variable non-neoplastic and neoplastic bowel entities.
In non-neoplastic bowel disorders,
pseudomembranous colitis may present greatest wall thickness as compared to other inflammation of bowels (eg.
neutropenic enterocolitis ,
viral or bacterial enterocolitis,
diverticulitis and inflammatory bowel disease),
ischemic bowel disease,
and graft-versus-host disease (GVHD) (Fig 12,
13).
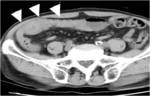
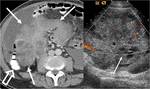
Contrast material trapped between polypoid thickened haustral folds presents “accordion sign” once thought specific for pseudomembranous colitis but also reported in other severe colitis (Fig 14).
Crohn disease shows thicker wall than does ulcerative colitis in inflammatory bowel disease (Fig 15,
16).
Intramural hemorrhage arising from trauma,
anticoagulation therapy or bleeding diathesis which sometimes need differentiation from bowel ischemia with subseroal hemorrhage at CT often has higher degree of wall thickening.
The size of neoplastic wall thickness varies from mild thickening,
small nodule to bulky or exophytic growing mass (Fig 17-19).
Obstructed colon carcinoma is not infrequently associated with proximal ischemic colitis,
which can be distinguished from tumoral segment by concentric,
smooth,
mild thickness (Fig 20).
Variable wall thickness of intestinal tuberculosis (TB) depends on the ulcerative,
hypertrophic or sclerotic forms.
Hypertrophic form of TB may manifest exophytic masses around the ulcerated ileocecal lumen,
mimicking lymphoma or carcinoma (Fig 21-23).
However,
lymphoma may be associated with aneurysmal luminal dilation,
and cecal carcinoma rarely extends beyond ileocecal valve.
Wall Attenuations
On precontrast CT,
although wall attenuation mostly contributes little for differential diagnosis,
some specific attenuation abnormalities may give hints.
High-attenuation wall (grater than 50-60 HU) usually denotes intramural hemorrhage (Fig 24,
25).
Fat-attenuation mass lesion of wall represents intestinal lipoma (Fig 26).
Bowel wall “fat halo sign” (wall stratification with a fat layer less than –10 HU) may be demonstrated in subacute or chronic inflammatory bowel disease (more often in ulcerative colitis [61%] vs.
Crohn disease [8%]) (Fig 27),
but also may represent a normal finding related to obesity.
Some rare neogrowths of bowel such as carcinoids and GISTs may occasionally calcify.
Gas attenuation within bowel wall (pneumatosis intestinalis) is reported a specific sign for diagnosis of bowel ischemia (Fig 28).
However,
it may also be seen in overdistension,
ulcerative inflammatory bowels,
long-term steroid use or post-endoscopy condition.
Distribution and Length of Extent
Acute appendicitis and diverticulitis can be specifically diagnosed at CT by the evidence of vermiform appendix- or diverticulum-centered inflammatory thickening (Fig 29,
30).
Many other gastrointestinal diseases present a broad spectrum of intestinal distribution and length of disease involvement.
Pseudomembranous colitis tends to involve whole course colon,
though segmentally on occasion (Fig 31,
32).
Ischemic colitis tends to involve vascular territory (Fig 33).
Exclusive involvement of ileum and right colon is more frequent with Crohn disease,
TB and neutropenic enterocolitis.
Shock bowel,
reversible transient ischemia due to prolonged hypoperfusion,
typically demonstrates diffuse small bowel wall thickening,
but often sparing the colon.
Cirrhosis-related edematous bowel wall thickening predominantly involves right colon when caused by portal hypertension,
or diffusely involves both colon and small bowel when caused by hypoalbuminemia (Fig 34).
Longer length of bowel involvement favors inflammatory or ischemic condition rather than neoplasm or intramural hemorrhage.
Nevertheless,
on the contrary to short length of adenocarcinoma,
lymphoma can extend for longer bowel segment (Fig 35).
Discontinuity of lesional involvement with intervening normal segments (skip lesions) is the hallmark of Crohn disease (Fig 36),
yet it can also occur in gastrointestinal graft-versus-host disease,
mesenteric vasculitis,
TB,
lymphoma or metastasis.
Contrast-Enhancing Pattern
- Homogeneous and heterogeneous enhancing patterns
Although both homogeneous and heterogeneous patterns of contrast enhancement of bowel wall can occur in either neoplastic or non-neoplastic gastrointestinal diseases,
the former pattern is more frequent with non-neoplastic entities.
The exception is ulcerative colitis,
which presents inhomogeneous enhancement in 70% of cases (Fig 37),
while Crohn disease classically enhances homogeneously (Fig 38).
Malignant intestinal tumors are inclined to enhance more heterogeneously probably due to necrosis,
cystic degeneration or hemorrhage; none the less,
lymphoma enhances more homogeneously (Fig 39,
40).
“Target sign” or “double halo sign” of wall enhancing pattern demonstrates alternating concentric high- and low-attenuated layers in bowel wall.
This pattern consists of inner enhancing layer– mucosa,
lamina propria and hypertrophic muscularis mucosa,
intermediate hypodense layer– submucosal edema,
inflammation or fat,
and outer enhancing layer– muscularis propria & serosa.
Target sign is manifested exclusively in non-neoplastic intestinal diseases,
either acute or chronic,
including Crohn disease,
ulcerative colitis,
infectious enterocolitis,
pseudomembranous colitis,
neutropenic enterocolitis,
bowel ischemia,
radiation damage,
GVHD,
Henoch-Schonlein purpura,
bowel edema,
and even intramural hemorrhage (Fig 41- 44).
Conspicuous mucosal enhancement is most evident with shock bowel,
GVHD and visceral angioedema (Fig 45).
- Lack and delayed enhancing pattern
Lack of mural contrast enhancement is specific for transmural bowel infarct (Fig 46).
Delayed enhancing pattern of wall during bi-phasic dynamic CT scan associated with serrated thickened beaked transition zone has been reported as a sign for strangulation.
Perienteric Change
CT can superbly detect perienteric change related to bowel diseases.
The perienteric change accompanying inflammatory or ischemic events is often non-specific but conspicuous,
including mesenteric fat stranding,
edema or fluid.
Such perienteric change may help little for differential diagnosis of underlying disease,
but signifies the severity of disease activity.
Thickened bowel segment associated with mesenteric fibrofatty proliferation,
perienteric fistula (enterocutaneous,
enteroenteric,
enterovesical or enterovaginal),
sinus tract or abscess frequently denotes Crohn disease (Fig 47,
48).
Peri-lesional phlegmon or abscess is also common seen with ruptured appendicitis or diverticulitis.
Perienteric mesenteric infiltration may also be associated with malignant tumor,
implying either direct tumor invasion,
desmoplastic reaction or a complication of perforation.
Beaded,
nodular morphology of mesenteric vascular engorgement or “comb sign” is reported suggestive of mesenteric vasculitis (Fig 49),
but similar finding may also be shown with Crohn disease and GVHD in the literature.
Visualization of mesenteric arterial or venous occlusion adds credit to thickened bowel wall for diagnosis of ischemic bowel disease (Fig 50).
Small mesenteric lymph nodes are not uncommon in inflammatory diseases.
Necrotic or calcified mesenteric lymph nodes may relate to intestinal TB.
Mesenteric or retroperitoneal bulky lymphadenopathy is characteristically associated with malignant tumors.
Presence of parietal peritoneal nodular or plaque-like thickening,
enhancement,
mesenteric or omental masses and loculated ascites in association with thickened bowel wall denotes peritoneal carcinomatosis (Fig 51).
Bloody ascites is frequently associated with intramural hemorrhage or bowel ischemia.
Demonstration of extraluminal gas or extravasation of enteric contrast medium or feces is the evidence of perforation.