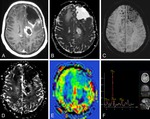
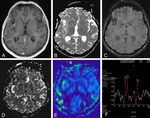
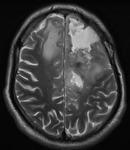
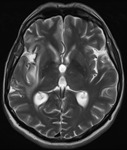
Follow-up brain MRI examinations were performed in 27 patients (19 men,
8 women; mean age 54.2 years) after surgery and/or radio and chemotherapy of gliomas.
From all of them,
21 patients had histological conformed glioblastoma multiforme (WHO grade IV),
5 had anaplastic astrocytomas (WHO grade III),
and 1 was with diffuse astrocytoma (WHO grade II).
All MR images were performed with a 3T system (Skyra,
Siemens,
Germany).
Imaging protocols included the following MR sequences: axial TSE T2 weighted imaging,
axial SE T1 weighted imaging,
coronal fluid-attenuated inversion recovery (FLAIR),
DWI,
ASL,
SWI,
dynamic susceptibility contrast-enhanced (DSC) MR perfusion imaging,
and contrast-enhanced axial,
coronal and sagital T1 weighted imaging.
The DSC MR perfusion imaging was performed during the intravenously administration of a standard dose of gadopentetate dimeglumine (Magnevist,
Schering,
Germany; 0.1 mmol per kilogram of body weight) with an MR-compatible power injector (Spectris,
Medrad,
Pittsburgh,
PA) through a 20-gauge angiocatheter at a rate of 5 mL/s.
The bolus of contrast material was followed by a 20 ml bolus of saline administered at the same injection rate.
Image processing was performed using commercially available software (Syngo Via,
Siemens,
Germany).
The DSC perfusion MR images were used in the production of cerebral blood volume (CBV) maps.
A single region of interest (ROI) was manually drawn around the entire contrast-enhanced region and contralateral normal-appearing white matter to standardize the cerebral hemodynamic measurements.
ASL represented with cerebral blood flow (CBF) map was analyzed visually.
The intralesion susceptibility signal (ILSS) seen on SWI was defined either as low-signal dot-like or tubular structures within a lesion.
It was classified as grade 0 (no ILSS),
grade I (1–5 dot-like or tubular ILSS),
grade II (6–10 dot-like or tubular ILSS) or grade III (>11 dot-like or tubular ILSS).
The gradation was based on the maximum of the ILSS on the selected imaging slice.
Based on multi-voxel 1H-MRS,
choline (Cho) to creatine (Cr) ratio was quantified.
Apparent diffusion coefficient (ADC) values were calculated by manually drawn ROI within the solid portion of the lesion.
All lesions were confirmed pathologically or by clinical-radiological follow-up after 3-months.
Three MRI protocols were reviewed by two neuroradiologists: (1) non-contrast,
including ASL,
SWI,
1H-MRS and DWI,
(2) contrast with DSC perfusion imaging,
and (3) combined contrast and non-contrast protocols.
The lesions were classified as posttreatment effects or recurrent tumors,
of the same grade or with progression to a higher grade.
Non-contrast protocol quantitative data included CBF,
degree of ILSS,
Cho/Cr ratio and ADC values within the region of interest.