Type:
Educational Exhibit
Keywords:
Abdomen, Liver, Ultrasound physics, Elastography, Ultrasound, Technology assessment, Education, Cirrhosis, Pathology
Authors:
O. S. Jaffer, G. Yusuf, P. Sidhu; London/UK
DOI:
10.1594/ecr2013/C-1984
Background
Currently,
liver biopsy to obtain histology is considered the ‘gold standard’ to quantify liver fibrosis. However,
biopsy is as an invasive procedure with recognised risks,
limitations and monetary costs.
Complications may range from pain,
to more unusual but serious complications such as intraperitoneal haemorrhage.
Furthermore,
there is potential for sampling errors given the heterogenous nature of fibrosis distribution and the small area sampled by a liver biopsy (approximately 1/50000th of total liver volume).
Studies have shown these factors may lead to misdiagnosis in 25% and discordance in fibrosis staging in up 33% 1. Recognition of these constraints has led to a search for a safe,
reliable and inexpensive non-invasive technique to quantify liver fibrosis.
While Imaging offers alternatives including MRI and CT for this purpose,
it is ultrasound elastography that has gained the widest interest.
Favourable qualities of ultrasound quantification include: lack of ionising radiation; comparatively,
the examinations are relatively quick and inexpensive; and ease of accessibility.
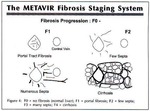
When performing liver biopsy,
two clinically relevant critical endpoints are sought: Firstly,
the detection of significant fibrosis (F2/ I3) (Image 1 and 2) which is an indication for the commencement of antiviral treatment in chronic hepatitis B and C; and secondly,
the presence of severe fibrosis (F3/ I5) or cirrhosis (F4/I6) which prompts the monitoring of associated complications such as portal hypertension and hepatocellular carcinoma.