I-INTRODUCTION AND EPIDEMIOLOGY
Acute ischemic stroke is caused by disruption of blood flow to one or more regions of the brain by vessel occlusion in 80% of cases (1) or by acute hemorrhage from diseased vessels in 20% of cases.
According to the “Instituto Nacional de Estadística” (INE) of Spain,
cerebrovascular disease was the second most common cause of death in the Spanish population in the year 2006 (the latest year statistics were available at the time of writing),
the first cause of death in the Spanish female population and the most important cause of permanent disability in Spanish society (2,3).
In Spain,
approximately 120,000 cases of acute ischemic stroke occur per year,
and approximately 795,000 cases per year are recorded in the United Stated of America (US) (4).
In the year 2003,
114,498 cases of acute stroke where recorded in Spain,
with and incidence of 268 per 100,000 inhabitants.
In the year 2006,
according to the INE,
there were 114,807 inpatient hospitalizations secondary to acute strokes and 1,288,010 hospital visits for the same reason,
this last figure being higher than the cases of ischemic heart disease (2).
In terms of economic cost,
in the US,
it is estimated to be around 503,000 million dollars in the year 2010,
which includes both direct costs and indirect costs such as wages lost (4).
In Spain,
the cost oscillates between 801.7 and 1,243.0 million Euros (2).
Using the Markov Model,
it has been determined that aggressive treatment of acute ischemic strokes reduces the cost of rehabilitation by 1.4 million per 1,000 patients and the cost of general treatment by 4.8 million per 1,000 patients (5).
Epidemiologically,
the “Number Needed to Treat” (NNT) is a measure of treatment efficacy,
and indicates the number of patients that need to be treated per favorable outcome.
It has been estimated that in the US the NNT increases from 2 in the first 90 minutes after the onset of an acute ischemic stroke to 7 in the first three hours and to 14 in the first 4.5 hours (6).
Thus,
according to the “Sociedad Española de Neurología” (SEN),
early treatment of acute ischemic stroke could save up to 6,000 lives from the approximately 40,000 that die each year in Spain from this disease (7).
In the US,
approximately 25-29% of patients reach an emergency department (ED) within the first three hours of symptom onset and 33-66% do so within 6 hours (8,9),
without having any reliable epidemiologic numbers available for Spain.
According to the document "Estrategias en Ictus del Sistema Nacional Español" (2) published by the Spanish Health Ministry,
after 6 months of an acute ischemic stroke,
26.1% of patients have died,
32.4% are dependent on some form of care and 41.5% are considered independent.
It is estimated that approximately 44% of stroke survivors in Spain have a functional disability (2).
The prognosis does not only depend on the severity of the ictus,
but on other factors such as age,
pre-existing conditions and co-morbidities,
and the sequelae of the stroke itself including epilepsy,
pain,
sleep apnea,
infections of the urinary tract,
heart arrhythmias,
etc.
(10).
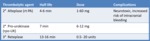
The current treatment paradigm states that if a patient presents within 4.5 hours of symptom onset he or she is treated with IV thrombolysis,
as long as there are no contraindications (Table 1,
Fig. 1).
For patients who meet exclusion criteria or arrive after the 4.5 hour window for IV thrombolysis has closed,
there are not many treatment alternatives available in most centers,
which significantly increased the risk of death or functional impairment.

Fig. 1: Table 1- Exclusion criteria for IV thrombolysis
Table 1- Exclusion criteria for IV thrombolysis.
In Spain,
there is a very severe shortage of specialized treatment centers for acute ischemic stroke beyond IV thrombolysis.
Only 48% of the Spanish provinces have one or more comprehensive stroke center.
The situation is especially grave in Andalucía,
where only 2 out of its 8 provinces have a stroke unit (7).
II- DIAGNOSIS
This is a brief summary of the clinical diagnosis of acute stroke,
since an extensive analysis is beyond the scope of this work.
The following references provide an excellent overview for those interested: "Australian Government.
National Stroke Foundation: Clinical Guidelines for Stroke Management 2010” (11); “Ministerio de Sanidad y Consumo de España: Estrategia en ictus del Sistema Nacional de Salud” (2) and “Sociedad Española de Neurroradiología: Guía de práctica clínica en neurorradiología: Ictus isquémico” (12).
These same references also describe in detail preventive treatments and rehabilitation programs for stroke patients.
Clinical diagnosis:
The acute stroke scale developed by the National Institute of Health (NIH) called the "National Institute of Health Stroke Scale" or NIHSS (Table 2,
Fig. 2) and the Glasgow Scale (Table 3,
Fig. 3) are the most commonly used to clinically determine the severity of a stroke.
If a patient survives,
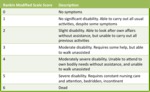
his or her level of disability is determined by the “Modified Rankin Scale" or mRS (Table 4,
Fig. 4).
Approximately 20% of acute stroke patients present with an NIHSS of 10 or more (10) (Table 5, Fig. 5) (13).
A good outcome is a mRS score of 2 or more.
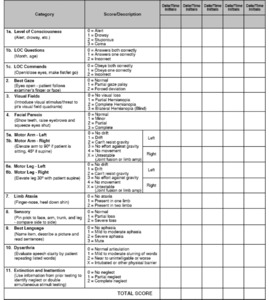
Fig. 2: Table 2- National Institute of Health Stroke Scale, courtesy of the National Institute of Neurological Disorders and Stoke.
References: National Institute of Neurological Disorders and Stroke
Table 2- National Institute of Health Stroke Scale,
courtesy of the National Institute of Neurological Disorders and Stoke.
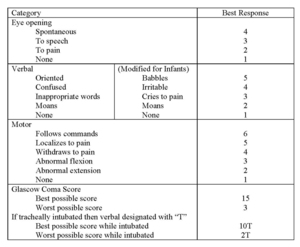
Fig. 3: Table 3- Glasgow Coma Scale, courtesy of the National Institute of Neurological Disorders and Stoke.
References: National Institute of Neurological Disorders and Stroke
Table 3- Glasgow Coma Scale,
courtesy of the National Institute of Neurological Disorders and Stoke.
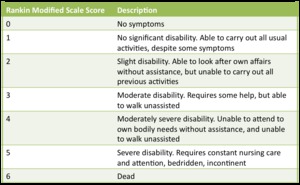
Fig. 4: Table 4- Modified Rankin Scale.
Table 4- Modified Rankin Scale.
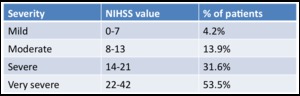
Fig. 5: Table 5- NIHSS of patients at presentation.
Table 5- Patients' NIHSS at presentation.
Radiological Diagnosis:
Once the patient arrives in the ED and the stroke code is activated,
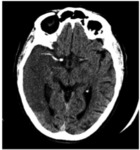
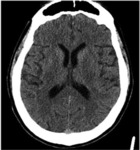
a non-enhanced CT of the brain is performed to rule out a hemorrhagic infarct or a completed hypodense infarct that affects more than 1/3 of a vascular territory,
both of which exclude the patient from thrombolytic treatment (Image 1,
Fig. 6 and Image 2,
Fig. 7.
In image 1 the hyperdense middle cerebral artery (MCA) sign can be appreciated).
In cases where a hypodense region is identified,
the size can be determined quantitatively using the Alberta Stroke Program Early CT Score (ASPECT),
although it must be noted that this method can be onerous,
and thus is not often utilized (1).
Readers interested in ASPECT can view the article by Warwick et al.
(14),
which explains this methodology in great detail.
If the patient does not have any radiological or clinical contraindication for thrombolytic treatment,
a stroke protocol is performed with angiography and perfusion,
either with CTA or MRI.
Currently CTA is the preferred method due to its rapid acquisition time and availability.
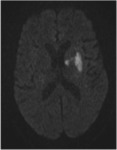
The advantage of utilizing MRI is that it easily permits identification of infracted areas in diffusion-weighted sequences (DWI),
although it important to know that hyperacute infarcts of one hour or less may not be seen in DWI images.
The greatest disadvantages of MRI are its long acquisition time (in acute stroke “time is brain”),
and its reduced availability in medical centers as compared to CT.
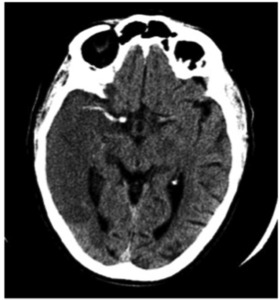
Fig. 6: Image 1 – Axial unenhanced CT shows greater that 1/3 hypodensity in the right MCA vascular territory secondary to a thrombus that occludes the artery. This radiological finding in known as the “MCA hyperdense sign”.
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 1 – Axial unenhanced CT shows greater that 1/3 hypodensity in the right middle cerebral artery (MCA) vascular territory secondary to a hyperdense thrombus that occludes the artery.
This radiological finding in known as the “MCA hyperdense sign”.
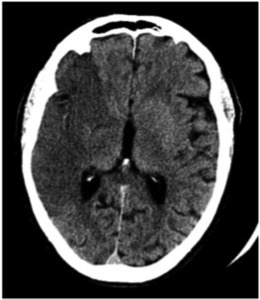
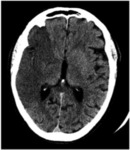
Fig. 7: Image 2 – Axial unenhanced CT shows greater that 1/3 hypodensity in the right MCA vascular territory.
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 2 – Axial unenhanced CT shows greater that 1/3 hypodensity in the right MCA vascular territory.
Once the images are acquired,
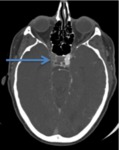
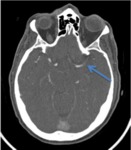
the angiographic portion of the study is evaluated to identify the presence of thrombi (Image 3,
Fig. 8,
thrombus in the right supraclinoid interior carotid artery (ICA),
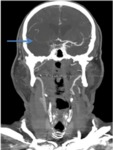
blue arrow; Image 4,
Fig. 9 thrombus that occludes the right ACM,
blue arrow).
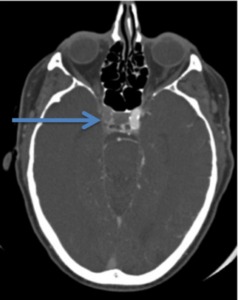
Fig. 8: Image 3- Axial CTA. Thrombus in the right supraclinoid interior carotid artery (ICA), indicated with the blue arrow.
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 3- Axial CTA.
Thrombus in the right supraclinoid interior carotid artery (ICA),
indicated with the blue arrow.
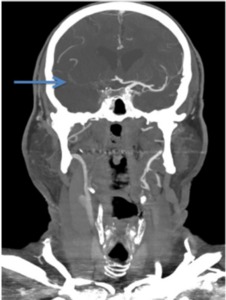
Fig. 9: Image 4- Coronal CTA. Thrombus in the entire course of the M1 segment of the right MCA, indicated with the blue arrow.
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 4- Coronal CTA.
Thrombus in the entire course of the M1 segment of the right MCA,
indicated with the blue arrow.
After identifying the location of the thrombus (if it is present),
the perfusion images are evaluated.
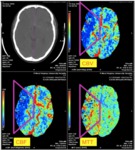
There are three perfusion maps and/or parameters that are utilized to determine the presence and size of the region of ischemia.
The first parameter is "cerebral blood flow” (CBF),
which determines the velocity of blood flow,
primarily in the main arteries; the second parameter is "mean transit time” (MTT),
which measures the time it takes the contrast bolus to pass from the arterial to the venous phase,
or in other words,
the time it takes to perfuse a vascular territory and; the third parameter known as "cerebral blood volume” (CBV) determines the volume of blood that perfuses a brain region,
including the main arteries and collaterals.
The relationship between these three parameters is (15):
MTT (min)= CBF (ml/g/min) ÷ CBV (ml/g)
It is recommended that the first map analyzed is the MTT,
since it is the most sensitive to perfusion alterations.
The size of the perfusion defect is then calculated in each of the three perfusion maps.
The two most important maps to compare are CBV and CBF.
If the size of the perfusion defects are similar ("matching defects",
Image 5,
Fig. 10),
then that means there is no significant collateral circulation in the brain region affected,
and thus no salvageable brain parenchyma is present (1). This is the case because not enough collateral circulation is perfusing the ischemic brain (which would be seen as preserved or minimally compromised blood volume in the CBV map).
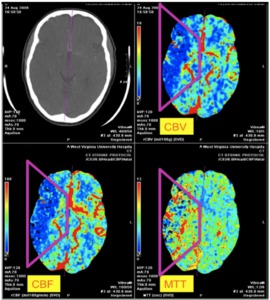
Fig. 10: Image 5- CTA perfusion maps. "Matching defect" in the right MCA territory with similar sized perfusion defects in the three perfusion maps: MTT, CBV and CBF.
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 5- CTA perfusion maps.
"Matching defect" in the right MCA territory with similar sized perfusion defects in the three perfusion maps: MTT,
CBV and CBF.
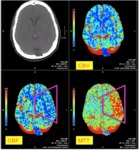
If,
on the contrary,
a patient with stroke symptoms does not present with a hypodense region on the unenhanced CT greater that 1/3 of the vascular territory (Image 6,
Fig. 11),
demonstrates an intraluminal thrombus in CTA (Image 7,
Fig. 12,
blue arrow),
and the defect in the CBV map is smaller that the other two perfusion maps,
especially the CBF map,
then collateral flow exist,
the ischemic tissue is partially perfused and thus is salvageable (1) ("mismatch defect",
Image 8,
Fig. 13.
Note complete preservation of CBV on the corresponding perfusion map).
When a mismatch defect exist it can also be described as a penumbra.
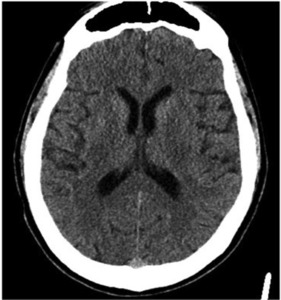
Fig. 11: Image 6- Axial unenhanced CT. No hypodensity or loss of grey-white matter differentiation appreciated.
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 6- Axial unenhanced CT.
No hypodensity or loss of grey-white matter differentiation appreciated.
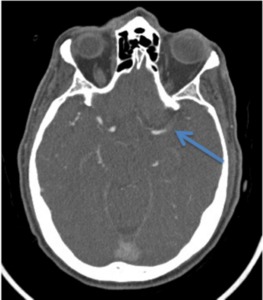
Fig. 12: Image 7- Axial CTA. Thrombus in the course of the M1 segment of the left MCA, indicated with the blue arrow.
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 7- Axial CTA.
Thrombus in the course of the M1 segment of the left MCA,
indicated with the blue arrow.
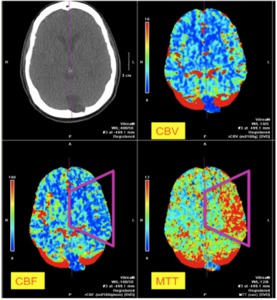
Fig. 13: Image 8- "Mismatching defect" in the territory of the left ACM. MTT and CBF demonstrate a perfusion defect that is not seen in the CBV map, thus a penumbra exists and the ischemic tissue can be salvageable
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 8- "Mismatching defect" in the territory of the left ACM.
MTT and CBF demonstrate a perfusion defect that is not seen in the CBV map,
thus a penumbra exists and the ischemic tissue might be salvageable.
III- INTRA-ARTERIAL (IA) TREATMENT
In patients that do not meet the requirements for IV thrombolysis because they present outside the 4.5 hour window,
or in patients that do not recanalize after IV thrombolysis,
IA thrombolysis is a treatment option.
Based on the incidence of stoke in the US,
it is estimated that only 2.5% of cases are eligible for IA thrombolysis (20,000 out of 795,000 annual cases) (8).
The general indications for IA thrombolysis include patients that present to the ED 4.5 to 12 hours after the onset of symptoms of stroke with occlusion of main anterior circulation arteries or between 4.5 to 24 hours after the onset of symptoms of stroke with occlusion of main posterior circulation arteries.
Patients should have an NIHSS of 10 to 25 (i.e.
significant neurological deficit),
as it has been determined that patients with an NIHSS score of less than 10 do not benefit significantly from IA thrombolysis (8,
16).
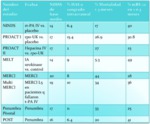
This treatment has been validated in multiple past (Table 6,
Fig. 14) and present (Table 7,
Fig. 15) trials,
and is recommended by the American Stroke Association,
the American Heart Association and the American College of Chest Physicians as an appropriate treatment for carefully selected patients.
For an extensive list of all the prior and current clinical investigations on this subject,
the reader should visit the Internet Stoke Center’s webpage: www.strokecenter.org.
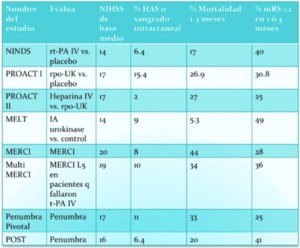
Fig. 14: Table 6 – Prior clinical trials evaluating IA and mechanical thrombolysis.
Table 6 – Prior clinical trials evaluating IA and mechanical thrombolysis.
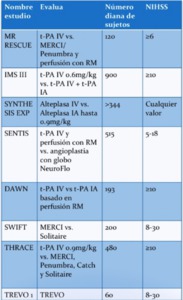
Fig. 15: Table 7- Current clinical trials evaluating IA and mechanical thrombolysis.
Table 7- Current clinical trials evaluating IA and mechanical thrombolysis.
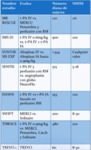
Intra-arterial thrombolysis is carried out in two ways: 1) with thrombolytic chemical agents that work in the plasminogen to plasmin coagulation cascade,
which degrades the fibrin found within the thrombi or 2) by mechanical means of thrombus extraction.
These two forms of intra-arterial thrombolysis can be used separately or together.
All treatments are administered through a microcatheter lodged within a guiding catheter.
First generation thrombolytics (urokinase and streptokinase) which activate plasminogen are currently not utilized as they resulted in hipofibrinogenemia,
which significantly increased the risk of massive hemorrhage.
Second generation thrombolytics (alteplase and pro-urokinase) are not antigenic and function by turning the pro-enzyme plasminogen into active plasmin,
although only alteplase is used in IA thrombolysis.
Third generation thrombolytics (reteplase and tenecteplase) are more potent than second generation ones because they have a longer half-life.
Only reteplase is FDA approved for IA thrombolysis.
(6,
19,
20)- see Table 8,
Fig. 16.

Fig. 16: Table 8- Second and third generation IA thrombolytic agents.
Table 8- Second and third generation IA thrombolytic agents.
The mechanical methods of thrombus extraction are especially useful because they forgo the need to use a chemical thrombolytic agent,
thus decreasing the risk of clinically significant hemorrhages.
The disadvantages are that there is a greater risk of vessel dissection,
perforation or tear,
or of proximal or distal emboli shower from the mechanical disruption of the thrombus (6,
21).
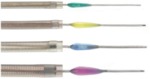
The devices most currently used and clinically validated are the MERCI (Image 9,
Fig. 17),
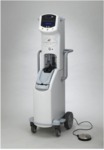
Penumbra (Image 10, Fig. 18 and video),
Solitare and AngioJet (Image 11,
Fig. 19).
Only MERCI and Penumbra are FDA approved for IA thrombolysis (at the time of writing),
although AngioJet has an “off-label” use designation.
MERCI (Concentric Medical,
Mountain View,
CA,
USA): It was the first mechanical device built for the mechanical extraction of intracranial arterial thrombi.
It received FDA approval in 2004 and has had multiple versions,
the latest one being series V.
Series V has,
in comparison to its spiral predecessors,
variable tension loops that allow more efficient capture of thrombi.
The device goes through the thrombus,
which is trapped within the variable tension loops,
and then is retracted into the microcatheter,
allowing for mechanical extraction of the blood clot.
A balloon is inflated just distal to the guide catheter to avoid a retrograde emboli shower.

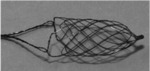
Fig. 17: Image 9- MERCI device series V. Courtesy of Dr. Thanh Nguyen.
References: Thanh N. Nguyen, Viken L. Babikian et al. Intra-arterial treatment methods in acute stroke therapy. Front Neurol 2011; 2:9.
Image 9- MERCI device series V.
Courtesy of Dr.
Thanh Nguyen.
Thanh N.
Nguyen,
Viken L.
Babikian et al.
Intra-arterial treatment methods in acute stroke therapy.
Front Neurol 2011; 2:9.
SOLITAIRE (ev3,
Plymouth,
Minnesota,
USA): It utilizes a mechanism similar to MERCI,
as it uses an uncovered stent with a microcatheter,
trapping the thrombus within its lumen.
PENUMBRA (Penumbra,
Alameda,
CA,
USA): It is a device that combines thrombus aspiration and mechanical extraction.
It was approved by the FDA for IA thrombolysis in 2008.
Similar to the MERCI device,
it is introduced within the mass of the thrombus,
utilizing a combination of both aspiration and mechanical extraction.
Like MERCI,
a balloon is inflated just distal to the guide catheter to avoid a proximal retrograde emboli shower.
Currently (at the time of writing) there are four different sizes available (0.054”,
0.041”,
0.032” and 0.026”) that allow for different sized vessels to be treated.
It should be mentioned that the smaller the catheter size,
the smaller the aspiration power (6).
Link to the online video here,
courtesy of Penumbra Inc.: http://www.penumbrainc.com/site.asp?content_id=158&menu_id=152&category=products_na&sub_menu_id=154&click2=158
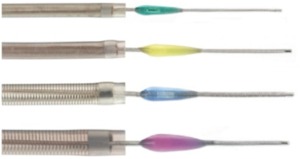
Fig. 18: Image 10- Penumbra device in four different sized. Courtesy of Penumbra, Inc.
References: Penumbra, Inc.
Image 10- Penumbra device in four different sized.
Courtesy of Penumbra,
Inc.
ANGIOJET (Possis Medical,
Inc.,
Minneapolis,
MN,
USA): It directs jets of high-pressure saline water towards the thrombus at the same time as it suctions,
aspirating the clot through the microcatheter.
It is currently (at time of writing) not FDA approved for IA thrombolysis,
although it does have an off-label use designation.
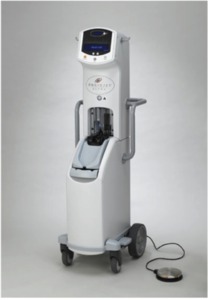
Fig. 19: Image 11- AngioJet device. Courtesy of Medrad Interventional / Possis.
References: Medrad Interventional / Possis
Image 11- AngioJet device.
Courtesy of Medrad Interventional / Possis.
There are other mechanical extraction apparatuses in the market that at time of writing had not been FDA approved for IA thrombolysis,
and that have not been as extensively used as the previously mentioned devices.
Some of these products are the CATCH Thromboembolectomy System (Balt Extrusion,
Montmorency,
France,
Image 12,
Fig. 20); multiple dispositives from Phenox,
among them the Phoenox clot retriever,
the CRC clot retriever CAGE,
BONNET,
BONNET short and pREset (Phenox,
Bochum,
Germany); the Alligator (Chestnut Medical Technologies,
Menlo Park,
CA,
USA; and the TREVO (Concentric Medical,
Mountain View,
CA,
USA),
which has a functionality and design similar to the SOLITAIRE.
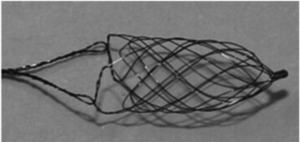
Fig. 20: Image 12- CATCH device. Courtesy of BALT Extrusion.
References: BALT Extrusion
Image 12- CATCH device.
Courtesy of BALT Extrusion.
Similarly,
there have been clinical trials using balloon devices for angioplasty (Hyperglide,
eV3,
Irvine CA,
USA and Gateway,
Boston Scientific,
Fremont CA,
USA,
among others),
although they have not been very effective,
as there is a high incidence of re-stenosis and they must be used in conjunction with chemical thrombolytic agents (6).
Finally,
stents have been used to treat acute ischemic stroke patients,
using such devices as the Enterprise Stent (Cordis,
Raynham,
MA,
USA,
Image 13,
Fig. 21) among others.
The downside of stents is that they require lifelong anticoagulation,
and there are no prospective studies on the long-effects and morbidities associated with permanent intracranial stent placement (6).
Thus it is preferable to use removable stents for treatment of acute ischemic stroke patients using devices similar to the Solitaire or TREVO.
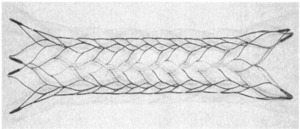
Fig. 21: Image 13- Enterprise Stent, courtesy of Cordis, Inc.
References: Cordis, Inc.
Image 13- Enterprise Stent,
courtesy of Cordis,
Inc.
The clinical results are promising,
although it is necessary to validate the results in a greater population of patients.
In the clinical trials done up to this point,
patients treated with IA thrombolysis had a 35% higher rate of complete or almost complete recuperation (NIHSS 0-1) at 90 days post-treatment in comparison to the control population (16,
23).
In a meta-analysis of five randomized clinical trials,
IA thrombolysis was associated to a better clinical response of mRS 0-2 (43 vs.
28%; OR 2,
95% CI 1.3- 3.1; NNT 6.8) and an almost complete clinical response of mRS 0-1 (31 vs.
18%; OR 2.1; 95% CI 1.3-3.5; NNT 7.7) as compared to the control group (6,
24).
Obviously these invasive treatments are not benign.
It is estimated that approximately 10% of patients treated with IA thrombolysis develop a clinically significant intracranial bleed,
and of these,
50% are deadly (6,25).
Clinically significant complications are seen in 5-7% of patients beyond the intracranial hemorrhages.
These treatments can also cause reperfusion injury that can result in significant intracranial edema,
hemorrhage or death (16).
As was mentioned before,
with the mechanical devices there is an increased risk of vessel dissection,
puncture or tear,
of proximal or distal emboli shower,
or of subarachnoid hemorrhage secondary to vessel trauma,
which have been observed during clinical trials with MERCI and Penumbra devices (26).
In the original MERCI clinical trial a 4.3% incidence of vessel perforation was observed (26).
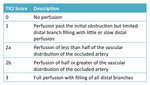
The angiographic result scale used to evaluate IA treatment sucess is called the Thrombolysis in Cerebral Infarction (TICI,
Table 9,
Fig. 22),
adapted from the Thrombolysis in Myocardial Infarction (TIMI) scale used in cardiac catheterization.
A good angiographic result is a TICI of 2b or 3.

Fig. 22: Table 9- Thrombolysis in Cerebral Infarction angiographic response scale.
Table 9- Thrombolysis in Cerebral Infarction angiographic response scale.
The patient with the left MCA clot that we saw in Images 6-8 presented outside the window of treatment with IV thrombolysis.
He woke up with his stroke symptoms and the exact time of stroke onset could not be determined,
although 12 hours had not passed since he was last seen normal.
A stroke code was activated for IA thrombolysis.
A diagnostic angiogram was performed (Image 14,
Fig. 23) that confirmed the presence of a left MCA thrombus with lack of distal perfusion.
After mechanical thrombus removal with a MERCI device,
blood flow TICI 3 is re-established (Image 15,
Fig. 24 and Image 16,
Fig. 25).
A post treatment MRI (Image 17,
Fig. 26) showed the small amount of infracted tissue as compared to the compromised area of perfusion seen on his CBF maps (Image 8,
Fig. 13).
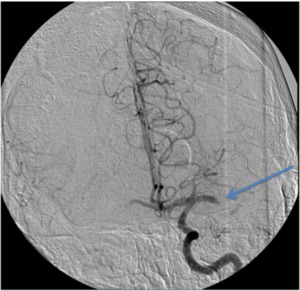
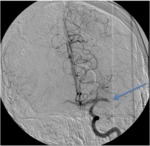
Fig. 23: Image 14: Catheter angiogram AP projection shows a left MCA thrombus with absence of distal flow, blue arrow.
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 14: Catheter angiogram AP projection shows a left MCA thrombus with absence of distal flow,
blue arrow.
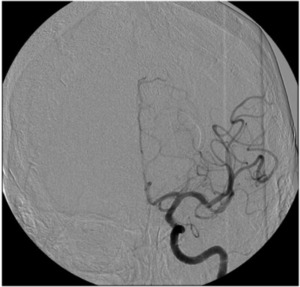
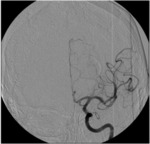
Fig. 24: Image 15- Catheter angiogram AP projection shows restored blood flow to the M2 and M3 branches of the left MCA after mechanical thrombus extraction with the MERCI device. TICI score of 3.
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 15- Catheter angiogram AP projection shows restored blood flow to the M2 and M3 branches of the left MCA after mechanical thrombus extraction with the MERCI device.
TICI score of 3.
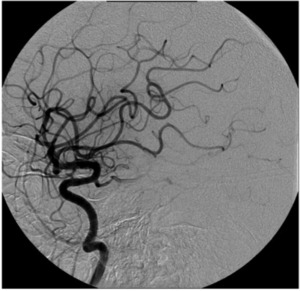
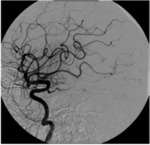
Fig. 25: Image 16- Catheter angiogram lateral projection shows restored blood flow to the M2 and M3 branches of the left MCA after mechanical thrombus extraction with the MERCI device. TICI score of 3.
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 16- Catheter angiogram lateral projection shows restored blood flow to the M2 and M3 branches of the left MCA after mechanical thrombus extraction with the MERCI device.
TICI score of 3.
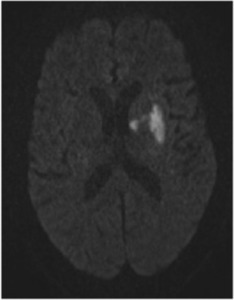
Fig. 26: Image 17- MR DWI axial image post treatment demonstrates the final region of infarct, which is significantly smaller than the regions of abnormal perfusion seen on CBF and MTT in Image 8.
References: Dr. Jeffery Hogg, Radiology Department, West Virginia University
Image 17- MR DWI axial image post treatment demonstrates the final region of infarct,
which is significantly smaller than the regions of abnormal perfusion seen on CBF and MTT in Image 8.
IV- BRIDGING THERAPY
IV thrombolysis has been validated in multiple clinical trials as clearly beneficial in most cases of acute ischemic stroke,
but it has demonstrated a limited benefit in patients with large clots within the main intracranial vessels,
even when they present within the 4.5 hour window (18).
In these patients,
as well as in patients treated with IA thrombolysis,
there is a movement towards “bridging therapy” that combines both IV and IA chemical thrombolysis or IV and mechanical IA thrombolysis.
Currently,
bridging treatments are being evaluated by the Interventional Management Stroke Trial III (IMS III),
a Phase III trial,
where low dose IV t-PA is followed by IA thrombolysis.
In the MR RESCUE protocol,
patients with failed IV thrombolysis received within a 4.5 hour window that present with persistent thrombus in a subsequent MRI are then treated with a mechanical extraction device within an 8 hour window.
V- STROKE CENTERS
In Spain (2) as well as in the US (27),
there are guidelines to integrate comprehensive treatment for stroke patients that include both acute interventions in a dedicated critical care stroke unit and chronic rehabilitation care.
As well,
some of these centers have a public health and educational role,
providing the general public with stroke prevention campaign as well as training emergency medical personal on how to provide the key hyperacute out-of-hospital stroke care (28).
In the US there are two types of designations that allow medical centers to treat stroke patients.
They are known as "Primary Stroke Centers” and "Comprehensive Stroke Center" (29).
The first designation is for more basic type of stroke care,
as its name implies,
while the second designation is what is properly known as a stroke center.
There are multiple differences between them.
The most important are that in a primary stroke center there is no IA thrombolysis treatment offered and there is no interventional neuroradiologist on call 24/7 (8) (Table 10).
In Spain,
as we mentioned in the introduction,
there are stroke centers similar to the Comprehensive Stroke Centers established in the US,
and the requirements to set up such units are described in the "Guía de Práctica Clínica en Neurorradiología - Ictus Isquémico" from the “Sociedad Española de Neurorradiología” (12) and in the "Estrategias en Ictus del Sistema Nacional Español" from the Spanish Health Ministry (2).
Unfortunately,
only in Catalonia,
Spain is there an autonomous community wide system to transfer patients from primary to comprehensive stroke centers (Oral Presentation,
VII Curso de Neurorradiología,
Lleida,
España).
As we already mentioned,
there are only two provinces in Andalucía with an advanced stroke center,
a deficiency which should be remedied in the shortest time possible,
although Andalucía's autonomous government,
the Junta de Andalucía,
has begun this process by implementing the "Plan Andaluz de Ataque Cerebral Agudo (PLACA)” (30).
In the US,
there is a central system that helps medical centers acquire and develop the necessary infrastructure to establish either primary or comprehensive stroke centers (26,
27,
31).
Once these stroke centers are established,
they are certified by the Joint Commission (32) in collaboration with the American Stroke Association and the American Heart Association (“Get with the Guidelines Program”) (28).
As well,
there are rigorous requirements for the training of the interventional neuroradiologists that work in these centers -please see the article by J.J.
Connors et al (17).
|
Requirements
|
Primary Center
|
Comprehensive Center
|
|
Stroke team
|
√
|
√
|
|
Stroke treatment algorithm established
|
√
|
√
|
|
ED
|
√
|
√
|
|
Dedicate critical care stroke unit
|
√
|
√
|
|
Neurosurgery
|
√
|
√
|
|
Neuroradiology (24/7)
|
√
|
√
|
|
Laboratory (24/7)
|
√
|
√
|
|
Quality Assurance
|
√
|
√
|
|
Continuing Medical Education
|
√
|
√
|
|
Coordination with 911 / EMS services
|
√
|
√
|
|
Minimum number of cases per year of the following: 20 subarachnoid bleeds,
10 craniotomies for aneurysm treatments,
15 neurointerventional aneurysm treatments,
25 IV thrombolysis treatments)
|
|
√
|
|
Interventional Neuroradiology (24/7)
|
|
√
|
|
Carotid and transcranial Dupplex
|
|
√
|
|
Echocardiogram,
transthoracic and transesophageal
|
|
√
|
|
Angio-MR (24/7)
|
|
√
|
|
Research Program
|
|
√
|
|
Peer-review
|
|
√
|
|
Outpatient rehabilitation coordination
|
|
√
|
Table 10- Requirements for US Primary and Comprehensive Stroke Centers.